Abstract
Purpose
This study aimed to determine the safety, tolerability, and recommended phase II doses of trametinib plus uprosertib (GSK2141795) in patients with solid tumors likely to be sensitive to MEK and/or AKT inhibition.
Methods
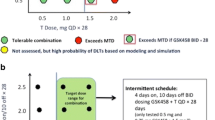
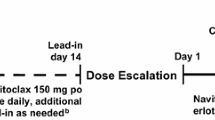
This was a phase I, open-label, dose-escalation, and dose-expansion study in patients with triple-negative breast cancer or BRAF-wild type advanced melanoma. The primary outcome of the expansion study was investigator-assessed response. Among 126 enrolled patients, 63 received continuous oral daily dosing of trametinib and uprosertib, 29 received various alternative dosing schedules, and 34 were enrolled into expansion cohorts. Doses tested in the expansion cohort were trametinib 1.5 mg once daily (QD) + uprosertib 50 mg QD.
Results
Adverse events (AEs) were consistent with those reported in monotherapy studies but occurred at lower doses and with greater severity. Diarrhea was the most common dose-limiting toxicity; diarrhea and rash were particularly difficult to tolerate. Overall, 59% and 6% of patients reported AEs with a maximum severity of grade 3 and 4, respectively. Poor tolerability prevented adequate delivery of uprosertib with trametinib at a concentration predicted to have clinical activity. The study was terminated early based on futility in the continuous-dosing expansion cohorts and a lack of pharmacological or therapeutic advantage with intermittent dosing. The objective response rate was < 5% (1 complete response, 5 partial responses).
Conclusions
Continuous and intermittent dosing of trametinib in combination with uprosertib was not tolerated, and minimal clinical activity was observed in all schedules tested.


Similar content being viewed by others
References
Burotto M, Chiou VL, Lee JM, Kohn EC (2014) The MAPK pathway across different malignancies: a new perspective. Cancer 120:3446–3456. https://doi.org/10.1002/cncr.28864
Wee S, Jagani Z, Xiang KX, Loo A, Dorsch M, Yao YM et al (2009) PI3K pathway activation mediates resistance to MEK inhibitors in KRAS mutant cancers. Cancer Res 69:4286–4293. https://doi.org/10.1158/0008-5472.CAN-08-4765
Lim SY, Menzies AM, Rizos H (2017) Mechanisms and strategies to overcome resistance to molecularly targeted therapy for melanoma. Cancer 123:2118–2129. https://doi.org/10.1002/cncr.30435
Hu Y, Gu Y, Wang H, Huang Y, Zou YM (2015) Integrated network model provides new insights into castration-resistant prostate cancer. Sci Rep 5:17280. https://doi.org/10.1038/srep17280
Liu Y, Sheikh MS (2014) Melanoma: molecular pathogenesis and therapeutic management. Mol Cell Pharmacol 6:228
Lan YT, Jen-Kou L, Lin CH, Yang SH, Lin CC, Wang HS et al (2015) Mutations in the RAS and PI3K pathways are associated with metastatic location in colorectal cancers. J Surg Oncol 111:905–910. https://doi.org/10.1002/jso.23895
Greger JG, Eastman SD, Zhang V, Bleam MR, Hughes AM, Smitheman KN et al (2012) Combinations of BRAF, MEK, and PI3K/mTOR inhibitors overcome acquired resistance to the BRAF inhibitor GSK2118436 dabrafenib, mediated by NRAS or MEK mutations. Mol Cancer Ther 11:909–920. https://doi.org/10.1158/1535-7163.MCT-11-0989
McCubrey JA, Steelman LS, Chappell WH, Abrams SL, Franklin RA, Montalto G et al (2012) Ras/Raf/MEK/ERK and PI3K/PTEN/Akt/mTOR cascade inhibitors: how mutations can result in therapy resistance and how to overcome resistance. Oncotarget 3:1068–1111. https://doi.org/10.18632/oncotarget.659
Sanchez-Hernandez I, Baquero P, Calleros L, Chiloeches A (2012) Dual inhibition of (V600E)BRAF and the PI3K/AKT/mTOR pathway cooperates to induce apoptosis in melanoma cells through a MEK-independent mechanism. Cancer Lett 314:244–255. https://doi.org/10.1016/j.canlet.2011.09.037
Tolcher AW, Khan K, Ong M, Banerji U, Papadimitrakopoulou V, Gandara DR et al (2015) Antitumor activity in RAS-driven tumors by blocking AKT and MEK. Clin Cancer Res 21:739–748. https://doi.org/10.1158/1078-0432.CCR-14-1901
