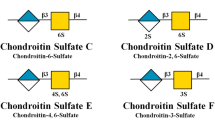
Abstract
Glycosaminoglycans are linear anionic polysaccharides that exhibit a number of important biological and pharmacological activities. The two most prominent members of this class of polysaccharides are heparin/heparan sulfate and the chondroitin sulfates (including dermatan sulfate). These polysaccharides, having complex structures and polydispersity, are biosynthesized in the Golgi of most animal cells. The chemical synthesis of these glycosaminoglycans is precluded by their structural complexity. Today, we depend on food animal tissues for their isolation and commercial production. Ton quantities of these glycosaminoglycans are used annually as pharmaceuticals and nutraceuticals. The variability of animal-sourced glycosaminoglycans, their inherent impurities, the limited availability of source tissues, the poor control of these source materials, and their manufacturing processes suggest a need for new approaches for their production. Over the past decade, there have been major efforts in the biotechnological production of these glycosaminoglycans. This mini-review focuses on the use of recombinant enzymes and metabolic engineering for the production of heparin and chondroitin sulfates.






Similar content being viewed by others
References
Adebowale AO, Cox DS, Liang Z, Eddington ND (2000) Analysis of glucosamine and chondroitin sulfate content in marketed products and the Caco-2 permeability of chondroitin sulfate raw materials. J Am Nutraceutical Assoc 3:37–44
Baik JY, Gasimli L, Yang B, Datta P, Zhang F, Glass CA, Esko JD, Linhardt RJ, Sharfstein ST (2012) Metabolic engineering of Chinese hamster ovary cells: towards a bioengineered heparin. Metab Eng 14:81–90. doi:10.1016/j.ymben.2012.01.008
Bernfield M, Götte M, Park PW, Reizes O, Fitzgerald ML, Lincecum J, Zako M (1999) Functions of cell surface heparan sulfate proteoglycans. Annu Rev Biochem 68:729–777. doi:10.1146/annurev.biochem.68.1.729
Berninsone P, Hirschberg C (1998) Heparan sulfate/heparin N-deacetylase/N-sulfotransferase: the N-sulfotransferase activity domain is at the carboxyl half of the holoenzyme. J Biochem 273:25556–25559
Bhan N, Xu P, Koffas MAG (2013) Pathway and protein engineering approaches to produce novel and commodity small molecules. Curr Opin Biotechnol 24:1137–1143. doi:10.1016/j.copbio.2013.02.019
Bhaskar U, Sterner E, Hickey AM, Onishi A, Zhang F, Dordick JS, Linhardt RJ (2012) Engineering of routes to heparin and related polysaccharides. Appl Microbiol Biotechnol 93:1–16. doi:10.1007/s00253-011-3641-4
Bhaskar U, Li G, Fu L, Onishi A, Suflita M, Dordick J, Linhardt RJ (2015) Combinatorial one-pot chemoenzymatic synthesis of heparin. Carbohydr Polymer 122:399–407
Bick RL, Frenkel EP, Walenga J, Fareed J, Hoppensteadt DA (2005) Unfractionated heparin, low molecular weight heparins, and pentasaccharide: basic mechanism of actions, pharmacology, and clinical use. Hematol Oncol Clin North Am 19:1–51. doi:10.1016/j.hoc.2004.09.003
Burkart M, Wong C (1999) A continuous assay for the spectrophotometric analysis of sulfotransferases using aryl sulfotransferase IV. Anal Biochem 274:131–137. doi:10.1006/abio.1999.4264
Burkart M, Izumi M, Chapman E, Lin C, Wong C (2000) Regeneration of PAPS for the enzymatic synthesis of sulfated oligosaccharides. J Org Chem 65:5565–5574
