Abstract
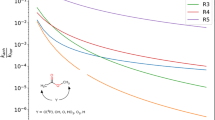
Although vinyl alcohol(CH2=CHOH)molecule was found to be an important intermediate in the combustion flames of hydrocarbon (Taatjes et al. in Science 308:1887, 2005), the removal mechanism of vinyl alcohol has not been established yet. The removal mechanism is critical to characterize the kinetics behavior of hydrocarbon in combustion chemistry and to develop the chemical models of hydrocarbon oxidation. In this work, the potential energy surface for the unimolecular decomposition of syn-CH2=CHOH reaction has been first studied by ab initio. The kinetics and product branching ratios for the decomposition reaction are evaluated by Variflex code in the temperature range of 500–3,000 K at 0.1, 1.0, and 100.0 atmosphere pressure. The results show that the formation of CH3 + CHO via the CH3CHO intermediate is dominant in the decomposition reaction and its branching ratios at 0.1, 1.0, and 100.0 atm are more than 99.90, 99.30, and 89.20%, respectively, through the whole temperature range investigated.



Similar content being viewed by others
References
Erlenmeyer E (1880) Chem Ber 13:305
Saito S (1976) Chem Phys Lett 42:399
Toullec J, El-Alaoui MJ (1986) Org Chem 51:4054
Capon B, Guo BZ, Kwok FC, Siddhanta AK, Zucco C (1988) Acc Chem Res 21:135
Hart H (1979) Chem Rev 79:515
Senosiain JP, Klippenstein SJ, Miller JA (2006) J Phys Chem A 110:6960
Chandra AK, Thérèse ZH (2003) J Org Chem 68:3618
Smith BJ, Nguyen MT, Bouma WJ, Radom L (1991) J Am Chem Soc 113:6452
Cool TA, Nakajima K, Mostefaoui TA, Qi F, McIlroy A, Westmoreland PR, Law ME, Poisson L, Peterka DS, Ahmed M (2003) J Chem Phys 119:8356
Taatjes CA, Hansen N, McIlroy A, Miller JA, Senosiain JP, Klippenstein SJ, Qi F, Sheng L, Zhang Y, Cool TA, Wang J, Westmoreland PR, Law ME, Kasper T, Kohse-Höinghaus K (2005) Science 308:1887
Yamada T, Bozzelli JW, Lay T (1999) J Phys Chem A 103:7646
Hippler H, Viskolcz B (2000) Phys Chem Chem Phys 2:3591
Taatjes CA, Hansen N, Miller JA (2006) J Phys Chem A 110:3254
Rodler M, Bauder A (1984) J Am Chem Soc 106:4025
Tureček F, Cramer CJ (1995) J Am Chem Soc 117:12243
Silva GD, Kim CH, Bozzelli JW (2006) J Phys Chem A 110:7925
Becke AD (1992) J Chem Phys 96:2155
Becke AD (1992) J Chem Phys 97:9173
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785
Hehre W, Radom L, Schleyer PVR, Pople JA (1986) Ab initio molecular orbital theory. Wiley, New York
Andersson MP, Uvdal P (2005) J Phys Chem A 109:2937
Gonzalez C, Schlegel HB (1989) J Phys Chem 90:2154
Raghavachari K, Trucks GW, Pople JA, Head-Gordon G (1989) Chem Phys Lett 157:479
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA Jr, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2003) Gaussian 03 (revision B.05)
Klippenstein SJ, Wagner AF, Dunbar RC, Wardlaw DM, Robertson SH (1999) VariFlex version 1.0. Argonne National Laboratory, Argonne
Beyer T, Swinehart DF (1973) Commun Assoc Comput Mach 16:379
Stein SE, Rabinovitch BS (1973) J Phys Chem 58:2438
Gilbert RG, Smith SC (1990) Theory of unimolecular and recombination reactions. Blackwell, Carlton
Holbrook KA, Pilling MJ, Robertson SH (1996) Unimolecular reactions. Wiley, New York
Miller JA, Klippenstein SJ (2003) J Phys Chem A 107:2680
Miller JA, Klippenstein SJ, Raffy C (2002) J Phys Chem A 106:4904
Miller JA, Klippenstein SJ, Robertson SH (2000) J Phys Chem A 104:7525
Fernandez-Ramos A, Miller J, Klippenstein S, Truhlar D (2006) Chem Rev 106:4518
Miller J, Klippenstein S (2006) J Phys Chem A 110:10528
Zhou CW, Li ZR, Liu CX, Li XY (2008) J Chem Phys 129:234301
Park J, Zhu RS, Lin MC (2002) J Chem Phys 117:3224
Matti GY, Osman OI, Upham JE, Suffolk RJ, Kroto HW (1989) J Electron Spectrosc Relat Phenom 49:195
Rodler M (1985) J Mol Spectrosc 114:23
Bouma WJ, Adom L (1978) J Mol Struct 43:267
Yasunaga K, Kubo S, Hoshikawa H, Kamesawa T, Hidaka Y (2008) Int J Chem Kinet 40:73
Gupte KS, Kiefer JH, Tranter RS, Klippenstein SJ, Harding LB (2007) Proc Combust Inst 31:167
Bentz T, Striebel F, Olzmann M (2008) J Phys Chem A 112:6120
Horowltz A, Calvert JG (1982) J Phys Chem 86:3105
Joshi A, You XQ, Barckholtz TA, Wang H (2005) J Phys Chem A 109:8016
Yadav JS, Goddard JD (1986) J Chem Phys 84:2682
Nilsson EJK, Bache-Andreassen L, Johnson MS, Nielsen CJ (2009) J Phys Chem A 113:3498
Harding LB, Georgievskii Y, Klippenstein SJ (2010) J Phys Chem A 114:765
Tardy DC, Rabinovitch BS (1977) Chem Rev 77:369
Quack M, Troe J (1977) In: Asmore PG, Donovan RJ (eds) Gas kinetics and energy transfer, vol 2. The Chemical Society, London
Poling BE, Prausnitz JM, O’Connell JP (2001) The properties of gases and liquids, 5th edn. McGraw-Hill, New York
Mourits FM, Rummens HA (1977) Can J Chem 55:3007
Zhu RS, Lin MC (2004) Chem Phys Chem 5:1864
Eckart C (1930) Phys Rev 35:1303
Acknowledgments
This work has been supported by the National Natural Science Foundation of China (Nos. 91016002, 20973118) and by the Natural Science Foundation of the Education Department of Sichuan Province in China (No.09ZA144).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shao, JX., Gong, CM., Li, XY. et al. Unimolecular decomposition mechanism of vinyl alcohol by computational study. Theor Chem Acc 128, 341–348 (2011). https://doi.org/10.1007/s00214-010-0860-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00214-010-0860-1