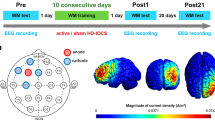
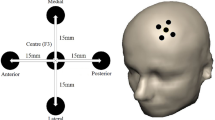
Abstract
Neuroimaging and electrophysiological studies have unequivocally identified the dorsolateral prefrontal cortex (DLPFC) as a crucial structure for top-down control of working memory (WM) processes. By modulating the excitability of neurons in a targeted cortical area, transcranial magnetic stimulation (TMS) offers a unique way to modulate DLPFC function, opening the possibility of WM facilitation. Even though TMS neuromodulation effects over the left DLPFC have successfully improved WM performance in patients with depression and schizophrenia in a multitude of studies, raising the potential of TMS as a safe efficacious treatment for WM deficits, TMS interventions in healthy individuals have produced mixed and inconclusive results. Here, we stimulated the left DLPFC of healthy individuals using a high-frequency repetitive TMS protocol and evaluated behavioral performance in a battery of cognitive tasks. We found that TMS treatment enhanced WM performance in a verbal digit span and a visuospatial 2-back task.


Similar content being viewed by others
References
Aben B, Stapert S, Blokland A (2012) About the distinction between working memory and short-term memory. Front Psychol 3(301):1–9
Andrews SC, Hoy KE, Enticott PG, Daskalakis ZJ, Fitzgerald PB (2011) Improving working memory: the effect of combining cognitive activity and anodal transcranial direct current stimulation to the left dorsolateral prefrontal cortex. Brain Stimul 4(2):84–89
Arnsten AF, Li BM (2005) Neurobiology of executive functions: catecholamine influences on prefrontal cortical functions. Biol Psychiatry 57(11):1377–1384
Balconi M (2013) Dorsolateral prefrontal cortex, working memory and episodic memory processes: insight through transcranial magnetic stimulation techniques. Neurosci Bull 29(3):381–389
Barch DM, Sheline YI, Csernansky JG, Snyder AZ (2003) Working memory and prefrontal cortex dysfunction: specificity to schizophrenia compared with major depression. Biol Psychiatry 53(5):376–384
Barr MS, Farzan F, Rajji TK, Voineskos AN, Blumberger DM, Arenovich T, Fitzgerald PB, Daskalakis ZJ (2013) Can repetitive magnetic stimulation improve cognition in schizophrenia? Pilot data from a randomized controlled trial. Biol Psychiatry 73(6):510–517
Beam W, Borckardt JJ, Reeves ST, George MS (2009) An efficient and accurate new method for locating the F3 position for prefrontal TMS applications. Brain Stimul 2(1):50–54
Bloch Y, Grisaru N, Harel EV, Beitler G, Faivel N, Ratzoni G, Stein D, Levkovitz Y (2008) Repetitive transcranial magnetic stimulation in the treatment of depression in adolescents: an open-label study. J ECT 24(2):156–159
Boggio PS, Fregni F, Bermpohl F, Mansur CG, Rosa M, Rumi DO, Barbosa ER, Rosa MO, Pascual-Leone A, Rigonatti SP, Marcolin MA, Araujo Silva MT (2005) Effect of repetitive TMS and fluoxetine on cognitive function in patients with Parkinson’s disease and concurrent depression. Mov Disord 20(9):1178–1184
Boroojerdi B, Phipps M, Kopylev L, Wharton CM, Cohen LG, Grafman J (2001) Enhancing analogic reasoning with rTMS over the left prefrontal cortex. Neurology 56(4):526–528
Bostrom N, Sandberg A (2009) Cognitive enhancement: methods, ethics, regulatory challenges. Sci Eng Ethics 15(3):311–341
Brunoni AR, Vanderhasselt MA (2014) Working memory improvement with non-invasive brain stimulation of the dorsolateral prefrontal cortex: a systematic review and meta-analysis. Brain Cogn 86:1–9
Chen YN, Mitra S (2009) The spatial-verbal difference in the n-back task: an ERP study. Acta Neurol Taiwanica 18(3):170–179
Chervyakov A, Sinitsyn D, Chernyavsky A, Piradov M (2015) Possible mechanisms underlying the therapeutic effects of transcranial magnetic stimulation. Front Hum Neurosci 9:303