Castelli R, Porro F, Tarsia P (2004) The heparins and cancer: review of clinical trials and biological properties. Vasc Med 9:205–213. doi:10.1191/1358863x04vm566ra
Chavaroche AAE, Van Den Broek LAM, Boeriu C, Eggink G (2012) Synthesis of heparosan oligosaccharides by Pasteurella multocida PmHS2 single-action transferases. Appl Microbiol Biotechnol 95:1199–1210. doi:10.1007/s00253-011-3813-2
Chen M, Bridges A, Liu J (2006) Determination of the substrate specificities of N-acetyl-d-glucosaminyltransferase. Biochemistry 45:12358–12365
Chen Y, Li Y, Yu H, Sugiarto G, Thon V, Hwang J, Ding L, Hie L, Chen X (2013) Tailored design and synthesis of heparan sulfate oligosaccharide analogues using sequential one-pot multienzyme systems. Angew Chem Int Ed Engl 52:11852–11856. doi:10.1002/anie.201305667
Cimini D, Restaino OF, Catapano A, De Rosa M, Schiraldi C (2010a) Production of capsular polysaccharide from Escherichia coli K4 for biotechnological applications. Appl Microbiol Biotechnol 85:1779–1787. doi:10.1007/s00253-009-2261-8
Cimini D, De Rosa M, Viggiani A, Restaino OF, Carlino E, Schiraldi C (2010b) Improved fructosylated chondroitin production by kfoC overexpression in E. coli K4. J Biotechnol 150:324–331. doi:10.1016/j.jbiotec.2010.09.954
Cimini D, De Rosa M, Carlino E, Ruggiero A, Schiraldi C (2013) Homologous overexpression of rfaH in E. coli K4 improves the production of chondroitin-like capsular polysaccharide. Microb Cell Factories 12:46. doi:10.1186/1475-2859-12-46
Clarke BR, Esumeh F, Roberts IS (2000) Cloning, expression, and purification of the K5 capsular polysaccharide lyase (KflA) from coliphage K5A: evidence for two distinct K5 lyase enzymes. J Bacteriol 182:3761–3766
Cress BF, Englaender JA, He W, Kasper D, Linhardt RJ, Koffas MAG (2014) Masquerading microbial pathogens: capsular polysaccharides mimic host-tissue molecules. FEMS Microbiol Rev 38:660–697. doi:10.1111/1574-6976.12056
Cress BF, Toparlak OD, Guleria S, Lebovich M, Stieglitz JT, Englaender JA, Jones JA, Linhardt RJ, Koffas MAG (2015) CRISPathBrick: modular combinatorial assembly of type II-A CRISPR arrays for dCas9-mediated multiplex transcriptional repression in E. coli. ACS Synth Biol. doi:10.1021/acssynbio.5b00012
Datta P, Li G, Yang B, Zhao X, Baik JY, Gemmill TR, Sharfstein ST, Linhardt RJ (2013a) Bioengineered Chinese hamster ovary cells with Golgi-targeted 3-O-sulfotransferase-1 biosynthesize heparan sulfate with an antithrombin-binding site. J Biol Chem 288:37308–37318. doi:10.1074/jbc.M113.519033
Datta P, Linhardt RJ, Sharfstein ST (2013b) An ’omics approach towards CHO cell engineering. Biotechnol Bioeng 110:1255–1271. doi:10.1002/bit.24841
DeAngelis PL, White CL (2002) Identification and molecular cloning of a heparosan synthase from Pasteurella multocida type D. J Biol Chem 277:7209–7213. doi:10.1074/jbc.M112130200
Deangelis PL, Liu J, Linhardt RJ (2013) Chemoenzymatic synthesis of glycosaminoglycans: re-creating, re-modeling and re-designing nature’s longest or most complex carbohydrate chains. Glycobiology 23:764–777. doi:10.1093/glycob/cwt016
Doherty DH, Weaver CA, Miyamoto K, Minamisawa T (2011) Compositions and methods for bacterial production of chondroitin. US Patent# US20110244520 A1
Esko JD, Lindahl U (2001) Molecular diversity of heparan sulfate. J Clin Invest 108:169–173. doi:10.1172/JCI13530
