Abstract
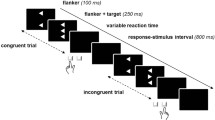
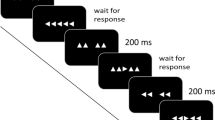
The concept of segregated basal ganglia–cortical loops entails that functional disturbances may result from abnormal processing within loops, but also from abnormal communication between loops. Cognitive and motor processes subserved by different basal ganglia–frontal loops may interfere with one another as a result of such abnormal communication, leakage, between loops. In Parkinson’s disease, movement execution has been found susceptible to decision uncertainty, attributed to this mechanism. Here, we evaluate whether this mechanism of abnormal coupling or leakage extends to perceptual decision-making with trial-by-trial control of decision uncertainty. We examined 10 Parkinson’s disease (PD) patients and healthy control subjects in a random-dot motion direction discrimination task with concurrent EEG recording. Random-dot motion was manipulated to make direction discrimination easy or difficult. Reaction times (RT) and movement times (MT) were recorded, and EEG was analysed to extract movement-related potentials. Easy versus difficult direction discrimination produced robust, equally large RT differences in patients and controls (>400 ms), along with a marked difference in error rates, confirming the efficacy of the task. Effects of easy versus difficult discrimination on MT were comparatively small (<50 ms) and did not differ between groups, despite robustly slower MT in patients. Lateralised movement-related EEG potentials reproduced the MT difference between patients and controls. Together, the results do not demonstrate an enhanced effect of decision uncertainty onto movement execution in PD. We surmise that leakage of decision uncertainty into movement execution is probably task-dependent, consistent with the view that the degree to which partial information is allowed to influence the motor system is under strategic control.



Similar content being viewed by others
References
Alexander GE, DeLong MR, Strick PL (1986) Parallel organization of functionally segregated circuits linking basal ganglia and cortex. Annu Rev Neurosci 9:357–381
Bar-Gad I, Bergman H (2001) Stepping out of the box: information processing in the neural networks of the basal ganglia. Curr Opin Neurobiol 11:689–695
Berg P, Scherg M (1994) A multiple source approach to the correction of eye artifacts. Electroencephalogr Clin Neurophysiol 90:229–241
Bergman H, Feingold A, Nini A, Raz A, Slovin H, Abeles M, Vaadia E (1998) Physiological aspects of information processing in the basal ganglia of normal and parkinsonian primates. Trends Neurosci 21:32–38
Brainard DH (1997) The psychophysics toolbox. Spat Vis 10:433–436
Burle B, Vidal F, Tandonnet C, Hasbroucq T (2004) Physiological evidence for response inhibition in choice reaction time tasks. Brain Cogn 56:153–164
Caligiuri MP, Heindel WC, Lohr JB (1992) Sensorimotor disinhibition in Parkinson’s disease: effects of levodopa. Ann Neurol 31:53–58
Cheyne DO (2013) MEG studies of sensorimotor rhythms: a review. Exp Neurol 245:27–39
Coles MGH, Smid HGOM, Scheffers MK, Otten LJ (1995) Mental chronometry and the study of human information processing. In: Rugg MD, Coles MGH (eds) Electrophysiology of mind: event-related brain potentials and cognition. Oxford University Press, Oxford, pp 86–131
De Lange FP, Rahnev DA, Donner TH, Lau H (2013) Pre-stimulus oscillatory activity over motor cortex reflects perceptual expectations. J Neurosci 33:1400–1410
Donner TH, Siegel M, Fries P, Engel AK (2009) Buildup of choice-predictive activity in human motor cortex during perceptual decision making. Curr Biol 19:1581–1585
Draganski B, Kherif F, Klöppel S, Cook PA, Alexander DC, Parker GJ, Deichmann R, Ashburner J, Frackowiak RS (2008) Evidence for segregated and integrative connectivity patterns in the human Basal Ganglia. J Neurosci 28:7143–7152
Filion M, Tremblay L, Bédard PJ (1988) Abnormal influences of passive limb movement on the activity of globus pallidus neurons in parkinsonian monkeys. Brain Res 444:165–176
Filion M, Tremblay L, Matsumura M, Richard H (1994) Dynamic focusing of informational convergence in basal ganglia. Rev Neurol (Paris) 150:627–633
Gold JI, Shadlen MN (2007) The neural basis of decision making. Annu Rev Neurosci 30:535–574
