Abstract
This review highlights recent trends in the development of multiplexed bioanalyses using quantum dot bioconjugates and donor–acceptor interactions. In these methods, multiple optical signals are generated in response to biorecognition through modulation of the photoluminescence of populations of quantum dots with different emission colors. The donor–acceptor interactions that have been used include fluorescence resonance energy transfer, bioluminescence resonance energy transfer, charge transfer quenching, and quenching via proximal gold nanoparticles. Assays for the simultaneous detection of between two and eight target analytes have been developed, where spectral deconvolution is an important tool. Target analytes have included small molecules, nucleic acid sequences, and proteases. The unique optical properties of quantum dots offer several potential advantages in multiplexed detection, and a large degree of versatility, for example, one pot multiplexing at the ensemble level, where only wavelength discrimination is required to differentiate between detection channels. These methods are not being developed to compete with array-based technologies in terms of overall multiplexing capacity, but rather to enable new formats for multiplexed bioanalyses. In particular, quantum dot bioprobes based on donor–acceptor interactions are anticipated to provide future opportunities for multiplexed biosensing within living cells.

Figure
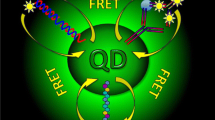
Multicolor optical bioanalyses using donor-acceptor interactions between quantum dots and gold nanoparticles, fluorescent dyes, or redox active complexes.





Similar content being viewed by others
Notes
QDs have also been used as electroactive labels for multiplexed anodic stripping voltammetry [5]; however, discussion of this is beyond the scope of this review.
Photoluminescence quenching by Au NPs has been primarily associated with dipole–surface interactions that have an inverse fourth power (r −4) distance dependence [16, 17], and also with changes in radiative rate induced by the Au NPs [18, 19]. These details are beyond the scope of this review, and the interested reader is referred to the cited references for more information.
References
Michalet X, Pinaud FF, Bentolila LA, Tsay JM, Doose S, Li JJ, Sundaresan G, Wu AM, Gambhir SS, Weiss S (2005) Science 307:538–544
Medintz IL, Uyeda HT, Goldman ER, Mattoussi H (2005) Nat Mater 4:435–446
Chan WCW, Maxwell DJ, Gao XH, Bailey RE, Han MY, Nie SM (2002) Curr Opin Biotechnol 13:40–46
Lee JA, Mardyani S, Hung A, Rhee A, Klostranec J, Mu Y, Li D, Chan WCW (2007) Adv Mater 19:3113–3118
Kim JH, Seo KS, Wang J (2006) IEEE Sens J 6:248–253
Wilson R, Cossins AR, Spiller DG (2006) Angew Chem Int Ed 45:6104–6117
Bentolila LA, Weiss A (2006) Cell Biochem Biophys 45:59–70
Tholouli E, Hoyland JA, Di Vizio D, O’Connell F, MacDermott SA, Twomey D, Levenson R, Liu Yin JA, Golub TR, Loda M, Byers R (2006) Biochem Biophys Res Commun 348:628–636
Chan PM, Yuen T, Ruf F, Gonzalez-Maeso J, Sealfon SC (2005) Nucleic Acids Res 33:e161
Nie S, Xing Y, Kim GJ, Simons JW (2007) Annu Rev Biomed Eng 9:257–288
Smith AM, Duan H, Mohs AM, Nie S (2008) Adv Drug Del Rev 60:1226–1240
Algar WR, Krull UJ (2008) Anal Bioanal Chem 391:1609–1618
Medintz IL, Mattoussi H (2009) Phys Chem Chem Phys 11:17–45
Algar WR, Tavares AJ, Krull UJ (2010) Anal Chim Acta (in press)
Xia ZY, Rao JH (2009) Curr Opin Biotechnol 20:37–44
Yun CS, Javier A, Jennings T, Fisher M, Hira S, Peterson S, Hopkins B, Reich NO, Strouse GF (2005) J Am Chem Soc 127:3115–3119
Li X, Qian J, Jiang L, He S (2009) Appl Phys Lett 94:063111
Dulkeith E, Ringler M, Klar TA, Feldmann J, Muñoz Javier A, Parak WJ (2005) Nano Lett 5:585–589
Schneider G, Decher G, Nerambourg N, Praho R, Werts MHV, Blanchard-Desce M (2006) Nano Lett 6:530–536
Pons T, Medintz IL, Sapsford KE, Higashiya S, Grimes AF, English DS, Mattoussi H (2007) Nano Lett 7:3157–3154