Esko JD, Selleck SB (2002) Order out of chaos: assembly of ligand binding sites in heparan sulfate. Annu Rev Biochem 71:435–471. doi:10.1146/annurev.biochem.71.110601.135458
Evans TD, Mozen MM (1962) Process for purifying heparin. US Patent #3058884
Fu L, Li G, Yang B, Onishi A, Li L, Sun P, Zhang F, Linhardt R (2013) Structural characterization of pharmaceutical heparins prepared from different animal tissues. J Pharm Sci 102:1447–1457
Fu L, Li L, Cai C, Li G, Zhang F, Linhardt RJ (2014a) Heparin stability by determining unsubstituted amino groups using hydrophilic interaction chromatography mass spectrometry. Anal Biochem 461:46–48. doi:10.1016/j.ab.2014.05.028
Fu L, Zhang F, Li G, Onishi A, Bhaskar U, Sun P, Linhardt RJ (2014b) Structure and activity of a new low-molecular-weight heparin produced by enzymatic ultrafiltration. J Pharm Sci 10(100):1375–1383. doi:10.1002/jps.23939
Gijsen HJM, Qiao L, Fitz W, Wong C-H (1996) Recent advances in the chemoenzymatic synthesis of carbohydrates and carbohydrate mimetics. Chem Rev 96:443–474. doi:10.1021/cr950031q
Griffiths G, Barrett B, Cook N, Roberts IS (1999) Biosynthesis of the Escherichia coli K5 capsular polysaccharide. Biochem Soc Trans 27:507–512
Guerrini M, Beccati D, Shriver Z, Naggi A, Viswanathan K, Bisio A, Capila I, Lansing JC, Guglieri S, Fraser B, Al-Hakim A, Gunay NS, Zhang Z, Robinson L, Buhse L, Nasr M, Woodcock J, Langer R, Venkataraman G, Linhardt RJ, Casu B, Torri G, Sasisekharan R (2008) Oversulfated chondroitin sulfate is a major contaminant in heparin associated with adverse clinical events. Nat Biotechnol 26:669–675
He W, Fu L, Li G, Jones A, Linhardt RJ, Koffas M (2015) Production of chondroitin in metabolically engineered E. coli. Metab Eng 27:92–100. doi:10.1016/j.ymben.2014.11.003
Higashi K, Hosoyama S, Ohno A, Masuko S, Yang B, Sterner E, Wang Z, Linhardt R, Toida T (2012) Photochemical preparation of a novel molecular weight heparin. Carbohydr Polym 87:1737–1743
Higashi K, Takeuchi Y, Mukuno A, Tomitori H, Miya M, Linhardt RJ, Toida T (2015) Composition of glycosaminoglycans in elasmobranchs including several deep-sea sharks: identification of chondroitin/dermatan sulfate from the dried fins of Isurus oxyrinchus and Prionace glauca. PLoS One 10, e0120860. doi:10.1371/journal.pone.0120860
Jones JA, Toparlak ÖD, Koffas MA (2015) Metabolic pathway balancing and its role in the production of biofuels and chemicals. Curr Opin Biotechnol 33:52–59. doi:10.1016/j.copbio.2014.11.013
Karst NA, Linhardt RJ (2003) Recent chemical and enzymatic approaches to the synthesis of glycosaminoglycan oligosaccharides | BenthamScience. Curr Med Chem 10:1993–2031
Kato M, Wang H, Bernfield M, Gallagher JT, Turnbull JE (1994) Cell surface syndecan-1 on distinct cell types differs in fine structure and ligand binding of its heparan sulfate chains. J Biol Chem 269:18881–18890
Keire DA, Buhse LF, Al-Hakim A (2013) Characterization of currently marketed heparin products: composition analysis by 2D-NMR. Anal Methods 5:2984–2994
Kuberan B, Lech MZ, Beeler DL, Wu ZL, Rosenberg RD (2003) Enzymatic synthesis of antithrombin III-binding heparan sulfate pentasaccharide. Nat Biotechnol 21:1343–1346. doi:10.1038/nbt885
Kusche-Gullberg M, Kjellén L (2003) Sulfotransferases in glycosaminoglycan biosynthesis. Curr Opin Struct Biol 13:605–611. doi:10.1016/j.sbi.2003.08.002