Tolcher AW, Patnaik A, Papadopoulos KP, Rasco DW, Becerra CR, Allred AJ et al (2015) Phase I study of the MEK inhibitor trametinib in combination with the AKT inhibitor afuresertib in patients with solid tumors and multiple myeloma. Cancer Chemother Pharmacol 75:183–189. https://doi.org/10.1007/s00280-014-2615-5
Flaherty KT, Robert C, Hersey P, Nathan P, Garbe C, Milhem M et al (2012) Improved survival with MEK inhibition in BRAF-mutated melanoma. N Engl J Med 367:107–114. https://doi.org/10.1056/NEJMoa1203421
Mekinist (trametinib) [prescribing information] Novartis Pharmaceuticals Corporation East Hanover, NJ, USA. 2018. Accessed 30 July 2019
Pachl F, Plattner P, Ruprecht B, Médard G, Sewald N, Kuster B (2013) Characterization of a chemical affinity probe targeting Akt kinases. J Proteome Res 12:3792–3800. https://doi.org/10.1021/pr400455j
Dumble M, Crouthamel MC, Zhang SY, Schaber M, Levy D, Robell K et al (2014) Discovery of novel AKT inhibitors with enhanced anti-tumor effects in combination with the MEK inhibitor. PLoS ONE 9:e100880. https://doi.org/10.1371/journal.pone.0100880
Gungor H, Saleem A, Babar S, Dina R, El-Bahrawy MA, Curry E et al (2015) Dose-finding quantitative 18F-FDG PET imaging study with the oral pan-AKT inhibitor GSK2141795 in patients with gynecological malignancies. J Nucl Med 56:1828–1835. https://doi.org/10.2967/jnumed.115.156505
Data on file (2015). Study TAC113886. https://www.gsk-clinicalstudyregister.com. Accessed 16 Feb 2016
Infante JR, Fecher LA, Falchook GS, Nallapareddy S, Gordon MS, Becerra C et al (2012) Safety, pharmacokinetic, pharmacodynamic, and efficacy data for the oral MEK inhibitor trametinib: a phase 1 dose-escalation trial. Lancet Oncol 13:773–781. https://doi.org/10.1016/S1470-2045(12)70270-X
Burris HA, Siu LL, Infante JR, Wheler JJ, Kurkjian C, Opalinska J et al (2011) Safety, pharmacokinetics (PK), pharmacodynamics (PD), and clinical activity of the oral AKT inhibitor GSK2141795 (GSK795) in a phase I first-in-human study. J Clin Oncol 29:3003. https://doi.org/10.1200/jco.2011.29.15_suppl.3003
Bedard PL, Tabernero J, Janku F, Wainberg ZA, Paz-Ares L, Vansteenkiste J et al (2015) A phase Ib dose-escalation study of the oral pan-PI3K inhibitor buparlisib (BKM120) in combination with the oral MEK1/2 inhibitor trametinib (GSK1120212) in patients with selected advanced solid tumors. Clin Cancer Res 21:730–738. https://doi.org/10.1158/1078-0432.CCR-14-1814
Huang X, Biswas S, Oki Y, Issa JP, Berry DA (2007) A parallel phase I/II clinical trial design for combination therapies. Biometrics 63:429–436. https://doi.org/10.1111/j.1541-0420.2006.00685.x
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R et al (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45:228–247. https://doi.org/10.1016/j.ejca.2008.10.026
Green SJ, Dahlberg S (1992) Planned versus attained design in phase II clinical trials. Stat Med 11:853–862. https://doi.org/10.1002/sim.4780110703
Algazi AP, Moon J, Chmielowski B, Lo R, Kendra KL, Lao CD et al (2017) SWOG S1221: a phase 1 dose escalation study co-targeting MAPK-dependent and MAPK-independent BRAF inhibitor resistance in BRAF mutant advanced solid tumors with dabrafenib, trametinib, and GSK2141795 (ClinicalTrials.gov NCT01902173). J Clin Oncol 35:2578. https://doi.org/10.1200/JCO.2017.35.15_suppl.2578
Do K, Speranza G, Bishop R, Khin S, Rubinstein L, Kinders RJ et al (2015) Biomarker-driven phase 2 study of MK-2206 and selumetinib (AZD6244, ARRY-142886) in patients with colorectal cancer. Investig New Drugs 33:720–728. https://doi.org/10.1007/s10637-015-0212-z
Tolcher AW, Baird RD, Patnaik A, Garcia VM, Papadopoulos KP, Garrett CR et al (2011) A phase I dose-escalation study of oral MK-2206 (allosteric AKT inhibitor) with oral selumetinib (AZD6244; MEK inhibitor) in patients with advanced or metastatic solid tumors. J Clin Oncol 29:3004. https://doi.org/10.1200/jco.2011.29.15_suppl.3004
Yap TA, Omlin A, de Bono JS (2013) Development of therapeutic combinations targeting major cancer signaling pathways. J Clin Oncol 31:1592–1605. https://doi.org/10.1200/JCO.2011.37.6418
