Abstract
Rationale and objective
We sought to examine the impact of differing cocaine administration schedules and dosing on the magnitude of cocaine conditioned place preference (CPP), extinction, and stress- and cocaine-induced reinstatement of CPP.
Methods
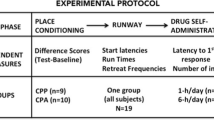
First, in C57Bl/6J mice, we investigated whether total cocaine administration or pattern of drug exposure could influence the magnitude of cocaine CPP by conditioning mice with a fixed-low dose (FL; 7.5 mg/kg; total of 30 mg/kg), a fixed-high dose (FH; 16 mg/kg; total of 64 mg/kg), or an ascending dosing schedule (Asc; 2, 4, 8, and 16 mg/kg; total of 30 mg/kg). Next, we investigated if cocaine or saline is more effective at extinguishing preference by reconditioning mice with either a descending dosing schedule (Desc; 8, 4, 2, and 1 mg/kg) or saline. Finally, we examined if prior conditioning and reconditioning history alters stress (~2–3-min forced swim test) or cocaine-induced (3.5 mg/kg) reinstatement.
Results
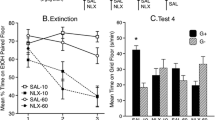
We replicated and extended findings by Itzhak and Anderson (Addict. Biol. 17(4): 706–16, 2011) demonstrating that Asc conditioning produces a greater CPP than either the FL or FH conditioning schedules. The magnitude of extinction expressed was similar in the Desc reconditioned and saline groups. Moreover, only the saline, and not the Desc reconditioned mice, showed stress and cocaine-induced reinstatement of CPP.
Conclusions
Our results suggest that the schedule of cocaine administration during conditioning and reconditioning can have a significant influence on the magnitude of CPP and extinction of preference and the ability of cocaine or a stressor to reinstate CPP.





Similar content being viewed by others
References
Ahmed SH, Kenny PJ, Koob GF, Markou A (2002) Neurobiological evidence for hedonic allostasis associated with escalating cocaine use. Nat Neurosci 5:625–626
Angarita GA, Pittman B, Gueorguieva R, Kalayasiri R, Lynch WJ, Sughondhabirom A et al (2010) Regulation of cocaine self-administration in humans: lack of evidence for loading and maintenance phases. Pharmacol Biochem Behav 95:51–55
Davis AR, Shields AD, Brigman JL, Norcross M, McElligott ZA, Holmes A et al (2008) Yohimbine impairs extinction of cocaine-conditioned place preference in an alpha2-adrenergic receptor independent process. Learn Mem 15:667–676
Ferrario CR, Gorny G, Crombag HS, Li Y, Kolb B, Robinson TE (2005) Neural and behavioral plasticity associated with the transition from controlled to escalated cocaine use. Biol Psychiatry 58:751–759
Gawin FH (1991) Cocaine addiction: psychology and neurophysiology. Science 251:1580–1586
Gawin FH, Ellinwood EH Jr (1989) Cocaine dependence. Annu Rev Med 40:149–161
Grimm JW, Hope BT, Wise RA, Shaham Y (2001) Neuroadaptation. Incubation of cocaine craving after withdrawal. Nature 412(6843):141–142
Itzhak Y, Anderson KL (2011) Changes in the magnitude of drug-unconditioned stimulus during conditioning modulate cocaine-induced place preference in mice. Addict Biol 17(4):706–716
Koob G, Kreek MJ (2007) Stress, dysregulation of drug reward pathways, and the transition to drug dependence. Am J Psychiatry 164:1149–1159
Kreibich AS, Blendy JA (2004) cAMP response element binding protein is required for stress but not cocaine-induced reinstatement. J Neurosci 24(30):6686–6692
Mantsch JR, Weyer A, Vranjkovic O, Beyer CE, Baker DA, Caretta H (2010) Involvement of noradrenergic neurotransmission in the stress- but not cocaine-induced reinstatement of extinguished cocaine-induced conditioned place preference in mice: role for beta-2 adrenergic receptors. Neuropsychopharmacology 35:2165–2178
Marks KR, Kearns DN, Christensen CJ, Silberberg A, Weiss SJ (2010) Learning that a cocaine reward is smaller than expected: a test of Redish's computational model of addiction. Behav Brain Res 212:204–207
Miner LL (1997) Cocaine reward and locomotor activity in C57BL/6J and 129/SvJ inbred mice and their F1 cross. Pharmacol Biochem Behav 58:25–30
Morgan D, Liu Y, Oleson EB, Roberts DC (2009) Cocaine self-administration on a hold-down schedule of reinforcement in rats. Psychopharmacology 201:601–609
Panlilio LV, Thorndike EB, Schindler CW (2007) Blocking of conditioning to a cocaine-paired stimulus: testing the hypothesis that cocaine perpetually produces a signal of larger-than-expected reward. Pharmacol Biochem Behav 86:774–777
Porrino LJ, Lyons D, Miller MD, Smith HR, Friedman DP, Daunais JB et al (2002) Metabolic mapping of the effects of cocaine during the initial phases of self-administration in the nonhuman primate. J Neurosci 22:7687–7694
Rush CR, Stoops WW, Hays LR (2009) Cocaine effects during d-amphetamine maintenance: a human laboratory analysis of safety, tolerability and efficacy. Drug Alcohol Depend 99:261–271
Schindler AG, Li S, Chavkin C (2010) Behavioral stress may increase the rewarding valence of cocaine-associated cues through a dynorphin/kappa-opioid receptor-mediated mechanism without affecting associative learning or memory retrieval mechanisms. Neuropsychopharmacology 35:1932–1942
Smirnov MS, Kiyatkin EA (2010) Cocaine action on peripheral, non-monoamine neural substrates as a trigger of electroencephalographic desynchronization and electromyographic activation following i.v. administration in freely moving rats. Neuroscience 165:500–514
Tzschentke TM (2007) Measuring reward with the conditioned place preference (CPP) paradigm: update of the last decade. Addict Biol 12:227–462
Vanderschuren LJ, Everitt BJ (2004) Drug seeking becomes compulsive after prolonged cocaine self-administration. Science 305:1017–1019
Zhou Y, Litvin Y, Piras AP, Pfaff DW, Kreek MJ (2011) Persistent increase in hypothalamic arginine vasopressin gene expression during protracted withdrawal from chronic escalating-dose cocaine in rodents. Neuropsychopharmacology 36:2062–2075
Zimmer BA, Dobrin CV, Roberts DC (2011) Brain-cocaine concentrations determine the dose self-administered by rats on a novel behaviorally dependent dosing schedule. Neuropsychopharmacology 36:2741–2749
Acknowledgments
The behavioral data presented in this manuscript were generated at the Vanderbilt Murine Neurobehavioral Laboratory (http://kc.vanderbilt.edu/mnlcore/). We would like to thank Dr. John Allison for his generous assistance and expertise. This work was supported by National Institutes of Health grants NS07491 and MH065215-08 (KLC) and AA019455 AND DA019112 (DGW).
Conflict of interest
All authors report no biomedical financial interests or potential conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Kelly L. Conrad and Katherine M. Louderback contributed equally to this work.
Rights and permissions
About this article
Cite this article
Conrad, K.L., Louderback, K.M., Milano, E.J. et al. Assessment of the impact of pattern of cocaine dosing schedule during conditioning and reconditioning on magnitude of cocaine CPP, extinction, and reinstatement. Psychopharmacology 227, 109–116 (2013). https://doi.org/10.1007/s00213-012-2944-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-012-2944-1