Lassen MR, Dahl OE, Mismetti P, Destrée D, Turpie AG (2009) AVE5026, a new hemisynthetic ultra-low-molecular-weight heparin for the prevention of venous thromboembolism in patients after total knee replacement surgery—TREK: a dose-ranging study. J Thromb Haemost 7:566–572. doi:10.1111/j.1538-7836.2009.03301.x
Lauder RM (2009) Chondroitin sulphate: a complex molecule with potential impacts on a wide range of biological systems. Complement Ther Med 17:56–62. doi:10.1016/j.ctim.2008.08.004
Li JP, Vlodavsky I (2009) Heparin, heparan sulfate and heparanase in inflammatory reactions. Thromb Haemost 102:823–828. doi:10.1160/TH09-02-0091
Li L, Zhang F, Zaia J, Linhardt RJ (2012) Top-down approach for the direct characterization of low molecular weight heparins using LC-FT-MS. Anal Chem 84:8822–8829. doi:10.1021/ac302232c
Li G, Li L, Tian F, Zhang L, Xue C, Linhardt RJ (2015) Glycosaminoglycanomics of cultured cells using a rapid and sensitive LC-MS/MS approach. ACS Chem Biol. doi:10.1021/acschembio.5b00011
Lindahl U, Kusche-Gullberg M, Kjellen L (1998) Regulated diversity of heparan sulfate. J Biol Chem 273:24979–24982. doi:10.1074/jbc.273.39.24979
Lindahl U, Li J, Kusche-Gullberg M, Salmivirta M, Alaranta S, Veromaa T, Emeis J, Roberts I, Taylor C, Oreste P, Zopetti G, Naggi A, Torri G, Casu B (2005) Generation of “neoheparin” from E. coli K5 capsular polysaccharide. J Med Chem 48(2):349--352. doi:10.1021/jm049812m
Linhardt RJ (2003) Heparin: strucuture and activity. J Med Chem 46:2521–2564
Linhardt RJ, Liu J (2012) Synthetic heparin. Curr Opin Pharmacol 12:217–219. doi:10.1016/j.coph.2011.12.002
Linhardt RJ, Ampofo SA, Fareed J, Hoppensteadt D, Folkman J, Mulliken JB (1992) Isolation and characterization of human heparin. Biochemistry 31:12441–12445. doi:10.1021/bi00164a020
Liu J, Shriver Z, Pope RM, Thorp SC, Duncan MB, Copeland RJ, Raska CS, Yoshida K, Eisenberg RJ, Cohen G, Linhardt RJ, Sasisekharan R (2002) Characterization of a heparan sulfate octasaccharide that binds to herpes simplex virus type 1 glycoprotein D. J Biol Chem 277:33456–33467. doi:10.1074/jbc.M202034200
Liu H, Zhang Z, Linhardt RJ (2009) Lessons learned from the contamination of heparin. Nat Prod Rep 26:313–321. doi:10.1039/b819896a
Liu R, Xu Y, Chen M, Weïwer M, Zhou X, Bridges AS, DeAngelis PL, Zhang Q, Linhardt RJ, Liu J (2010) Chemoenzymatic design of heparan sulfate oligosaccharides. J Biol Chem 285:34240–34249. doi:10.1074/jbc.M110.159152
Liu C, Sheng J, Krahn JM, Perera L, Xu Y, Hsieh PH, Dou W, Liu J, Pedersen LC (2014) Molecular mechanism of substrate specificity for heparan sulfate 2-O-sulfotransferase. J Biol Chem 289:13407–13418. doi:10.1074/jbc.M113.530535
Loganathan D, Wang HM, Mallis LM, Linhardt RJ (1990) Structural variation in the antithrombin III binding site region and its occurrence in heparin from different sources. Biochemistry 29:4362–4368
Manzoni M, Bergomi S, Molinari F, Cavazzoni V (1996) production and purification of an extracellularly produced K4 polysaccharide from Escherichia coli. Biotechnol Lett 4:383–386. doi:10.1007/BF00143456
Masuko S, Bera S, Green DE, Weïwer M, Liu J, Deangelis PL, Linhardt RJ (2012) Chemoenzymatic synthesis of uridine diphosphate-GlcNAc and uridine diphosphate-GalNAc analogs for the preparation of unnatural glycosaminoglycans. J Org Chem 77:1449–1456. doi:10.1021/jo202322k
