Abstract
Aim
Despite current treatments, high-grade meningiomas continue to have a poor prognosis. Immunotherapy targeting immune checkpoints, such as PD-L1, has demonstrated significant success in controlling numerous malignancies. In this study, we investigate the extent of systemic and local immunosuppression in meningiomas to assess the potential benefit of immune checkpoint inhibitors for the treatment of high-grade meningiomas.
Methods
Peripheral blood was collected from patients undergoing resection of meningiomas (WHO grade I, n = 18; grade II, n = 25; grade III, n = 10). Immunosuppressive myeloid cells (CD45+CD11b+PD-L1+), myeloid-derived suppressor cells (MDSCs) (CD11b+CD33+HLA-DRlow), and regulatory T cells (Tregs) (CD3+CD4+CD25+FoxP3+) were quantified through flow cytometry. Tissue sections from the same patients were assessed for PD-L1 expression and T cell infiltration via immunohistochemistry.
Results
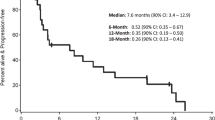
Patients with grade III meningiomas demonstrated increased peripheral monocyte PD-L1 compared to patients with grade I/II meningiomas and healthy controls. Peripheral MDSC abundance was increased in grades II and III meningioma patients. PD-L1 staining of meningioma tissue demonstrated increased positivity in grade III meningiomas. Intratumoral PD-L1 was not associated with progression-free survival. High-grade meningiomas had increased T-cell infiltration. However, a significant proportion of these T cells were exhausted PD1+ T cells and immunosuppressive Tregs.
Conclusions
Patients with meningiomas exhibit signs of peripheral immunosuppression, including increased PD-L1 on myeloid cells and elevated MDSC abundance proportional to tumor grade. Additionally, the tumors express substantial PD-L1 proportional to tumor grade. These results suggest a role for immune checkpoint inhibitors targeting the PD-L1/PD-1 pathway in combination with standard therapies for the treatment of high-grade meningiomas.




Similar content being viewed by others
Abbreviations
- CTLA-4:
-
Cytotoxic T lymphocyte antigen 4
- FFPE:
-
Formalin-fixed, paraffin-embedded
- HPFs:
-
High powered fields
- IFN-γ:
-
Interferon-γ
- MDSC:
-
Myeloid-derived suppressor cells
- NCI:
-
National Cancer Institute
- NINDS:
-
National Institute of Neurological Disorders and Stroke
- PBMC:
-
Peripheral blood mononuclear cells
- PD-1:
-
Programmed death 1
- PD-L1:
-
Programmed death-ligand 1
- PFS:
-
Progression-free survival
- Treg:
-
Regulatory T cell
- WHO:
-
World Health Organization
References
Li Y, Veliceasa D, Lamano J, Lamano JB, Kaur G, Smith B, DiDomenico J, Oyon D, Bloch O (2018) Systemic and local immunosuppression in patients with high-grade meningiomas. J Neurosurg 128(4):961–1272 (Abstract)
Saraf S, McCarthy BJ, Villano JL (2011) Update on meningiomas. Oncologist 16(11):1604–1613. https://doi.org/10.1634/theoncologist.2011-0193
Perry A, Scheithauer BW, Stafford SL, Lohse CM, Wollan PC (1999) “Malignancy” in meningiomas: a clinicopathologic study of 116 patients, with grading implications. Cancer 85(9):2046–2056
Clark VE, Erson-Omay EZ, Serin A, Yin J, Cotney J, Ozduman K, Avsar T, Li J, Murray PB, Henegariu O, Yilmaz S, Gunel JM, Carrion-Grant G, Yilmaz B, Grady C, Tanrikulu B, Bakircioglu M, Kaymakcalan H, Caglayan AO, Sencar L, Ceyhun E, Atik AF, Bayri Y, Bai H, Kolb LE, Hebert RM, Omay SB, Mishra-Gorur K, Choi M, Overton JD, Holland EC, Mane S, State MW, Bilguvar K, Baehring JM, Gutin PH, Piepmeier JM, Vortmeyer A, Brennan CW, Pamir MN, Kilic T, Lifton RP, Noonan JP, Yasuno K, Gunel M (2013) Genomic analysis of non-NF2 meningiomas reveals mutations in TRAF7, KLF4, AKT1, and SMO. Science 339(6123):1077–1080. https://doi.org/10.1126/science.1233009
Brastianos PK, Horowitz PM, Santagata S, Jones RT, McKenna A, Getz G, Ligon KL, Palescandolo E, Van Hummelen P, Ducar MD, Raza A, Sunkavalli A, Macconaill LE, Stemmer-Rachamimov AO, Louis DN, Hahn WC, Dunn IF, Beroukhim R (2013) Genomic sequencing of meningiomas identifies oncogenic SMO and AKT1 mutations. Nat Genet 45(3):285–289. https://doi.org/10.1038/ng.2526
Perng P, Lim M (2015) Immunosuppressive mechanisms of malignant gliomas: parallels at non-CNS sites. Front Oncol. https://doi.org/10.3389/fonc.2015.00153
