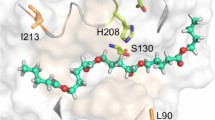
Abstract
Cutinases have been exploited for a broad range of reactions, from hydrolysis of soluble and insoluble esters to polymer synthesis. To further expand the biotechnological applications of cutinases for synthetic polyester degradation, we perform a comparative activity and stability analysis of five cutinases from Alternaria brassicicola (AbC), Aspergillus fumigatus (AfC), Aspergillus oryzae (AoC), Humicola insolens (HiC), and the well-characterized Fusarium solani (FsC). Of the cutinases, HiC demonstrated enhanced poly(ε-caprolactone) hydrolysis at high temperatures and under all pH values, followed by AoC and AfC. Both AbC and FsC are least stable and function poorly at high temperatures as well as at acidic pH conditions. Surface charge calculations and phylogenetic analysis reveal two important modes of cutinase stabilization: (1) an overall neutral surface charge within the “crowning area” by the active site and (2) additional disulfide bond formation. These studies provide insights useful for reengineering such enzymes with improved function and stability for a wide range of biotransformations.






Similar content being viewed by others
References
Alexov EG, Gunner MR (1997) Incorporating protein conformational flexibility into the calculation of pH-dependent protein properties. Biophys J 72:2075–2093
Araujo R, Silva C, O’Neill A, Micaelo N, Guebitz G, Soares CM, Casal M, Cavaco-Paulo A (2007) Tailoring cutinase activity towards polyethylene terephthalate and polyamide 6,6 fibers. J Biotechnol 128:849–857
Baptista RP, Santos AM, Fedorov A, Martinho JM, Pichot C, Elaissari A, Cabral JM, Taipa MA (2003) Activity, conformation and dynamics of cutinase adsorbed on poly(methyl methacrylate) latex particles. J Biotechnol 102:241–249
Bennett SJ, Pearson PJG (2009) From petrochemical complexes to biorefineries? The past and prospective co-evolution of liquid fuel and chemicals production in the UK. Chem Eng Res Des 87:1120–1139
Benson DA, Boguski MS, Lipman DJ, Ostell J (1997) Genbank. Nucleic Acid Res 25:1–6
Bornscheuer UT, Pohl M (2001) Improved biocatalysts by directed evolution and rational protein design. Curr Opin Chem Biol 5:137–143
Carvalho CM, Aires-Barros MR, Cabral JM (1999) Cutinase: from molecular level to bioprocess development. Biotechnol Bioeng 66:17–34
Chen Z, Franco CF, Baptista RP, Cabral JM, Coelho AV, Rodrigues CJ Jr, Melo EP (2007) Purification and identification of cutinases from Colletotrichum kahawae and Colletotrichum gloeosporioides. Appl Microbiol Biotechnol 73:1306–1313
Daggett V, Fersht A (2003) The present view of the mechanism of protein folding. Nat Rev Mol Cell Biol 4:497–502
de Barros DPC, Fonseca LP, Fernandes P, Cabral JMS, Mojovic L (2009) Biosynthesis of ethyl caproate and other short ethyl esters catalyzed by cutinase in organic solvents. J Mol Catal B Enzym 60:178–185
Eberl A, Heumann S, Kotek R, Kaufmann F, Mitsche S, Cavaco-Paulo A, Gübitz GM (2008) Enzymatic hydrolysis of PTT polymers and oligomers. J Biotechnol 135:45–51
Egmond MR, de Vlieg J (2000) Fusarium solani pisi cutinase. Biochimie 82:1015–1021
Fan C, Koller W (1998) Diversity of cutinases from plant pathogenic fungi: differential and sequential expression of cutinolytic esterases by Alternaria brassicicola. FEMS Microbiol Lett 158:33–38
Freire E, Murphy KP, Sanchez-Ruiz JM, Galisteo ML, Privalov PL (1992) The molecular basis of cooperativity in protein folding. Thermodynamic dissection of interdomain interactions in phosphoglycerate kinase. Biochemistry 31:250–256