Mikami T, Kitagawa H (2013) Biosynthesis and function of chondroitin sulfate. Biochim Biophys Acta Gen Subj 1830:4719–4733. doi:10.1016/j.bbagen.2013.06.006
Moon AF, Edavettal SC, Krahn JM, Munoz EM, Negishi M, Linhardt RJ, Liu J, Pedersen LC (2004) Structural analysis of the sulfotransferase (3-O-sulfotransferase isoform 3) involved in the biosynthesis of an entry receptor for herpes simplex virus 1. J Biol Chem 279:45185–45193. doi:10.1074/jbc.M405013200
Moon AF, Xu Y, Woody SM, Krahn JM, Linhardt RJ, Liu J, Pedersen LC (2012) Dissecting the substrate recognition of 3-O-sulfotransferase for the biosynthesis of anticoagulant heparin. Proc Natl Acad Sci 109:5265–5270. doi:10.1073/pnas.1117923109
Mousa SA, Fareed J (2001) Overview: from heparin to low molecular weight heparin: beyond anticoagulation. Curr Opin Investig Drugs 2:1077–1080
Murugesan S, Xie J, Linhardt RJ (2008) Immobilization of heparin: approaches and applications. Curr Top Med Chem 8:80–100. doi:10.2174/156802608783378891
Naggi A, Torri G, Casu B, Oreste P, Zoppetti G, Li JP, Lindahl U (2001) Toward a biotechnological heparin through combined chemical and enzymatic modification of the Escherichia coli K5 polysaccharide. Semin Thromb Hemost 27:437–443. doi:10.1055/s-2001-17954
Okuyama T, Yoshida K, Sakuraik, Ogurat, Horie K, Tawada A (1975) Method of separating and recovering mucopolysaccharides from connective tissues of animals. US Patent #3862003
Orellana A, Hirschberg CB, Wei Z, Swiedler SJ, Ishihara M (1994) Molecular cloning and expression of a glycosaminoglycan N-acetylglucosaminyl N-deacetylase/N-sulfotransferase from a heparin-producing cell line. J Biol Chem 269:2270–2276
Osterman MT, Lichtenstein GR (2007) Current and future anti-TNF therapy for inflammatory bowel disease. Curr Treat Options in Gastroenterol 10:195–207. doi:10.1007/s11938-007-0013-3
Pacheco B, Maccarana M, Goodlett DR, Malmström A, Malmström L (2009a) Identification of the active site of DS-epimerase 1 and requirement of N-glycosylation for enzyme function. J Biol Chem 284:1741–1747. doi:10.1074/jbc.M805479200
Pacheco B, Malmström A, Maccarana M (2009b) Two dermatan sulfate epimerases form iduronic acid domains in dermatan sulfate. J Biol Chem 284:9788–9795. doi:10.1074/jbc.M809339200
Papoutsaki M, Osório F, Morais P, Torres T, Magina S, Chimenti S, Costanzo A (2013) Infliximab in psoriasis and psoriatic arthritis. BioDrugs 27(Suppl 1):13–23. doi:10.1007/BF03325638
Parish CR, Freeman C, Hulett MD (2001) Heparanase: a key enzyme involved in cell invasion. Biochim Biophys Acta 1471:M99–M108
Petitou M, Duchaussoy P, Lederman I, Choay J, Sinaÿ P, Jacquinet J-C, Torri G (1986) Synthesis of heparin fragments. A chemical synthesis of the pentasaccharide O-(2-deoxy-2-sulfamido-6-O-sulfo-alpha-D-glucopyranosyl)-(1–4)-O-(beta-D-glucopyranosyluronic acid)-(1–4)-O-(2-O-sulfo-alpha-L-idopyranosyluronic acid)-(1–4)-2-deoxy-2-sulfamido-6-. Carbohydr Res 147:221–236
Qin Y, Ke J, Gu X, Fang J, Wang W, Cong Q, Li J, Tan J, Brunzelle JS, Zhang C, Jiang Y, Melcher K, Li J, Xu HE, Ding K (2015) Structural and functional study of D-glucuronyl C5-epimerase. J Biol Chem 290:4620–4630. doi:10.1074/jbc.M114.602201
