Abstract
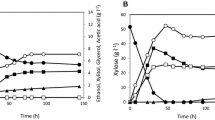
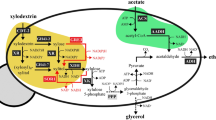
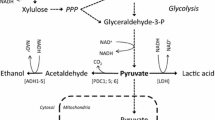
Production of lactic acid from renewable sugars has received growing attention as lactic acid can be used for making renewable and bio-based plastics. However, most prior studies have focused on production of lactic acid from glucose despite that cellulosic hydrolysates contain xylose as well as glucose. Microbial strains capable of fermenting both glucose and xylose into lactic acid are needed for sustainable and economic lactic acid production. In this study, we introduced a lactic acid-producing pathway into an engineered Saccharomyces cerevisiae capable of fermenting xylose. Specifically, ldhA from the fungi Rhizopus oryzae was overexpressed under the control of the PGK1 promoter through integration of the expression cassette in the chromosome. The resulting strain exhibited a high lactate dehydrogenase activity and produced lactic acid from glucose or xylose. Interestingly, we observed that the engineered strain exhibited substrate-dependent product formation. When the engineered yeast was cultured on glucose, the major fermentation product was ethanol while lactic acid was a minor product. In contrast, the engineered yeast produced lactic acid almost exclusively when cultured on xylose under oxygen-limited conditions. The yields of ethanol and lactic acid from glucose were 0.31 g ethanol/g glucose and 0.22 g lactic acid/g glucose, respectively. On xylose, the yields of ethanol and lactic acid were <0.01 g ethanol/g xylose and 0.69 g lactic acid/g xylose, respectively. These results demonstrate that lactic acid can be produced from xylose with a high yield by S. cerevisiae without deleting pyruvate decarboxylase, and the formation patterns of fermentations can be altered by substrates.






Similar content being viewed by others
References
Adachi E, Torigoe M, Sugiyama M, Nikawa J, Shimizu K (1998) Modification of metabolic pathways of Saccharomyces cerevisiae by the expression of lactate dehydrogenase and deletion of pyruvate decarboxylase genes for the lactic acid fermentation at low pH value. J Ferment Bioeng 86:284–289
Andrade RP, Casal M (2001) Expression of the lactate permease gene JEN1 from the yeast Saccharomyces cerevisiae. Fungal Genet Biol 32:105–111
Angermayr SA, Paszota M, Hellingwerf KJ (2012) Engineering a cyanobacterial cell factory for production of lactic acid. Appl Environ Microbiol 78:7098–7106
Bai D, Jia M, Zhao X, Ban R, Shen F, Li X, Xu S (2003) L(+)-lactic acid production by pellet-form Rhizopus oryzae R1021 in a stirred tank fermentor. Chem Eng Sci 58:785–791
Barnett JA (1976) The utilization of sugars by yeasts. Adv Carbohydr Chem Biochem 32:125–234
Bianchi MM, Brambilla L, Protani F, Liu C, Lievense J, Porro D (2001) Efficient homolactic fermentation by Kluyveromyces lactis strains defective in pyruvate utilization and transformed with the heterologous LDH gene. Appl Environ Microbiol 67:5621–5625
Boiteux A, Hess B (1970) Allosteric properties of yeast pyruvate decarboxylase. FEBS Lett 9:293–296
Casal M, Paiva S, Andrade RP, Gancedo C, Leao C (1999) The lactate-proton symport of Saccharomyces cerevisiae is encoded by JEN1. J Bacteriol 181:2620–2623
Castillo Martinez FA, Balciunas EM, Salgado JM, Dominguez Gonzalez JM, Converti A, Oliveira, de Souza Pinheiro R (2013) Lactic acid properties, applications and production: a review. Trends Food Sci Technol 30:70–83
Colombié S, Dequin S, Sablayrolles JM (2003) Control of lactate production by Saccharomyces cerevisiae expressing a bacterial LDH gene. Enzyme Microb Technol 33:38–46
