Abstract
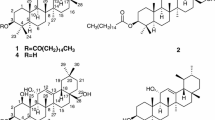
The EtOAc extract of Youngia koidzumiana significantly inhibited the diacylglycerol acyltransferase (DGAT) from rat liver microsomes. Bioactivity-guided fractionation led to the isolation of nine compounds, the structures of which were established using physicochemical and spectral data. Of the isolated compounds, oleanolic acid (2), methyl ursolate (7) and corosolic aicd (8) inhibited DGAT, with IC50 values of 31.7, 26.4, and 44.3 μM, respectively. However, sesquiterpenoids showed only weak inhibitory effects toward DGAT.
Similar content being viewed by others
References
Adegawa, S. and Miyase, T., Sesquiterpene glycosides fromCrepidiastrum keiskeanum Nakai.Chem. Pharm. Bull., 33, 4906–4911 (1985).
Cases, S., Smith, S., Zheng, Y., Myers, H., Lear, S. R., and Sande, E., Identification of gene encoding an acyl CoA: diacylglycerol acyltransferase, a key enzyme in triacyglycerol synthesis.Proc. Natl. Acad. Sci. U.S.A., 95, 13018–13023 (1998).
Coleman, R. A., Diacylglycerol acyltransferase and monoacyl-glycerol acyltransferase from liver and intestine.Methods Enzymol., 209, 98–103 (1992).
Dat, N. T., Cai, X. F., Bae, K. H., and Kim, Y. H., Terpenoid constituents fromYoungia koidzumiana.Nat. Pro. Sci., 8, 55–57 (2002).
Gray, G. A. and Tartaglia, L. A., Medicinal strategies in the treatment of obesity.Curr. Opin. Lipidol., 11, 229–234 (2000).
Kang, S. S. and Son, K. H.,Structure determination of natural products. Seoul National University Publisher, Seoul, (2000).
Ko, J. S., Rho, M-C, Chung, M. Y., Song, H. Y., Kang J. S., Lee, H. S., and Kim, Y. K., Quinolone alkaloids, diacylglycerol acyltransferase inhibitors from the fruits ofEvodia rutaecarpa.Planta Med., 68, 1131–1133 (2002).
Ko, J. S., Ryu, S. Y., Kim, Y. S., Chung, M. Y., Kang, J. S., Rho, M-C, Lee, H. S., and Kim, Y. K., Inhibitory activity of diacylglycerol acyltransferase by tanshinones from the root ofSalvia miltiorrhiza.Arch. Pharm. Res., 25, 446–448 (2002).
Kozima, H. and Ogura, H., Configurational studies on hydroxy groups at C-2, 3 and 23 or 24 of oleanene and ursane-type triterpenes by NMR spectroscopy.Phytochem., 28, 1703–1710 (1989).
Kuang, H. X. and Kasai, R., Chemical constituents of Pericarps ofRosa davurica Pall., a traditional Chinese medicine.Chem. Pharm. Bull., 37, 2232–2233 (1989).
Lehner, R. and Kuksis, A., Biosynthesis of triacylglycerols.Prog, Lipid Res., 35, 169–201 (1996).
Piozzi, F. and Paternostro, M., Triterpenes fromAmaracus dictamnus.Phytochem., 25, 539–541 (1986).
Smith, S. J., Cases, S., Jensen, D. R., Chen, H. C., Sande, E., and Tow, B., Obesity resistance and multiple mechanisms of triglyceride synthesis in mice lacking Dgat.Nat. Genet., 25, 87–90 (2000).
Tabata, N., Ito, M., Tomoda, H., and Omura, S., Xanthohumols, diacylglycerol acyltransferase inhibitors, fromHumulus lupulus.Phytochem., 46, 683–687 (1997).
Tomoda, H., Ito, M., Tabata, N., Masuma, R., Yamaguchi, Y., and Omura, S., Amidepsines, inhibitors of diacylglycerol acyltransferase produced byHumicola sp. FO-2942.J. Antibiot., 48, 937–941 (1995).
Tomoda, H., Ohyama, Y., Abe, T., Tabata, N., Namikoshi, M., Yamaguchi, Y., Masuma, R., and Omura, S., Roselipines, inhibitors of diacylglycerol acyltransferase, produced byGliocalidum roseum KF-1040.J. Antibiot., 52, 689–694 (1999).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dat, N.T., Cai, X.F., Rho, MC. et al. The inhibition of diacylglycerol acyltransferase by terpenoids fromYoungia koidzumiana . Arch Pharm Res 28, 164–168 (2005). https://doi.org/10.1007/BF02977708
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02977708