Georgescu RE, Alexov EG, Gunner MR (2002) Combining conformational flexibility and continuum electrostatics for calculating pK(a)s in proteins. Biophys J 83:1731–1748
Goncalves AP, Cabral JM, Aires-Barros MR (1996) Immobilization of a recombinant cutinase by entrapment and by covalent binding. Kinetic and stability studies. Appl Biochem Biotechnol 60:217–228
Greenfield NJ (2004) Analysis of circular dichroism data. Method Enzymol 383:282–317
Kim YH, Seo HS, Min J, Kim YC, Ban YH, Han KY, Park JS, Bae KD, Gu MB, Lee J (2007) Enhanced degradation and toxicity reduction of dihexyl phthalate by Fusarium oxysporum f. sp. pisi cutinase. J Appl Microbiol 102:221–228
Kirk O, Borchert TV, Fuglsang CC (2002) Industrial enzyme applications. Curr Opin Biotechnol 13:345–351
Koschorreck K, Liu D, Kazenwadel C, Schmid RD, Hauer B (2010) Heterologous expression, characterization and site-directed mutagenesis of cutinase CUTAB1 from Alternaria brassicicola. Appl Microbiol Biotechnol 87:991–997
Liu Z, Gosser Y, Baker PJ, Ravee Y, Lu Z, Alemu G, Li H, Butterfoss GL, Kong XP, Gross R, Montclare JK (2009) Structural and functional studies of Aspergillus oryzae cutinase: enhanced thermostability and hydrolytic activity of synthetic ester and polyester degradation. J Am Chem Soc 13:15711–15716
Livesay DR, Jambeck P, Rojnuckarin A, Subramaniam S (2003) Conservation of electrostatic properties within enzyme families and superfamilies. Biochemistry 42:3464–3473
Longhi S, Czjzek M, Lamzin V, Nicolas A, Cambillau C (1997) Atomic resolution (1.0 A) crystal structure of Fusarium solani cutinase: stereochemical analysis. J Mol Biol 268:779–799
Maeda H, Yamagata Y, Abe K, Hasegawa F, Machida M, Ishioka R, Gomi K, Nakajima T (2005) Purification and characterization of a biodegradable plastic-degrading enzyme from Aspergillus oryzae. Appl Microbiol Biotechnol 67:778–788
Martinez C, De Geus P, Lauwereys M, Matthyssens G, Cambillau C (1992) Fusarium solani cutinase is a lipolytic enzyme with a catalytic serine accessible to solvent. Nature 356:615–618
Marti-Renom MA, Stuart AC, Fiser A, Sanchez R, Melo F, Sali A (2000) Comparative protein structure modeling of genes and genomes. Annu Rev Biophys Biomol Struct 29:291–325
Matsumura S, Soeda Y, Toshima K (2006) Perspectives for synthesis and production of polyurethanes and related polymers by enzymes directed toward green and sustainable chemistry. Appl Microbiol Biotechnol 70:12–20
Melo EP, Aires-Barros MR, Cabral JM (1995) Triglyceride hydrolysis and stability of a recombinant cutinase from Fusarium solani in AOT-iso-octane reversed micelles. Appl Biochem Biotechnol 50:45–56
Mooney BP (2009) The second green revolution? Production of plant-based biodegradable plastics. Biochem J 418:219–232
Murphy CA, Cameron JA, Huang SJ, Vinopal RT (1996) Fusarium polycaprolactone depolymerase is cutinase. Appl Environ Microbiol 62:456–460
Neves-Petersen MT, Petersen SB (2003) Protein electrostatics: a review of the equations and methods used to model electrostatic equations in biomolecules—applications in biotechnology. Biotechnol Annu Rev 9:315–395
Nyon MP, Rice DW, Berrisford JM, Hounslow AM, Moir AJ, Huang H, Nathan S, Mahadi NM, Bakar FD, Craven CJ (2009) Catalysis by Glomerella cingulata cutinase requires conformational cycling between the active and inactive states of its catalytic triad. J Mol Biol 385:226–235
Pace CN, Grimsley GR, Thomson JA, Barnett BJ (1988) Conformational stability and activity of ribonuclease T1 with zero, one, and two intact disulfide bonds. J Biol Chem 263:11820–11825
