Abstract
Rationale
It has long been hypothesized that human as well as animal cocaine users titrate their intake to maintain a specific level of cocaine reward. This hypothesis predicts that the dose–injection function of each subject individually should be a decreasing function, with no initial, gradual ascending limb.
Objectives
The present study was designed to test this specific prediction.
Methods
Rats were trained to self-administer cocaine under a continuous reinforcement schedule. After stabilization of cocaine self-administration, all rats were tested with a wide range of i.v. cocaine doses (0.0078–1 mg). To accurately measure the threshold dose of each individual, the pharmacological resolution was set at 0.0078 mg at the four lowest doses.
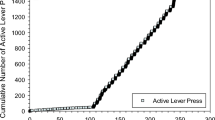
Results
As predicted, individual dose–behavior curves are discontinuous at a threshold dose, with a descending limb but no gradual, ascending limb. Below the threshold, there is no evidence for cocaine self-administration; at and above the threshold, the rate of injections spikes to its maximum and then decreases lawfully with the dose, a decrease that reflects cocaine titration. In all individuals, this critical transition occurred over a dose interval of less than 0.008 mg.
Conclusions
This study suggests that the cumulative effects of cocaine maintained during self-administration are all-or-nothing—a conclusion that confirms the regulation hypothesis of cocaine reward. The neurobehavioral consequences of this specific level of cocaine reward remain to be elucidated.





Similar content being viewed by others
References
Ahmed SH, Koob GF (1997) Cocaine- but not food-seeking behavior is reinstated by stress after extinction. Psychopharmacology 132:289–295
Ahmed SH, Koob GF (1998) Transition from moderate to excessive drug intake: change in hedonic set point. Science 282:298–300
Ahmed SH, Koob GF (1999) Long-lasting increase in the set point for cocaine self-administration after escalation in rats. Psychopharmacology 146:303–312
Ahmed SH, Koob GF (2005) Transition to drug addiction: a negative reinforcement model based on an allostatic decrease in reward function. Psychopharmacology 180:473–490
Ahmed SH, Kenny PJ, Koob GF, Markou A (2002) Neurobiological evidence for hedonic allostasis associated with escalating cocaine use. Nat Neurosci 5:625–626
Arnold JM, Roberts DC (1997) A critique of fixed and progressive ratio schedules used to examine the neural substrates of drug reinforcement. Pharmacol Biochem Behav 57:441–447
Bickel WK, De Grandpre RJ, Higgins ST, Hughes JR (1990) Behavioral economics of drug self-administration: I. Functional equivalence of response requirement and drug dose. Life Sci 47:1501–1510
Caine SB, Koob GF (1995) Pretreatment with the dopamine agonist 7-OH-DPAT shifts the cocaine self-administration dose-effect function to the left under different schedules in the rat. Behav Pharmacol 6:333–347
Carroll ME, Lac ST (1997) Acquisition of i.v. amphetamine and cocaine self-administration in rats as a function of dose. Psychopharmacology 129:206–214
Downs DA, Woods JH (1974) Codeine- and cocaine-reinforced responding in rhesus monkeys: effects of dose on response rates under a fixed-ratio schedule. J Pharmacol Exp Ther 191:179–188
Flory GS, Woods JH (2003) The ascending limb of the cocaine dose–response curve for reinforcing effect in rhesus monkeys. Psychopharmacology 166:91–94
Gallistel CR, Fairhurst S, Balsam P (2004) The learning curve: implications of a quantitative analysis. Proc Natl Acad Sci USA 101:13124–13131
Gerber GJ, Wise RA (1989) Pharmacological regulation of intravenous cocaine and heroin self-administration in rats: a variable dose paradigm. Pharmacol Biochem Behav 32:527–531
Hodos W (1961) Progressive ratio as a measure of reward strength. Science 134:943–944
Koob GF, Le Moal M (1997) Drug abuse: hedonic homeostatic dysregulation. Science 278:52–58