Gratton G, Coles MG, Sirevaag EJ, Eriksen CW, Donchin E (1988) Pre- and poststimulus activation of response channels: a psychophysiological analysis. J Exp Psychol Hum Percept Perform 14:331–344
Guthrie M, Leblois A, Garenne A, Boraud T (2013) Interaction between cognitive and motor cortico-basal ganglia loops during decision making: a computational study. J Neurophysiol 109:3025–3040
Haber SN (2003) The primate basal ganglia: parallel and integrative networks. J Chem Neuroanat 26:317–330
Hughes AJ, Daniel SE, Kilford L, Lees AJ (1992) Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: a clinico-pathological study of 100 cases. J Neurol Neurosurg Psychiatry 55:181–184
Liu T, Pleskac TJ (2011) Neural correlates of evidence accumulation in a perceptual decision task. J Neurophysiol 106:2383–2398
Lord S, Rochester L, Hetherington V, Allcock LM, Burn D (2010) Executive dysfunction and attention contribute to gait interference in ‘off’ state Parkinson’s disease. Gait Posture 31:169–174
Malapani C, Pillon B, Dubois B, Agid Y (1994) Impaired simultaneous cognitive task performance in Parkinson’s disease: a dopamine-related dysfunction. Neurology 44:319–326
Mazzoni P, Hristova A, Krakauer JW (2007) Why don’t we move faster? Parkinson’s disease, movement vigor, and implicit motivation. J Neurosci 27:7105–7116
Middleton FA, Strick PL (2002) Basal-ganglia ‘projections’ to the prefrontal cortex of the primate. Cereb Cortex 12:926–935
Oostenveld R, Praamstra P (2001) The five percent electrode system for high resolution EEG and ERP measurements. Clin Neurophysiol 112:713–719
Pelli DG (1997) The VideoToolbox software for visual psychophysics: transforming numbers into movies. Spat Vis 10:437–442
Pessiglione M, Czernecki V, Pillon B, Dubois B, Schüpbach M, Agid Y, Tremblay L (2005) An effect of dopamine depletion on decision-making: the temporal coupling of deliberation and execution. J Cogn Neurosci 17:1886–1896
Praamstra P, Plat FM (2001) Failed suppression of direct visuomotor activation in Parkinson’s disease. J Cogn Neurosci 13:31–43
Praamstra P, Seiss E (2005) The neurophysiology of response competition: motor cortex activation and inhibition following subliminal response priming. J Cogn Neurosci 17:483–493
Praamstra P, Stegeman DF, Horstink MWIM, Cools AR (1996) Dipole source analysis suggests selective modulation of the supplementary motor area contribution to the Readiness Potential. Electroencephalogr Clin Neurophysiol 98:468–477
Redgrave P, Rodriguez M, Smith Y, Rodriguez-Oroz MC, Lehericy S, Bergman H, Agid Y, DeLong MR, Obeso JA (2010) Goal-directed and habitual control in the basal ganglia: implications for Parkinson’s disease. Nat Rev Neurosci 11:760–772
Roitman JD, Shadlen MN (2002) Response of neurons in the lateral intraparietal area during a combined visual discrimination reaction time task. J Neurosci 22:9475–9489
Shadlen MN, Newsome WT (1996) Motion perception: seeing and deciding. Proc Natl Acad Sci U S A 93:628–633
Shadlen MN, Britten KH, Newsome WT, Movshon JA (1996) A computational analysis of the relationship between neuronal and behavioral responses to visual motion. J Neurosci 16:1486–1510
Song JH, Nakayama K (2009) Hidden cognitive states revealed in choice reaching tasks. Trends Cogn Sci 13:360–366
Tandonnet C, Garry MI, Summers JJ (2011) Selective suppression of the incorrect response implementation in choice behavior assessed by transcranial magnetic stimulation. Psychophysiology 48:462–469
Tomlinson CL, Stowe R, Patel S, Rick C, Gray R, Clarke CE (2010) Systematic review of levodopa dose equivalency reporting in Parkinson’s disease. Mov Disord 25:2649–2653
Ulrich R, Miller J (2001) Using the jackknife-based scoring method for measuring LRP onset effects in factorial designs. Psychophysiology 38:816–827
van den Wildenberg WP, Wylie SA, Forstmann BU, Burle B, Hasbroucq T, Ridderinkhof KR (2010) To head or to heed? Beyond the surface of selective action inhibition: a review. Front Hum Neurosci 4:222
Van Wijk B, Daffersthofer A, Roach N, Praamstra P (2009) A role of beta oscillatory synchrony in biasing response competition? Cereb Cortex 19:1294–1302
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Praamstra, P., Loing, A.F. & de Lange, F.P. Leakage of decision uncertainty into movement execution in Parkinson’s disease?. Exp Brain Res 232, 21–30 (2014). https://doi.org/10.1007/s00221-013-3715-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-013-3715-x