Petersen MTN, Fojan P, Petersen SB (2001a) How do lipases and esterases work: the electrostatic contribution. J Biotechnol 85:115–147
Petersen SB, Fojan P, Petersen EI, Petersen MT (2001b) The thermal stability of the Fusarium solani pisi cutinase as a function of pH. J Biomed Biotechnol 1:62–69
Pio TF, Macedo GA (2009) Cutinases: properties and industrial applications. Adv Appl Microbiol 66:77–95
Purdy RE, Kolattukudy PE (1973) Depolymerization of a hydroxy fatty acid biopolymer, cutin, by an extracellular enzyme from Fusarium solani f. pisi: isolation and some properties of the enzyme. Arch Biochem Biophys 159:61–69
Purdy RE, Kolattukudy PE (1975a) Hydrolysis of plant cuticle by plant pathogens. Properties of cutinase I, cutinase II, and a nonspecific esterase isolated from Fusarium solani pisi. Biochemistry 14:2832–2840
Purdy RE, Kolattukudy PE (1975b) Hydrolysis of plant cuticle by plant pathogens. Purification, amino acid composition, and molecular weight of two isozymes of cutinase and a nonspecific esterase from Fusarium solani f. pisi. Biochemistry 14:2824–2831
Quillaguaman J, Guzman H, Van-Thuoc D, Hatti-Kaul R (2009) Synthesis and production of polyhydroxyalkanoates by halophiles: current potential and future prospects. Appl Microbiol Biotechnol 85:1687–1696
Ripoll DR, Faerman CH, Axelsen PH, Silman I, Sussman JL (1993) An electrostatic mechanism for substrate guidance down the aromatic gorge of acetylcholinesterase. Proc Natl Acad Sci U S A 90:5128–5132
Ronkvist A, Lu W, Feder D, Gross RA (2009a) Cutinase-catalyzed deacetylation of poly(vinyl acetate). Macromolecules 42:6086–6097
Ronkvist A, Xie W, Lu W, Gross RA (2009b) Cutinase-catalyzed hydrolysis of poly(ethylene terephthalate). Macromolecules 42:5128–5138
Shah AA, Hasan F, Hameed A, Ahmed S (2008) Biological degradation of plastics: a comprehnesive review. Biotechnol Adv 26:246–265
Shashidhara KS, Gaikwad SM (2010) Conformational and functional transcription in class II α-mannoside from Aspergillus fischeri. J Fluoresc 20:827–836
Skamnioti P, Gurr SJ (2008) Cutinase and hydrophobin interplay: a herald for pathogenesis? Plant Signal Behav 3:248–250
Vidinha P, Harper N, Micaelo N, Lourenco NM, Gomaes da Silva MDR, Cabral JMS, Afonso CA, Soares CM, Barreiros S (2003) Effect on immobilization support, water, activity, and enzyme ionization state on cutinase activity and enantioselectivty in organic media. Biotechnol Bioeng 85:442–449
Vidinha P, Augusto V, Almeida M, Fonseca I, Fidalgo A, Ilharco L, Cabral JM, Barreiros S (2006) Sol-gel encapsulation: an efficient and versatile immobilization technique for cutinase in non-aqueous media. J Biotechnol 121:23–33
Acknowledgments
This work was supported in part by NYU:POLY Seed Fund (RG and JKM), AFOSR DURIP (FA-9550-08-1-0266) (JKM), NSF IUCRC (RG), and NSF GK-12 Fellows grant 0741714 (PJB). We thank Jeremy Minshull and Jonathan Ness from DNA 2.0 for their assistance in generating the cutinase DNA sequences as well as the production and purification of the enzymes.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplemental Materials
Below is the link to the electronic supplementary material.
ESM 1
(DOC 1267 kb)
Rights and permissions
About this article
Cite this article
Baker, P.J., Poultney, C., Liu, Z. et al. Identification and comparison of cutinases for synthetic polyester degradation. Appl Microbiol Biotechnol 93, 229–240 (2012). https://doi.org/10.1007/s00253-011-3402-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-011-3402-4