Ramani VC, Purushothaman A, Stewart M, Thompson C, Vlodavsky I, Au J, Sanderson RD (2013) The heparanase/syndecan-1 axis in cancer: mechanisms and therapies. FEBS J 280:2294–2306. doi:10.1016/j.biotechadv.2011.08.021
Restaino OF, Cimini D, De Rosa M, Catapano A, Schiraldi C, De Rosa M, Catapano A, Schiraldi C, De Rosa M, Catapano A, Schiraldi C (2011) High cell density cultivation of Escherichia coli K4 in a microfiltration bioreactor: a step towards improvement of chondroitin precursor production. Microb Cell Factories 10:1–11. doi:10.1186/1475-2859-10-10
Restaino OF, Bhaskar U, Paul P, Li L, De Rosa M, Dordick JS, Linhardt RJ (2013a) High cell density cultivation of a recombinant E. coli strain expressing a key enzyme in bioengineered heparin production. Appl Microbiol Biotechnol 97:3893–3900. doi:10.1007/s00253-012-4682-z
Restaino OF, di Lauro I, Cimini D, Carlino E, De Rosa M, Schiraldi C (2013b) Monosaccharide precursors for boosting chondroitin-like capsular polysaccharide production. Appl Microbiol Biotechnol 97:1699–1709. doi:10.1007/s00253-012-4343-2
Sakai S, Otake E, Toida T, Goda Y (2007) Identification of the origin of chondroitin sulfate in “health foods”. Chem Pharm Bull (Tokyo) 55:299–303
Saribaş AS, Mobasseri A, Pristatsky P, Chen X, Barthelson R, Hakes D, Wang J (2004) Production of N-sulfated polysaccharides using yeast-expressed N-deacetylase/N-sulfotransferase-1 (NDST-1). Glycobiology 14:1217–1228. doi:10.1093/glycob/cwh129
Sasisekharan R, Venkataraman G (2000) Heparin and heparan sulfate: biosynthesis, structure and function. Curr Opin Chem Biol 4:626–631. doi:10.1016/S1367-5931(00)00145-9
Sato N, Meijer L, Skaltsounis L, Greengard P, Brivanlou AH (2004) Maintenance of pluripotency in human and mouse embryonic stem cells through activation of Wnt signaling by a pharmacological GSK-3-specific inhibitor. Nat Med 10:55–63. doi:10.1038/nm979
Schiraldi C, Cimini D, De Rosa M (2010) Production of chondroitin sulfate and chondroitin. Appl Microbiol Biotechnol 87:1209–1220. doi:10.1007/s00253-010-2677-1
Schonberger L (1998) New variant Creutzfeldt-Jakob disease and bovine spongiform encephalopathy. Infect Dis Clin N Am 12:111–121
Sheng J, Liu R, Xu Y, Liu J (2011) The dominating role of N-deacetylase/N-sulfotransferase 1 in forming domain structures in heparan sulfate. J Biol Chem 286:19768–19776. doi:10.1074/jbc.M111.224311
Sheng J, Xu Y, Dulaney SB, Huang X, Liu J (2012) Uncovering biphasic catalytic mode of C5-epimerase in heparan sulfate biosynthesis. J Biol Chem 287:20996–21002. doi:10.1074/jbc.M112.359885
Shi X, Zaia J (2009) Organ-specific heparan sulfate structural phenotypes. J Biol Chem 284:11806–11814. doi:10.1074/jbc.M809637200
Silbert JE, Sugumaran G (2002) Biosynthesis of chondroitin/dermatan sulfate. IUBMB Life 54:177–186. doi:10.1080/15216540290114450
Sismey-Ragatz AE, Green DE, Otto NJ, Rejzek M, Field RA, DeAngelis PL (2007) Chemoenzymatic synthesis with distinct Pasteurella heparosan synthases: monodisperse polymers and unnatural structures. J Biol Chem 282:28321–28327. doi:10.1074/jbc.M701599200
Sterner E, Li L, Paul P, Beaudet JM, Liu J, Linhardt RJ, Dordick JS (2014) Assays for determining heparan sulfate and heparin O-sulfotransferase activity and specificity. Anal Bioanal Chem 406:525–536. doi:10.1007/s00216-013-7470-4