Borghaei H, Paz-Ares L, Horn L, Spigel DR, Steins M, Ready NE, Chow LQ, Vokes EE, Felip E, Holgado E, Barlesi F, Kohlhaufl M, Arrieta O, Burgio MA, Fayette J, Lena H, Poddubskaya E, Gerber DE, Gettinger SN, Rudin CM, Rizvi N, Crino L, Blumenschein GR Jr, Antonia SJ, Dorange C, Harbison CT, Graf Finckenstein F, Brahmer JR (2015) Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med 373(17):1627–1639. https://doi.org/10.1056/NEJMoa1507643
Ott PA, Hodi FS, Robert C (2013) CTLA-4 and PD-1/PD-L1 blockade: new immunotherapeutic modalities with durable clinical benefit in melanoma patients. Clin Cancer Res 19(19):5300–5309. https://doi.org/10.1158/1078-0432.Ccr-13-0143
Wolchok JD, Chiarion-Sileni V, Gonzalez R, Rutkowski P, Grob J-J, Cowey CL, Lao CD, Wagstaff J, Schadendorf D, Ferrucci PF, Smylie M, Dummer R, Hill A, Hogg D, Haanen J, Carlino MS, Bechter O, Maio M, Marquez-Rodas I, Guidoboni M, McArthur G, Lebbé C, Ascierto PA, Long GV, Cebon J, Sosman J, Postow MA, Callahan MK, Walker D, Rollin L, Bhore R, Hodi FS, Larkin J (2017) Overall survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med 377(14):1345–1356. https://doi.org/10.1056/NEJMoa1709684
Du Z, Abedalthagafi M, Aizer AA, McHenry AR, Sun HH, Bray MA, Viramontes O, Machaidze R, Brastianos PK, Reardon DA, Dunn IF, Freeman GJ, Ligon KL, Carpenter AE, Alexander BM, Agar NY, Rodig SJ, Bradshaw EM, Santagata S (2015) Increased expression of the immune modulatory molecule PD-L1 (CD274) in anaplastic meningioma. Oncotarget 6(7):4704–4716
Han SJ, Reis G, Kohanbash G, Shrivastav S, Magill ST, Molinaro AM, McDermott MW, Theodosopoulos PV, Aghi MK, Berger MS, Butowski NA, Barani I, Phillips JJ, Perry A, Okada H (2016) Expression and prognostic impact of immune modulatory molecule PD-L1 in meningioma. J Neurooncol 130(3):543–552. https://doi.org/10.1007/s11060-016-2256-0
Holmes M, Vides L, Chahine J, Harris B (2014) The role of microglia and T cells in different grades of meningioma. Am J Clin Pathol 142(suppl_1):A001. https://doi.org/10.1093/ajcp/142.suppl1.001
Vetsika E-K, Koinis F, Gioulbasani M, Aggouraki D, Koutoulaki A, Skalidaki E, Mavroudis D, Georgoulias V, Kotsakis A (2014) A circulating subpopulation of monocytic myeloid-derived suppressor cells as an independent prognostic/predictive factor in untreated non-small lung cancer patients. J Immunol Res 2014:12. https://doi.org/10.1155/2014/659294
Huang A, Zhang B, Wang B, Zhang F, Fan KX, Guo YJ (2013) Increased CD14(+)HLA-DR−/low myeloid-derived suppressor cells correlate with extrathoracic metastasis and poor response to chemotherapy in non-small cell lung cancer patients. Cancer Immunol Immunother 62(9):1439–1451. https://doi.org/10.1007/s00262-013-1450-6
Bloch O, Crane CA, Kaur R, Safaee M, Rutkowski MJ, Parsa AT (2013) Gliomas promote immunosuppression through induction of B7-H1 expression in tumor-associated macrophages. Clin Cancer Res 19(12):3165–3175. https://doi.org/10.1158/1078-0432.CCR-12-3314
Huang Y, Liao H, Zhang Y, Yuan R, Wang F, Gao Y, Wang P, Du Z (2014) Prognostic value of tumor-infiltrating FoxP3 + T cells in gastrointestinal cancers: a meta analysis. PLoS One 9(5):e94376. https://doi.org/10.1371/journal.pone.0094376
Santoiemma PP, Powell DJ Jr (2015) Tumor infiltrating lymphocytes in ovarian cancer. Cancer Biol Ther 16(6):807–820. https://doi.org/10.1080/15384047.2015.1040960
Zhao HQ, Li WM, Lu ZQ, Yao YM (2014) Roles of Tregs in development of hepatocellular carcinoma: a meta-analysis. World J Gastroenterol 20(24):7971–7978. https://doi.org/10.3748/wjg.v20.i24.7971
Miyan M, Schmidt-Mende J, Kiessling R, Poschke I, de Boniface J (2016) Differential tumor infiltration by T-cells characterizes intrinsic molecular subtypes in breast cancer. J Transl Med. https://doi.org/10.1186/s12967-016-0983-9
Zitron IM, Kamson DO, Kiousis S, Juhász C, Mittal S (2013) In vivo metabolism of tryptophan in meningiomas is mediated by indoleamine 2,3-dioxygenase 1. Cancer Biol Ther 14(4):333–339. https://doi.org/10.4161/cbt.23624
Chikuma S, Terawaki S, Hayashi T, Nabeshima R, Yoshida T, Shibayama S, Okazaki T, Honjo T (2009) PD-1-mediated suppression of IL-2 production induces CD8 + T cell anergy in vivo. J Immunol 182(11):6682–6689. https://doi.org/10.4049/jimmunol.0900080