Cho SS, Strafella AP (2009) rTMS of the left dorsolateral prefrontal cortex modulates dopamine release in the ipsilateral anterior cingulate cortex and orbitofrontal cortex. PLoS One 4(8):e6725
D’Esposito M, Aguirre GK, Zarahn E, Ballard D, Shin RK, Lease J (1998) Functional MRI studies of spatial and nonspatial working memory. Cogn Brain Res 7(1):1–13
De Jongh R, Bolt I, Schermer M, Olivier B (2008) Botox for the brain: enhancement of cognition, mood and pro-social behavior and blunting of unwanted memories. Neurosci Biobehav Rev 32(4):760–776
Demirtas-Tatlidede A, Vahabzadeh-Hagh AM, Pascual-Leone A (2013) Can noninvasive brain stimulation enhance cognition in neuropsychiatric disorders? Neuropharmacology 64:566–578
Dickstein DP, Treland JE, Snow J, McClure EB, Mehta MS, Towbin KE, Pine DS, Leibenluft E (2004) Neuropsychological performance in pediatric bipolar disorder. Biol Psychiatry 55(1):32–39
Dresler M, Sandberg A, Ohla K, Bublitz C, Trenado C, Mroczko-Wąsowicz A, Kuhn S, Repantis D (2013) Non-pharmacological cognitive enhancement. Neuropharmacology 64:529–543
Edin F, Klingberg T, Johansson P, McNab F, Tegnér J, Compte A (2009) Mechanism for top-down control of working memory capacity. Proc Natl Acad Sci 106(16):6802–6807
Epstein CM, Sekino M, Yamaguchi K, Kamiya S, Ueno S (2002) Asymmetries of prefrontal cortex in human episodic memory: effects of transcranial magnetic stimulation on learning abstract patterns. Neurosci Lett 320(1):5–8
Esslinger C, Schüler N, Sauer C, Gass D, Mier D, Braun U, Ochs E, Schulze T, Rietschel M, Kirsh P, Meyer-Lindenberg A (2014) Induction and quantification of prefrontal cortical network plasticity using 5 Hz rTMS and fMRI. Hum Brain Mapp 35(1):140–151
Fabre I, Galinowski A, Oppenheim C, Gallarda T, Meder JF, De Montigny C, Olie JP, Poirier MF (2004) Antidepressant efficacy and cognitive effects of repetitive transcranial magnetic stimulation in vascular depression: an open trial. Int J Geriatr Psychiatry 19(9):833–842
Floel A, Poeppel D, Buffalo EA, Braun A, Wu CWH, Seo HJ, Stefan K, Knecht S, Cohen LG (2004) Prefrontal cortex asymmetry for memory encoding of words and abstract shapes. Cereb Cortex 14(4):404–409
Fregni F, Pascual-Leone A (2007) Technology insight: noninvasive brain stimulation in neurology—perspectives on the therapeutic potential of rTMS and tDCS. Nat Clin Pract Neurol 3(7):383–393
Fregni F, Boggio PS, Nitsche M, Bermpohl F, Antal A, Feredoes E, Marcoli MA, Rigonatti SP, Silva MTA, Paulus W, Pascual-Leone A (2005) Anodal transcranial direct current stimulation of prefrontal cortex enhances working memory. Exp Brain Res 166(1):23–30
Fujiki M, Kobayashi H, Abe T, Kamida T (2003) Repetitive transcranial magnetic stimulation for protection against delayed neuronal death induced by transient ischemia. J Neurosurg 99(6):1063–1069
Funamizu H, Ogiue-Ikeda M, Mukai H, Kawato S, Ueno S (2005) Acute repetitive transcranial magnetic stimulation reactivates dopaminergic system in lesion rats. Neurosci Lett 383(1):77–81
Gaudeau-Bosma C, Moulier V, Allard AC, Sidhoumi D, Bouaziz N, Braha S, Volle E, Januel D (2013) Effect of two weeks of rTMS on brain activity in healthy subjects during an n-back task: a randomized double blind study. Brain Stimul 6(4):569–575
Gazzaley A, Nobre AC (2012) Top-down modulation: bridging selective attention and working memory. Trends Cogn Sci 16(2):129–135
Grosbras MH, Paus T (2002) Transcranial magnetic stimulation of the human frontal eye field: effects on visual perception and attention. J Cogn Neurosci 14(7):1109–1120
Guse B, Falkai P, Wobrock T (2010) Cognitive effects of high-frequency repetitive transcranial magnetic stimulation: a systematic review. J Neural Transm 117(1):105–122
