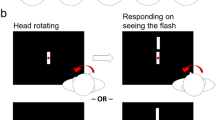
Abstract
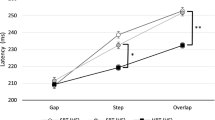
Knowing when the head moves is crucial information for the central nervous system to maintain a veridical representation of the self in the world for perception and action. Previous studies have shown that active head movement onset has to precede a sound by approximately 80 ms to be perceived as simultaneous, suggesting that the perceived timing of head movement is slow. However, this research was conducted with closed eyes. Given that visual information is available for most natural head movements, could perceptual delays in head movement onset be related to removing vision? Here, we examined whether visual information affects the perceived timing of active head movement onset. Participants performed a series of temporal order judgment tasks between their active head movement and an auditory tone presented at various stimulus onset asynchronies. Visual information was either absent (eyes closed) or present while either maintaining fixation on an earth or head-fixed target in the dark or in the light. Results show that head movement onset has to precede a sound by ~76 ms with eyes closed confirming previous work. The results also suggest that head movement onset must still precede a sound when fixating targets in the dark with a trend for the head having to move with less lead time with visual information and with the vestibulo-ocular reflex active or suppressed (~70 to 48 ms). Together, these results suggest that the perception of head movement onset is persistently delayed and is not fully resolved even with full field visual input.







Similar content being viewed by others
References
Angelaki DE, Cullen KE (2008) Vestibular system: the many facets of a multimodal sense. Annu Rev Neurosci 31:125–150
Aschersleben G, Müsseler J (1999) Dissociations in the timing of stationary and moving stimuli. J Exp Psychol Hum Percept Perform 25:1709–1720
Barnett-Cowan M (2013) Vestibular perception is slow: a review. Multisens Res 26:387–403
Barnett-Cowan M, Harris LR (2009) Perceived timing of vestibular stimulation relative to touch, light and sound. Exp Brain Res 198:221–231
Barnett-Cowan M, Harris LR (2011) Temporal processing of active and passive head movement. Exp Brain Res 214:27–35
Barnett-Cowan M, Raeder SM, Bülthoff HH (2012) Persistent perceptual delay for head movement onset relative to auditory stimuli of different durations and rise times. Exp Brain Res 220:41–50
Brooks JX, Cullen KE (2014) Early vestibular processing does not discriminate active from passive self-motion if there is a discrepancy between predicted and actual proprioceptive feedback. J Neurophysiol 111:2465–2478
Butler JS, Smith ST, Campos JL, Bülthoff HH (2010) Bayesian integration of visual and vestibular signals for heading. J Vis 10:23
Carriot J, Brooks JX, Cullen KE (2013) Multimodal integration of self-motion cues in the vestibular system: active versus passive translations. J Neurosci 33:19555–19566
Clemens IAH, Selen LP, Pomante A, MacNeilage PR, Medendorp WP (2017) Eye movements in darkness modulate self-motion perception. ENeuro 4:1–12
Clément G, Maciel F (2004) Adjustment of the vestibulo-ocular reflex gain as a function of perceived target distance in humans. Neurosci Lett 366:115–119
Cullen KE (2012) The vestibular system: multimodal integration and encoding of self-motion for motor control. Trends Neurosci 35:185–196
Fetsch CR, Turner AH, DeAngelis GC, Angelaki DE (2009) Dynamic reweighting of visual and vestibular cues during self-motion perception. J Neurosci 29:15601–15612
Figliozzi F, Guariglia P, Silvetti M, Siegler I, Doricchi F (2005) Effects of vestibular rotatory accelerations on covert attentional orienting in vision and touch. J Cogn Neurosci 17:1638–1651
Hine T, Thorn F (1987) Compensatory eye movements during active head rotation for near targets: effects of imagination, rapid head oscillation and vergence. Vis Res 27:1639–1657
Huisman M (2000) Imputation of missing item responses: some simple techniques. Qual Quant 34:331–351
Karmali F, Lim K, Merfeld DM (2014) Visual and vestibular perceptual thresholds each demonstrate better precision at specific frequencies and also exhibit optimal integration. J Neurophysiol 111:2393–2403
Kim J, Barnett-Cowan M, Macpherson EA (2013) Integration of auditory input with vestibular and neck proprioceptive information in the interpretation of dynamic sound localization cues. In: Proceedings of meetings on acoustics ICA2013, vol 19, no 1. ASA, p 050142
MacNeilage PR, Banks MS, Berger DR, Bülthoff HH (2007) A Bayesian model of the disambiguation of gravitoinertial force by visual cues. Exp Brain Res 179:263–290
Medendorp WP, Van Gisbergen JAM, Van Pelt S, Gielen CCAM (2000) Context compensation in the vestibuloocular reflex during active head rotations. J Neurophysiol 84:2904–2917
Medrea I, Cullen KE (2013) Multisensory integration in early vestibular processing in mice: the encoding of passive versus active motion. J Neurophysiol 110:2704–2717
Ohmi M (1996) Egocentric perception through interaction among many sensory systems. Cogn Brain Res 5:87–96
Paige GD, Telford L, Seidman SH, Barnes GR (1998) Human vestibuloocular reflex and its interactions with vision and fixation distance during linear and angular head movement. J Neurophysiol 80:2391–2404
Riecke BE, Cunningham DW, Bülthoff HH (2007) Spatial updating in virtual reality: the sufficiency of visual information. Psychol Res 71:298–313
Roy JE, Cullen KE (2001) Selective processing of vestibular reafference during self-generated head motion. J Neurosci 21:2131–2142
Roy JE, Cullen KE (2004) Dissociating self-generated from passively applied head motion: neural mechanisms in the vestibular nuclei. J Neurosci 24:2102–2111
Royden CS, Banks MS, Crowell JA (1992) The perception of heading during eye movements. Nature 360:583–585
Sanders MC, Chang NYN, Hiss MM, Uchanski RM, Hullar TE (2011) Temporal binding of auditory and rotational stimuli. Exp Brain Res 210:539–547
Soyka F, Giordano PR, Barnett-Cowan M, Bülthoff HH (2012) Modeling direction discrimination thresholds for yaw rotations around an earth-vertical axis for arbitrary motion profiles. Exp Brain Res 220:89–99
Tong J, Patel SS, Bedell HE (2006) The attenuation of perceived motion smear during combined eye and head movements. Vis Res 46:4387–4397
Tong J, Aydin M, Bedell HE (2007) Direction-of-motion discrimination is facilitated by visible motion smear. Atten Percept Psychophys 69:48–55
von Holst E, Mittelstaedt H (1950) Das Reafferenzprinzip. Naturwissenschaften 37:464–476
Warren WH, Hannon DJ (1988) Direction of self-motion is perceived from optical flow. Nature 336:162–163
Acknowledgements
This work was generously supported by a Natural Sciences and Engineering Research Council of Canada (NSERC) Discovery Grant (#RGPIN-05435-2014) and a Canadian Foundation for Innovation (CFI) John R. Evans Leaders Fund Grant (#32618) to MB-C. We thank Adrienne Wise for helping to test participants and Jessy Parokaran Varghese for comments on an earlier version of the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Chung, W., Barnett-Cowan, M. Persistent perceptual delay for active head movement onset relative to sound onset with and without vision. Exp Brain Res 235, 3069–3079 (2017). https://doi.org/10.1007/s00221-017-5026-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-017-5026-0