Dequin S, Baptista E, Barre P (1999) Acidification of grape musts by Saccharomyces cerevisiae wine yeast strains genetically engineered to produce lactic acid. Am J Enol Vitic 50:45–50
Fortman J, Chhabra S, Mukhopadhyay A, Chou H, Lee TS, Steen E, Keasling JD (2008) Biofuel alternatives to ethanol: pumping the microbial well. Trends Biotechnol 26:375–381
Garlotta D (2001) A literature review of poly (lactic acid). J Polym Environ 9:63–84
Gietz DR, Woods RA (2002) Transformation of yeast by lithium acetate/single-stranded carrier DNA/polyethylene glycol method. Meth Enzymol 350:87–96
Ida Y, Furusawa C, Hirasawa T, Shimizu H (2012) Stable disruption of ethanol production by deletion of the genes encoding alcohol dehydrogenase isozymes in Saccharomyces cerevisiae. J Biosci Bioeng 113:192–195
Ilmén M, Koivuranta K, Ruohonen L, Suominen P, Penttilä M (2007) Efficient production of L-lactic acid from xylose by Pichia stipitis. Appl Environ Microbiol 73:117–123
Ishida N, Saitoh S, Tokuhiro K, Nagamori E, Matsuyama T, Kitamoto K, Takahashi H (2005) Efficient production of L-lactic acid by metabolically engineered Saccharomyces cerevisiae with a genome-integrated L-lactate dehydrogenase gene. Appl Environ Microbiol 71:1964–1970
Jin Y, Jeffries TW (2003) Changing flux of xylose metabolites by altering expression of xylose reductase and xylitol dehydrogenase in recombinant Saccharomyces cerevisiae. Appl Biochem Biotechnol 106:277–285
Jin Y, Ni H, Laplaza JM, Jeffries TW (2003) Optimal growth and ethanol production from xylose by recombinant Saccharomyces cerevisiae require moderate D-xylulokinase activity. Appl Environ Microbiol 69:495–503
Jin YS, Laplaza JM, Jeffries TW (2004) Saccharomyces cerevisiae engineered for xylose metabolism exhibits a respiratory response. Appl Environ Microbiol 70:6816–6825
Kim SR, Ha S, Kong II, Jin Y (2012) High expression of XYL2 coding for xylitol dehydrogenase is necessary for efficient xylose fermentation by engineered Saccharomyces cerevisiae. Metab Eng 14:336–343
Kim SR, Skerker JM, Kang W, Lesmana A, Wei N, Arkin AP, Jin Y (2013) Rational and evolutionary engineering approaches uncover a small set of genetic changes efficient for rapid xylose fermentation in Saccharomyces cerevisiae. PLoS One 8, e57048
Kötter P, Ciriacy M (1993) Xylose fermentation by Saccharomyces cerevisiae. Appl Microbiol Biotechnol 38:776–783
Kulkarni R, Moore E, Hegyeli A, Leonard F (1971) Biodegradable poly (lactic acid) polymers. J Biomed Mater Res 5:169–181
Kuyper M, Hartog MM, Toirkens MJ, Almering MJ, Winkler AA, Dijken JP, Pronk JT (2005) Metabolic engineering of a xylose‐isomerase‐expressing Saccharomyces cerevisiae strain for rapid anaerobic xylose fermentation. FEMS Yeast Res 5:399–409
Lodi T, Fontanesi F, Guiard B (2002) Co-ordinate regulation of lactate metabolism genes in yeast: the role of the lactate permease gene JEN1. Mol Gen Genomics 266:838–847
Maas RH, Bakker RR, Eggink G, Weusthuis RA (2006) Lactic acid production from xylose by the fungus Rhizopus oryzae. Appl Microbiol Biotechnol 72:861–868
Ostergaard S, Olsson L, Nielsen J (2000) Metabolic engineering of Saccharomyces cerevisiae. Microbiol Mol Biol Rev 64:34–50
Ozcan S, Johnston M (1999) Function and regulation of yeast hexose transporters. Microbiol Mol Biol Rev 63:554–569
Parekh RN, Shaw MR, Wittrup KD (1996) An integrating vector for tunable, high copy, stable integration into the dispersed Ty δ sites of Saccharomyces cerevisiae. Biotechnol Prog 12:16–21