Guse B, Falkai P, Gruber O, Whalley H, Gibson L, Hasan A, Obst K, Dechent P, Mclntosh A, Suchan B, Wobrock T (2013) The effect of long-term high frequency repetitive transcranial magnetic stimulation on working memory in schizophrenia and healthy controls—a randomized placebo-controlled, double-blind fMRI study. Behav Brain Res 237:300–307
Hamidi M, Slagter HA, Tononi G, Postle BR (2009) Repetitive transcranial magnetic stimulation affects behavior by biasing endogenous cortical oscillations. Front Integr Neurosci 3(14):1–12
Hausmann A, Weis C, Marksteiner J, Hinterhuber H, Humpel C (2000) Chronic repetitive transcranial magnetic stimulation enhances c-fos in the parietal cortex and hippocampus. Mol Brain Res 76(2):355–362
Hausmann A, Kemmler G, Walpoth M, Mechtcheriakov S, Kramer-Reinstadler K, Lechner T, Walsh T, Deisenhammer EA, Kofler M, Rupp CI, Hinterhuber H, Conca A (2004) No benefit derived from repetitive transcranial magnetic stimulation in depression: a prospective, single centre, randomised, double blind, sham controlled “add on” trial. J Neurol Neurosurg Psychiatry 75(2):320–322
Hilgetag CC, Théoret H, Pascual-Leone A (2001) Enhanced visual spatial attention ipsilateral to rTMS-induced ‘virtual lesions’ of human parietal cortex. Nat Neurosci 4(9):953–957
Hill EL (2004) Executive dysfunction in autism. Trends Cogn Sci 8(1):26–32
Hodsoll J, Mevorach C, Humphreys GW (2009) Driven to less distraction: rTMS of the right parietal cortex reduces attentional capture in visual search. Cereb Cortex 19(1):106–114
Hoy KE, Fitzgerald PB (2010) Brain stimulation in psychiatry and its effects on cognition. Nat Rev Neurol 6(5):267–275
Hoy KE, Bailey N, Michael M, Fitzgibbon B, Rogasch NC, Saeki T, Fitzgerald PB (2015) Enhancement of working memory and task-related oscillatory activity following intermittent theta burst stimulation in healthy controls. Cerebral Cortex. doi:10.1093/cercor/bhv193
Kirschen MP, Davis-Ratner MS, Jerde TE, Schraedley-Desmond P, Desmond JE (2006) Enhancement of phonological memory following transcranial magnetic stimulation (TMS). Behav Neurol 17(3–4):187–194
Klimesch W, Sauseng P, Gerloff C (2003) Enhancing cognitive performance with repetitive transcranial magnetic stimulation at human individual alpha frequency. Eur J Neurosci 17(5):1129–1133
Ko JH, Monchi O, Ptito A, Bloomfield P, Houle S, Strafella AP (2008) Theta burst stimulation-induced inhibition of dorsolateral prefrontal cortex reveals hemispheric asymmetry in striatal dopamine release during a set-shifting task—a TMS-[11C] raclopride PET study. Eur J Neurosci 28(10):2147–2155
Kojima S, Goldman-Rakic PS (1982) Delay-related activity of prefrontal neurons in rhesus monkeys performing delayed response. Brain Res 248(1):43–50
Koski L, Molnar-Szakacs I, Iacoboni M (2005) Exploring the contributions of premotor and parietal cortex to spatial compatibility using image-guided TMS. Neuroimage 24(2):296–305
Krishnan C, Santos L, Peterson MD, Ehinger M (2015) Safety of noninvasive brain stimulation in children and adolescents. Brain Stimul 8(1):76–87
Kuroda Y, Motohashi N, Ito H, Ito S, Takano A, Nishikawa T, Suhara T (2006) Effects of repetitive transcranial magnetic stimulation on [11 C] raclopride binding and cognitive function in patients with depression. J Affect Disord 95(1):35–42
Levkovitz Y, Rabany L, Harel EV, Zangen A (2011) Deep transcranial magnetic stimulation add-on for treatment of negative symptoms and cognitive deficits of schizophrenia: a feasibility study. Int J Neuropsychopharmacol 14(7):991–996
Lisanby SH, Belmaker RH (2000) Animal models of the mechanisms of action of repetitive transcranial magnetic stimulation(RTMS): comparisons with electroconvulsive shock(ECS). Depress Anxiety 12(3):178–187