Sugiura N, Tawada A, Sugimoto K, Watanabe H (2002) Molecular cloning and characterization of chondroitin polymerase from Escherichia coli strain K4. J Biol Chem 277:21567–21575. doi:10.1074/jbc.M201719200
Sugiura N, Shioiri T, Chiba M, Sato T, Narimatsu H, Kimata K, Watanabe H (2012) Construction of a chondroitin sulfate library with defined structures and analysis of molecular interactions. J Biol Chem 287:43390–43400. doi:10.1074/jbc.M112.412676
Sun X, Li L, Overdier KH, Ammons LA, Douglas IS, Burlew CC, Zhang F, Schmidt EP, Chi L, Linhardt RJ (2015) Analysis of total human urinary glycosaminoglycan disaccharides by liquid chromatography–tandem mass spectrometry. Anal Chem 87:6220–6227. doi:10.1021/acs.analchem.5b00913
Taipale J, Keski-Oja J (1997) Growth factors in the extracellular matrix. FASEB J Off Publ Fed Am Soc Exp Biol 11:51–59
Thelin MA, Bartolini B, Axelsson J, Gustafsson R, Tykesson E, Pera E, Oldberg A, MacCarana M, Malmstrom A (2013) Biological functions of iduronic acid in chondroitin/dermatan sulfate. FEBS J 280:2431–2446. doi:10.1111/febs.12214
Toschi V, Lettino M (2007) Fondaparinux: pharmacology and clinical experience in cardiovascular medicine. Mini - Rev Med Chem 7:383–387
Tully SE, Rawat M, Hsieh-Wilson LC (2006) Discovery of a TNF-alpha antagonist using chondroitin sulfate microarrays. J Am Chem Soc 128:7740–7741. doi:10.1021/ja061906t
Van Gorp CL, Vosburgh F, Schubert RL (1997) Protein hydrolysate derived from mucosa tissue. US Patent #5607840
Vidic H-J (1981) Process for the preparation of heparin. US Patent # 4283530.
Viskov C, Just M, Laux V, Mourier P, Lorenz M (2009) Description of the chemical and pharmacological characteristics of a new hemisynthetic ultra-low-molecular-weight heparin, AVE5026. J Thromb Haemost 7:1143–1151. doi:10.1111/j.1538-7836.2009.03447.x
Vlodavsky I, Friedmann Y (2001) Heparan sulfate proteoglycans molecular properties and involvement of heparanase in cancer metastasis and angiogenesis. J Clin Investig 108:341–347. doi:10.1172/JCI200113662
Wang Z, Yang B, Zhang Z, Mellisa L, Takieddin M, Mousa S, Liu J, Dordick JS, Linhardt RJ (2011) Control of the heparosan N-deacetylation leads to an improved bioengineered heparin. Appl Microbiol Biotechnol 91:91–99
Wang W, Englaender JA, Xu P, Mehta KK, Suwan J, Dordick JS, Zhang F, Yuan Q, Linhardt RJ, Koffas M (2013) Expression of low endotoxin 3-o-sulfotransferase in Bacillus subtilis and Bacillus megaterium. Appl Biochem Biotechnol 171:954–962. doi:10.1007/s12010-013-0415-8
Williams RE (1967) Process for the recovery of heparin. US Patent #3337409
Wu Q, Yang A, Zou W, Duan Z, Liu J, Chen J, Liu L (2013) Transcriptional engineering of Escherichia coli K4 for fructosylated chondroitin production. Biotechnol Prog 29:1140–1149. doi:10.1002/btpr.1777
Xiong J, Bhaskar U, Li G, Fu L, Li L, Zhang F, Dordick JS, Linhardt RJ (2013) Immobilized enzymes to convert N-sulfo, N-acetyl heparosan to a critical intermediate in the production of bioengineered heparin. J Biotechnol 167:241–247. doi:10.1016/j.jbiotec.2013.06.018
Xu Y, Masuko S, Takieddin M, Xu H, Liu R, Jing J, Mousa SA, Linhardt RJ, Liu J, Xu Y, Masuko S, Takieddin M, Xu H, Liu R, Jing J, Mousa S, Linhardt RJ, Liu J (2011) Chemoenzymatic synthesis of structurally homogeneous ultra-low molecular weight heparins. Science 334(80-):498–501. doi:10.1126/science.1207478