Parekh RN, Wittrup KD (1997) Expression level tuning for optimal heterologous protein secretion in Saccharomyces cerevisiae. Biotechnol Prog 13:117–122
Payot T, Chemaly Z, Fick M (1999) Lactic acid production by Bacillus coagulans—kinetic studies and optimization of culture medium for batch and continuous fermentations. Enzyme Microb Technol 24:191–199
Pritchard G (1973) Factors affecting the activity and synthesis of NAD-dependent lactate dehydrogenase in Rhizopus oryzae. J Gen Microbiol 78:125–137
Qin J, Zhao B, Wang X, Wang L, Yu B, Ma Y, Ma C, Tang H, Sun J, Xu P (2009) Non-sterilized fermentative production of polymer-grade L-lactic acid by a newly isolated thermophilic strain Bacillus sp. 2–6. PLoS One 4, e4359
Roy A, Kim JH (2014) Endocytosis and vacuolar degradation of the yeast cell surface glucose sensors Rgt2 and Snf3. J Biol Chem
Saitoh S, Ishida N, Onishi T, Tokuhiro K, Nagamori E, Kitamoto K, Takahashi H (2005) Genetically engineered wine yeast produces a high concentration of L-lactic acid of extremely high optical purity. Appl Environ Microbiol 71:2789–2792
Sauer M, Porro D, Mattanovich D, Branduardi P (2008) Microbial production of organic acids: expanding the markets. Trends Biotechnol 26:100–108
Schmitt HD, Zimmermann FK (1982) Genetic analysis of the pyruvate decarboxylase reaction in yeast glycolysis. J Bacteriol 151:1146–1152
Skory CD (2003) Lactic acid production by Saccharomyces cerevisiae expressing a Rhizopus oryzae lactate dehydrogenase gene. J Ind Microbiol Biotechnol 30:22–27
Tamakawa H, Ikushima S, Yoshida S (2012) Efficient production of l-lactic acid from xylose by a recombinant Candida utilis strain. J Biosci Bioeng 113:73–75
Tylicki A, Ziolkowska G, Bolkun A, Siemieniuk M, Czerniecki J, Nowakiewicz A (2008) Comparative study of the activity and kinetic properties of malate dehydrogenase and pyruvate decarboxylase from Candida albicans, Malassezia pachydermatis, and Saccharomyces cerevisiae. Can J Microbiol 54:734–741
van Urk H, Schipper D, Breedveld GJ, Mak PR, Alexander Scheffers W, van Dijken JP (1989) Localization and kinetics of pyruvate-metabolizing enzymes in relation to aerobic alcoholic fermentation in Saccharomyces cerevisiae CBS 8066 and Candida utilis CBS 621. Biochimica et Biophysica Acta (BBA)-Gen Subjects 992:78–86
Wee Y, Kim J, Ryu H (2006) Biotechnological production of lactic acid and its recent applications. Food Technol Biotechnol 44:163
Zhang Z, Lohr L, Escalante C, Wetzstein M (2010) Food versus fuel: what do prices tell us? Energy Policy 38:445–451
Acknowledgments
TLT would like to thank the JBT Graduate Fellowship and acknowledge that this project was supported by the Agriculture and Food Research Initiative Competitive Grant No. 2015-67011-22806 from the USDA National Institute of Food and Agriculture. YSJ is affiliated with the Energy Biosciences Institute (EBI).
Compliance with ethical standards
ᅟ
Funding
This study was funded by the Agriculture and Food Research Initiative Competitive Grant No. 2016-67011-22806 from the USDA National Institute of Food and Agriculture. TLT received additional funding support from the University of Illinois at Urbana-Champaign’s JBT Graduate Fellowship.
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Timothy L. Turner and Guo-Chang Zhang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Turner, T.L., Zhang, GC., Kim, S.R. et al. Lactic acid production from xylose by engineered Saccharomyces cerevisiae without PDC or ADH deletion. Appl Microbiol Biotechnol 99, 8023–8033 (2015). https://doi.org/10.1007/s00253-015-6701-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-015-6701-3