Lisanby SH, Gutman D, Luber B, Schroeder C, Sackeim HA (2001) Sham TMS: intracerebral measurement of the induced electrical field and the induction of motor-evoked potentials. Biol Psychiatry 49(5):460–463
Lowe C, Rabbitt P (1998) Test re-test reliability of the CANTAB and ISPOCD neuropsychological batteries: theoretical and practical issues. Neuropsychologia 36(9):915–923
Luber B, Lisanby SH (2014) Enhancement of human cognitive performance using transcranial magnetic stimulation (TMS). Neuroimage 85:961–970
Machii K, Cohen D, Ramos-Estebanez C, Pascual-Leone A (2006) Safety of rTMS to non-motor cortical areas in healthy participants and patients. Clin Neurophysiol 117(2):455–471
Maeda F, Keenan JP, Tormos JM, Topka H, Pascual-Leone A (2000) Interindividual variability of the modulatory effects of repetitive transcranial magnetic stimulation on cortical excitability. Exp Brain Res 133(4):425–430
McKinley RA, Bridges N, Walters CM, Nelson J (2012) Modulating the brain at work using noninvasive transcranial stimulation. Neuroimage 59(1):129–137
Meegan DV, Honsberger MJ (2005) Spatial information is processed even when it is task-irrelevant: implications for neuroimaging task design. NeuroImage 25(4):1043–1055
O’Connor M, Brenninkmeyer C, Morgan A, Bloomingdale K, Thall M, Vasile R, Leone AP (2003) Relative effects of repetitive transcranial magnetic stimulation and electroconvulsive therapy on mood and memory: a neurocognitive risk–benefit analysis. Cogn Behav Neurol 16(2):118–127
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9(1):97–113
Owen AM, Evans AC (1996) Evidence for a two-stage model of spatial working memory processing within the lateral frontal cortex: a positron emission tomography study. Cerebral Cortex 6(1):31–38
Owen AM, McMillan KM, Laird AR, Bullmore E (2005) N-back working memory paradigm: a meta-analysis of normative functional neuroimaging studies. Hum Brain Mapp 25(1):46–59
Paivio A (1991) Dual coding theory: retrospect and current status. Can J Psychol 45(3):255
Pascual-Leone A, Hallett M (1994) Induction of errors in a delayed response task by repetitive transcranial magnetic stimulation of the dorsolateral prefrontal cortex. NeuroReport 5(18):2517–2520
Pascual-Leone A, Tormos JM, Keenan J, Tarazona F, Cañete C, Catalá MD (1998) Study and modulation of human cortical excitability with transcranial magnetic stimulation. J Clin Neurophysiol 15(4):333–343
Preston G, Anderson E, Silva C, Goldberg T, Wassermann EM (2010) Effects of 10 Hz rTMS on the neural efficiency of working memory. J Cogn Neurosci 22(3):447–456
Quednow BB (2010) Ethics of neuroenhancement: a phantom debate. BioSocieties 5(1):153
Radhu N, de Jesus DR, Ravindran LN, Zanjani A, Fitzgerald PB, Daskalakis ZJ (2013) A meta-analysis of cortical inhibition and excitability using transcranial magnetic stimulation in psychiatric disorders. Clin Neurophysiol 124(7):1309–1320
Robbins TW, James M, Owen AM, Sahakian BJ, McInnes L, Rabbitt P (1994) Cambridge Neuropsychological Test Automated Battery (CANTAB): a factor analytic study of a large sample of normal elderly volunteers. Dement Geriatr Cogn Disord 5(5):266–281
Rossi S, Hallett M, Rossini PM, Pascual-Leone A, Safety of TMS Consensus Group (2009) Safety, ethical considerations, and application guidelines for the use of transcranial magnetic stimulation in clinical practice and research. Clin Neurophysiol 120(12):2008–2039
Sandberg A, Bostrom N (2006) Converging cognitive enhancements. Ann N Y Acad Sci 1093(1):201–227
Sandrini M, Rossini PM, Miniussi C (2008) Lateralized contribution of prefrontal cortex in controlling task-irrelevant information during verbal and spatial working memory tasks: rTMS evidence. Neuropsychologia 46(7):2056–2063