Subbiah V, Sen S, Hess KR, Janku F, Hong DS, Khatua S et al (2018) Phase I study of the BRAF inhibitor vemurafenib in combination with the mammalian target of rapamycin inhibitor everolimus in patients with BRAF-mutated malignancies. JCO Precis Oncol 2:1–12. https://doi.org/10.1200/PO.18.00189
Hoeflich KP, Merchant M, Orr C, Chan J, Den Otter D, Berry L et al (2012) Intermittent administration of MEK inhibitor GDC-0973 plus PI3K inhibitor GDC-0941 triggers robust apoptosis and tumor growth inhibition. Cancer Res 72:210–219. https://doi.org/10.1158/0008-5472.CAN-11-1515
Wang X, Guda C (2016) Integrative exploration of genomic profiles for triple negative breast cancer identifies potential drug targets. Medicine (Baltimore) 95:e4321. https://doi.org/10.1097/MD.0000000000004321
Bose R, Kavuri SM, Searleman AC, Shen W, Shen D, Koboldt DC et al (2013) Activating HER2 mutations in HER2 gene amplification negative breast cancer. Cancer Discov 3:224–237. https://doi.org/10.1158/2159-8290.CD-12-0349
Song MS, Salmena L, Pandolfi PP (2012) The functions and regulation of the PTEN tumour suppressor. Nat Rev Mol Cell Biol 13:283–296. https://doi.org/10.1038/nrm3330
Acknowledgements
The authors thank all participating patients and their families. The authors also thank William Fazzone, Ph.D., of ArticulateScience, LLC, for editorial assistance, which was funded by Novartis Pharmaceuticals Corporation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
A. W. T. has received fees for consulting and board memberships, as well as research funding (paid to NEXT Oncology) from AbbVie, ADC Therapeutics, Adagene, Agenus, Aminex, AroBioTX, Arrys, Asana, Ascentage, Aximmune, Bayer, Biolnvent, Birdie, Boston Bio, CStone, Deciphera, EMD Serono, Forbius, GSK, HBM Partners, Ignyta, Inhibrx, Immunome, Immunomet, Innate, Jazz Pharmaceuticals, Kiromet, Mekanistic, Mersana, Nanobiotix, NatureWise, NBE Therapeutics, NextCure, Nitto Biopharma, Nuvalent, Pelican, Pfizer, Pierre Fabre, Pieris, Ridgeway, Scitemex, Sesen Bio, Seattle Genetics, Sunshine Guojian, Symphogen, Syndax, Syneos, Tizone, and Zymeworks. R. K. has received research funding from Boehringer Ingelheim, DeBiopharm, Foundation Medicine, Genentech, Grifols, Guardant Health, Incyte, Konica Minolta, Merck Serono, OmniSeq, Pfizer, and Sequenom; has consulting or advisory role in Actuate Therapeutics, Gaido, LOXO, NeoMed, Pfizer, Roche, Soluventis, and X-Biotech; has received speaker fees from Roche; is a board member of CureMatch, Inc.; and has stock and other equity interests in CureMatch, Inc., IDbyDNA, and Soluventis. V. V. received grant and honorarium from Novartis. R. G. received research funding from Novartis, GSK and grant and personal fees from Array, Bristol-Myers Squibb, Merck, and Roche/Genentech. R. S. H. received consulting honoraria from Boehringer Ingelheim, Novartis, Tarveda, Apollomics and received research funding (to his institution) from Daiichi Sankyo, Agios, Novartis, Corvus, Mirati, Millennium, Genentech Roche, AbbVie, Exelixis, Celgene, and Incyte. A. R. T. received research funding from GSK. C. W. and S. K-S. are employees of GSK. C. L. and L. Y. were former employees of GSK. J. F. K. was an employee of Novartis. J. G. and A. M. D. Jr. are employees of Novartis Pharmaceuticals and have stock ownership in Novartis and GSK. C. E. is an employee and stock owner of GSK. N. I. was an employee of GSK and currently is an employee of Merck. All remaining authors have declared no conflicts of interest. All authors received assistance with manuscript preparation from ArticulateScience, LLC.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tolcher, A.W., Kurzrock, R., Valero, V. et al. Phase I dose-escalation trial of the oral AKT inhibitor uprosertib in combination with the oral MEK1/MEK2 inhibitor trametinib in patients with solid tumors. Cancer Chemother Pharmacol 85, 673–683 (2020). https://doi.org/10.1007/s00280-020-04038-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-020-04038-8