Xu P, Bhan N, Koffas MAG (2013) Engineering plant metabolism into microbes: from systems biology to synthetic biology. Curr Opin Biotechnol 24:291–299. doi:10.1016/j.copbio.2012.08.010
Xu Y, Cai C, Chandarajoti K, Hsieh P-H, Li L, Pham TQ, Sparkenbaugh EM, Sheng J, Key NS, Pawlinski R, Harris EN, Linhardt RJ, Liu J (2014) Homogenous low-molecular-weight heparins with reversible anticoagulant activity. Nat Chem Biol 10:248–250. doi:10.1038/nchembio.1459
Yang B, Chang Y, Weyers AM, Sterner E, Linhardt RJ (2012) Disaccharide analysis of glycosaminoglycan mixtures by ultra-high-performance liquid chromatography-mass spectrometry. J Chromatogr A 1225:91–98. doi:10.1016/j.chroma.2011.12.063
Zanfardino A, Restaino OF, Notomista E, Cimini D, Schiraldi C, De Rosa M, De Felice M, Varcamonti M (2010) Isolation of an Escherichia coli K4 kfoC mutant over-producing capsular chondroitin. Microb Cell Factories 9:34. doi:10.1186/1475-2859-9-34
Zhang Z, McCallum SA, Xie J, Nieto L, Corzana F, Jimenez-Barbero J, Chen M, Liu J, Linhardt RJ (2008) Solution structures of chemoenzymatically synthesized heparin and its precursors. J Am Chem Soc 130:12998–13007
Zhang F, Yang B, Mellisa L, Kemal S, Xiao Z, Wang Z, Beaudet JM, Torelli AY, Dordick JS, Linhardt RJ (2011) Structural characterization of heparins from different commercial sources. Anal Bioanal Chem 401:2793–2803
Zhang C, Liu L, Teng L, Chen J, Liu J, Li J, Du G, Chen J (2012) Metabolic engineering of Escherichia coli BL21 for biosynthesis of heparosan, a bioengineered heparin precursor. Metab Eng 14:521–527. doi:10.1016/j.ymben.2012.06.005
Zhang J, Suflita M, Fiaschetti CM, Li G, Li L, Zhang F, Dordick JS, Linhardt RJ (2015a) High cell density cultivation of a recombinant Escherichia coli strain expressing a 6-O-sulfotransferase for the production of bioengineered heparin. J Appl Microbiol 118:92–98. doi:10.1111/jam.12684
Zhang J, Suflita M, Li G, Zhong W, Li L, Dordick JS, Linhardt RJ, Zhang F (2015b) High cell density cultivation of recombinant Escherichia coli strains expressing 2-O-sulfotransferase and C5-epimerase for the production of bioengineered heparin. Appl Biochem Biotechnol 175:2986–2995. doi:10.1007/s12010-014-1466-1
Zhou YJ, Hao XF, Tian ZJ, Tong GZ, Yoo D, An TQ, Zhou T, Li GX, Qiu HJ, Wei TC, Yuan XF (2008) Highly virulent porcine reproductive and respiratory syndrome virus emerged in China. Transbound Emerg Dis 55:152–164. doi:10.1111/j.1865-1682.2008.01020.x
Zhou X, Chandarajoti K, Pham TQ, Liu R, Liu J (2011) Expression of heparan sulfate sulfotransferases in Kluyveromyces lactis and preparation of 3′-phosphoadenosine-5′-phosphosulfate. Glycobiology 21:771–780. doi:10.1093/glycob/cwr001
Zoppetti G, Oreste P (2004) process for the preparation of chondroitin sulfates from K4 polysaccharide and obtained products. US Patent 6777398 B2
Acknowledgments
The authors are grateful for support from the National Institutes of Health (HL094463, GM102137, HL62244, HL096972) and the National Science Foundation (MCB-1448657).
Conflict of interest
The authors declare no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Suflita, M., Fu, L., He, W. et al. Heparin and related polysaccharides: synthesis using recombinant enzymes and metabolic engineering. Appl Microbiol Biotechnol 99, 7465–7479 (2015). https://doi.org/10.1007/s00253-015-6821-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-015-6821-9