Lynch WJ, Sughondhabirom A, Pittman B, Gueorguieva R, Kalayasiri R, Joshua D, Morgan P, Coric V, Malison RT (2006) A paradigm to investigate the regulation of cocaine self-administration in human cocaine users: a randomized trial. Psychopharmacology 185:306–314
Mantsch JR, Yuferov V, Mathieu-Kia AM, Ho A, Kreek MJ (2004) Effects of extended access to high versus low cocaine doses on self-administration, cocaine-induced reinstatement and brain mRNA levels in rats. Psychopharmacology 175:26–36
Mello NK, Negus SS (1996) Preclinical evaluation of pharmacotherapies for treatment of cocaine and opioid abuse using drug self-administration procedures. Neuropsychopharmacology 14:375–424
Panlilio LV, Katz JL, Pickens RW, Schindler CW (2003) Variability of drug self-administration in rats. Psychopharmacology 167:9–19
Parsons LH, Koob GF, Weiss F (1995) Serotonin dysfunction in the nucleus accumbens of rats during withdrawal after unlimited access to intravenous cocaine. J Pharmacol Exp Ther 274:1182–1191
Peoples LL, Uzwiak AJ, Guyette FX, West MO (1998) Tonic inhibition of single nucleus accumbens neurons in the rat: a predominant but not exclusive firing pattern induced by cocaine self-administration sessions. Neuroscience 86:13–22
Pettit HO, Justice JB Jr (1989) Dopamine in the nucleus accumbens during cocaine self–administration as studied by in vivo microdialysis. Pharmacol Biochem Behav 34:899–904
Ranaldi R, Pocock D, Zereik R, Wise RA (1999) Dopamine fluctuations in the nucleus accumbens during maintenance, extinction, and reinstatement of intravenous d-amphetamine self-administration. J Neurosci 19:4102–4109
Rothman RB, Baumann MH, Dersch CM, Romero DV, Rice KC, Carroll FI, Partilla JS (2001) Amphetamine-type central nervous system stimulants release norepinephrine more potently than they release dopamine and serotonin. Synapse 39:32–41
Sizemore GM, Martin TJ (2000) Toward a mathematical description of dose-effect functions for self-administered drugs in laboratory animal models. Psychopharmacology 153:57–66
Tornatzky W, Miczek KA (2000) Cocaine self-administration “binges”: transition from behavioral and autonomic regulation toward homeostatic dysregulation in rats. Psychopharmacology 148:289–298
Tsibulsky VL, Norman AB (1999) Satiety threshold: a quantitative model of maintained cocaine self-administration. Brain Res 839:85–93
Weeks JR (1962) Experimental morphine addiction: method for automatic intravenous injections in unrestrained rats. Science 138:143–144
Wise RA, Newton P, Leeb K, Burnette B, Pocock D, Justice JB Jr (1995) Fluctuations in nucleus accumbens dopamine concentration during intravenous cocaine self-administration in rats. Psychopharmacology 120:10–20
Yokel RA, Pickens R (1973) Self-administration of optical isomers of amphetamine and methylamphetamine by rats. J Pharmacol Exp Ther 187:27–33
Yokel RA, Pickens R (1974) Drug level of d- and l-amphetamine during intravenous self-administration. Psychopharmacologia 34:255–264
Acknowledgments
This work was supported by grants from Université Victor-Segalen Bordeaux 2, CNRS, MILDT and Région Aquitaine. We thank Lydie Goutière for animal care, Pierre Gonzalez for technical assistance, Marie-Hélène Bruyères for administrative assistance, Christian Darrack (Imetronic, Pessac) for his help with data extraction, Dr Luis Stinus for general support and excellent laboratory management and, finally, two anonymous reviewers for helpful comments. We also thank the laboratories Sarget, Sanofi–Synthelabo and Bristol–Myers–Squibb for their generous gift of betadine, heparin, and totapen, respectively.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zittel-Lazarini, A., Cador, M. & Ahmed, S.H. A critical transition in cocaine self-administration: behavioral and neurobiological implications. Psychopharmacology 192, 337–346 (2007). https://doi.org/10.1007/s00213-007-0724-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-007-0724-0