Sauseng P, Klimesch W, Heise KF, Gruber WR, Holz E, Karim AA, Hummel FC (2009) Brain oscillatory substrates of visual short-term memory capacity. Curr Biol 19(21):1846–1852
Schulze-Rauschenbach SC, Harms U, Schlaepfer TE, Maier W, Falkai P, Wagner M (2005) Distinctive neurocognitive effects of repetitive transcranial magnetic stimulation and electroconvulsive therapy in major depression. Br J Psychiatry 186(5):410–416
Skrdlantova L, Horacek J, Dockery C, Lukavsky J, Kopecek M, Preiss M, Novak T, Hoschl C (2005) The influence of low-frequency left prefrontal repetitive transcranial magnetic stimulation on memory for words but not for faces. Physiol Res 54(1):123–128
Snyder AW, Mulcahy E, Taylor JL, Mitchell DJ, Sachdev P, Gandevia SC (2003) Savant-like skills exposed in normal people by suppressing the left fronto-temporal lobe. J Integrat Neurosci 2(02):149–158
Stahl SM (2013) Stahl’s essential psychopharmacology: neuroscientific basis and practical applications. Cambridge University Press, Cambridge
Strafella AP, Paus T, Barrett J, Dagher A (2001) Repetitive transcranial magnetic stimulation of the human prefrontal cortex induces dopamine release in the caudate nucleus. J Neurosci 21(15):1–4
Taghavi SMR (2002) Validity and reliability of the general health questionnaire (ghq-28) in college students of Shiraz University. J Psychol 5(4):381–398
Tavares JVT, Clark L, Cannon DM, Erickson K, Drevets WC, Sahakian BJ (2007) Distinct profiles of neurocognitive function in unmedicated unipolar depression and bipolar II depression. Biol Psychiatry 62(8):917–924
Tegenthoff M, Ragert P, Pleger B, Schwenkreis P, Forster A, Nicolas V, Dinse HR (2005) Improvement of tactile discrimination performance and enlargement of cortical somatosensory maps after 5 Hz rTMS. PLoS Biol 3(11):2031
Thut G, Nietzel A, Pascual-Leone A (2005) Dorsal posterior parietal rTMS affects voluntary orienting of visuospatial attention. Cereb Cortex 15(5):628–638
Thut G, Miniussi C (2009) New insights into rhythmic brain activity from TMS–EEG studies. Trends in Cognitive Sciences 13(4):182–189
Vanderhasselt MA, De Raedt R, Leyman L, Baeken C (2009) Acute effects of repetitive transcranial magnetic stimulation on attentional control are related to antidepressant outcomes. J Psychiatry Neurosci 34(2):119
Wagenmakers EJ, Van Der Maas HL, Grasman RP (2007) An EZ-diffusion model for response time and accuracy. Psychon Bull Rev 14(1):3–22
Wager TD, Smith EE (2003) Neuroimaging studies of working memory. Cogn Affect Behav Neurosci 3(4):255–274
Walsh V, Ellison A, Battelli L, Cowey A (1998) Task–specific impairments and enhancements induced by magnetic stimulation of human visual area V5. Proc R Soc Lond B Biol Sci 265(1395):537–543
Wechsler D (1981) The psychometric tradition: developing the Wechsler adult intelligence scale. Contemp Educ Psychol 6(2):82–85
Yekutieli D, Benjamini Y (1999) Resampling-based false discovery rate controlling multiple test procedures for correlated test statistics. J Stat Plan Infer 82(1–2):171–196
Young RL, Ridding MC, Morrell TL (2004) Switching skills on by turning off part of the brain. Neurocase 10(3):215–222
Zanto TP, Rubens MT, Thangavel A, Gazzaley A (2011) Causal role of the prefrontal cortex in top-down modulation of visual processing and working memory. Nat Neurosci 14(5):656–661
Acknowledgments
We are grateful to Santani Teng for assistance in editing this manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bagherzadeh, Y., Khorrami, A., Zarrindast, M.R. et al. Repetitive transcranial magnetic stimulation of the dorsolateral prefrontal cortex enhances working memory. Exp Brain Res 234, 1807–1818 (2016). https://doi.org/10.1007/s00221-016-4580-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-016-4580-1