Kansy BA, Concha-Benavente F, Srivastava RM, Jie H-B, Shayan G, Lei Y, Moskovitz J, Moy J, Li J, Brandau S, Lang S, Schmitt NC, Freeman GJ, Gooding WE, Clump DA, Ferris RL (2017) PD-1 status in CD8 + T cells associates with survival and anti-PD-1 therapeutic outcomes in head and neck cancer. Cancer Res 77(22):6353–6364. https://doi.org/10.1158/0008-5472.Can-16-3167
Branca MA (2016) Rekindling cancer vaccines. Nat Biotechnol 34:1019. https://doi.org/10.1038/nbt.3690
Dewan R, Pemov A, Dutra AS, Pak ED, Edwards NA, Ray-Chaudhury A, Hansen NF, Chandrasekharappa SC, Mullikin JC, Asthagiri AR, Heiss JD, Stewart DR, Germanwala AV (2017) First insight into the somatic mutation burden of neurofibromatosis type 2-associated grade I and grade II meningiomas: a case report comprehensive genomic study of two cranial meningiomas with vastly different clinical presentation. BMC Cancer 17(1):127. https://doi.org/10.1186/s12885-017-3127-6
Kanno H, Nishihara H, Wang L, Yuzawa S, Kobayashi H, Tsuda M, Kimura T, Tanino M, Terasaka S, Tanaka S (2013) Expression of CD163 prevents apoptosis through the production of granulocyte colony-stimulating factor in meningioma. Neuro-oncology 15(7):853–864. https://doi.org/10.1093/neuonc/not028
Nelson BH (2010) CD20 + B cells: the other tumor-infiltrating lymphocytes. J Immunol 185(9):4977–4982. https://doi.org/10.4049/jimmunol.1001323
Fang L, Lowther DE, Meizlish ML, Anderson RCE, Bruce JN, Devine L, Huttner AJ, Kleinstein SH, Lee JY, Stern JNH, Yaari G, Lovato L, Cronk KM, O’Connor KC (2013) The immune cell infiltrate populating meningiomas is composed of mature, antigen-experienced T and B cells. Neuro-oncology 15(11):1479–1490. https://doi.org/10.1093/neuonc/not110
Ding Y, Qiu L, Xu Q, Song L, Yang S, Yang T (2014) Relationships between tumor microenvironment and clinicopathological parameters in meningioma. Int J Clin Exp Pathol 7(10):6973–6979
Acknowledgements
The authors would like to thank the Nervous System Tumor Bank at Northwestern University, without which the current study would not be possible. Imaging work was performed at the Northwestern University Center for Advanced Microscopy generously supported by National Cancer Institute (NCI) CCSG P30 CA060553 awarded to the Robert H. Lurie Comprehensive Cancer Center.
Funding
This work was supported by the Howard Hughes Medical Institute Medical Student Research Fellows program (Yuping Li) as well as by the NIH/NCI (National Cancer Institute) R01 (CA164714; Orin Bloch) and NIH/National Institute of Neurological Disorders and Stroke (NINDS) R00 (NS078055; Orin Bloch). The funding was received by National Cancer Institute (CA206413; Jonathan B. Lamano) and National Institute of Neurological Disorders and Stroke (NS101884; Gurvinder Kaur).
Author information
Authors and Affiliations
Contributions
YDL took part in conceptualization, experimental design, data curation, formal analysis, funding acquisition, methodology, validation, and writing the manuscript; DV took part in experimental design and data curation; JBL took part in data curation, formal analysis, methodology, and validation. JBL took part in data curation and formal analysis; GK, and DB took part in data curation; CMH took part in formal analysis and validation; TJK took part in conceptualization and methodology; OB took part in conceptualization, funding acquisition, investigation, methodology, and supervision. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval and ethical standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study was approved by the Institutional Review Board of Northwestern University, Feinberg School of Medicine (STU00203854).
Informed consent
Written informed consent was obtained from all individual participants included in the study for the use of their blood and tumor specimen for research. Consent was not required for collection of patient characteristics as information was de-identified.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This work was previously published as an abstract at the 2018 American Association of Neurological Surgeons (AANS) Annual Scientific Meeting in New Orleans, LA, USA on April 29–May 2, 2018 [1].
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, Y.D., Veliceasa, D., Lamano, J.B. et al. Systemic and local immunosuppression in patients with high-grade meningiomas. Cancer Immunol Immunother 68, 999–1009 (2019). https://doi.org/10.1007/s00262-019-02342-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-019-02342-8