Pan B, Cui D, Ozkan CS, Ozkan M, Xu P, Huang T, Liu F, Chen H, Li Q, He R, Gao F (2008) J Phys Chem C 112:939–944
Medintz IL, Farrell D, Susumu K, Trammell SA, Deschamps JR, Brunel FM, Dawson PE, Mattoussi H (2009) Anal Chem 81:4831–4839
Sandros MG, Shete V, Benson DE (2006) Analyst 131:229–235
Swain MD, Octain J, Benson DE (2008) Bioconjug Chem 19:2520–2526
Bailey VJ, Easwaran H, Zhang Y, Griffiths E, Belinsky SA, Herman JG, Baylin SB, Carraway HE, Wang TH (2009) Genome Res 19:1455–1461
Algar WR, Krull UJ (2007) Anal Chim Acta 581:193–201
Algar WR, Krull UJ (2009) Anal Chem 81:4113–4120
Algar WR, Krull UJ (2010) Anal Chem 82:400–405
Algar WR, Krull UJ (2010) Langmuir 26:6041–6047
Algar WR, Krull UJ (2010) In: Alexandrou A, Cheon J, Mattoussi H, Rotello V (eds) Biological imaging and sensing using nanoparticle assemblies. symposium proceedings, vol 1241E. Materials Research Society, Warrendale, paper no 1241-XX09-04
Tang B, Cao L, Xu K, Zhuo L, Ge J, Li Q, Yu L (2008) Chem Eur J 14:3637–3644
Cady NC, Strickland AD, Batt CA (2007) Mol Cell Probes 21:116–124
Chang E, Miller JS, Sun JT, Yu WW, Colvin VL, Drezek R, West KL (2005) Biochem Biophys Res Commun 334:1317–1321
Liu J, Lee JH, Lu Y (2007) Anal Chem 79:4120–4125
Kim YP, Oh YH, Oh E, Ko S, Han MK, Kim HS (2008) Anal Chem 80:4634–4641
Clapp AR, Medintz IL, Fisher BR, Anderson GP, Mattoussi H (2005) J Am Chem Soc 127:1242–1250
So MK, Xu C, Loening AM, Gambhir SS, Rao J (2006) Nat Biotechnol 24:339–343
Xia Z, Xing Y, So MK, Koh AL, Sinclair R, Rao J (2008) Anal Chem 80:8649–8655
Allen PM, Bawendi M (2008) J Am Chem Soc 130:9240–9241
Allen PM, Liu W, Chauhan VP, Lee J, Ting AY, Fukumura D, Jain RK, Bawendi MG (2010) J Am Chem Soc 132:470–471
Jiang W, Singhal A, Zheng J, Wang C, Chan WCW (2006) Chem Mater 18:4845–4854
Medintz IL, Pons T, Trammell SA, Grimes AF, English DS, Blanco-Canosa JB, Dawson PE, Mattoussi H (2008) J Am Chem Soc 130:16745–16756
Pons T, Medintz IL, Sykora M, Mattoussi H (2006) Phys Rev B 73:245302
Xu H, Sha MY, Wong EY, Uphoff J, Xu Y, Treadway JA, Truong A, O’Brien E, Asquith S, Stubbins M, Spurr NK, Lai EH, Mahoney W (2003) Nucleic Acids Res 31:e43
Han M, Gao X, Su JZ, Nie S (2001) Nat Biotechnol 19:631–635
Fournier-Bidoz S, Jennings TL, Klostranec JM, Fung W, Rhee A, Li D, Chan WCW (2008) Angew Chem Int Ed 47:5577–5581
Eastman PS, Ruan W, Doctolero M, Nuttall R, de Feo G, Park JS, Chu JSF, Cooke P, Gray JW, Li S, Chen FF (2006) Nano Lett 6:1059–1064
Hohng S, Ha T (2005) Chem Phys Chem 6:956–960
Pons T, Medintz IL, Wang X, English DS, Mattoussi H (2006) J Am Chem Soc 128:15324–15331
Zhang CY, Yeh HC, Kuroki MT, Wang TH (2005) Nat Mater 4:826–831
Zhang CY, Johnson LW (2009) Anal Chem 81:3051–3055
Klostranec JM, Xiang Q, Farcas GA, Lee JA, Rhee A, Lafferty EI, Perrault SD, Kain KC, Chan WCW (2007) Nano Lett 7:2812–2818
Delehanty JB, Mattoussi H, Medintz IL (2009) Anal Bioanal Chem 393:1091–1105
Delehanty JB, Medintz IL, Pons T, Brunel FM, Dawson PE, Mattoussi H (2006) Bioconjug Chem 17:920–927
Horton KL, Stewart KM, Fonseca SB, Guo Q, Kelley SO (2008) Chem Biol 15:375–382
Acknowledgements
The authors are grateful to the Natural Sciences and Engineering Research Council of Canada (NSERC) for funding their research program. W.R.A. is also grateful to the University of Toronto and the Ontario Ministry of Education and Training for a graduate fellowship (OGSST).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in the special issue on Focus on Bioanalysis with Guest Editors Antje J. Baeumner, Günter Gauglitz, Frieder W. Scheller.
Rights and permissions
About this article
Cite this article
Algar, W.R., Krull, U.J. New opportunities in multiplexed optical bioanalyses using quantum dots and donor–acceptor interactions. Anal Bioanal Chem 398, 2439–2449 (2010). https://doi.org/10.1007/s00216-010-3837-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-010-3837-y