Summary
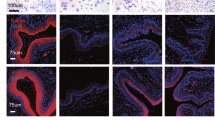
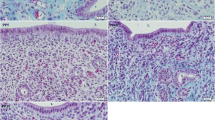
Mouse vaginal epithelial cells were isolated from intact 21-day-old BALB/cCrgl mice and cultured in a serum-free medium (SF20: basal medium supplemented with insulin, epidermal growth factor, transferrin, and bovine serum albumin—fraction V) to examine the proliferation, differentiation, and specificity of estrogen-induced growth retardation in vitro. Histologic and ultrastructural studies showed that vaginal epithelial cells undergo differentiative changes in vitro in the absence of estrogen, and that these changes are similar to those induced in vivo by estrogen. Addition of 17β-estradiol inhibited cellular proliferation in a dose-dependent manner. Whereas other estrane derivatives (17α-estradiol and estriol) also significantly retarded cellular proliferation, cholesterol, testosterone, and progesterone had no effect. Keoxifene, an antiestrogen, significantly reversed estrogen-induced growth inhibition, resulting in proliferation of estrogen-treated cells equivalent to that of the untreated control. The results suggest that both proliferation and differentiation of prepubertal mouse vaginal epithelial cells in vitro are estrogen-independent, and that the growth inhibition is a specific estrogen-induced response.
Similar content being viewed by others
References
Adler, A. J.; Nelson, J. F. Aging and chronic exposure impair estradiol-induced cornification but not proliferation of vaginal epithelium in C57BL/6J mice. Biol. Reprod. 38:175–182; 1988.
Bern, H. A. Is estrogen a cellular signal for female genital tract epithelium? In: Lavia, L. A., ed. Cellular signals controlling uterine function. New York: Plenum. In press; 1991.
Bindal, R. D.; Carlson, K. E.; Katzenellenbogen, B. S., et al. Lipophilic impurities, not phenolsulfonphthalein, account for the estrogenic activity in commercial preparations of phenol red. J. Steroid Biochem. 31:287–293; 1988.
Black, L. J.; Jones, C. D.; Falcone, J. F. Antagonism of estrogen action with a new benzothiophene derived antiestrogen. Life Sci. 32:1031–1036; 1982.
Buckley, A.; Davidson, J. M.; Kamerath, C. D., et al. Sustained release of epidermal growth factor accelerates wound repair. Proc. Natl. Acad. Sci. USA 82:7340–7344; 1985.
Conti, C. J.; Tasat, D. R. Regulation of cultured rat vaginal epithelial cells by 17β-estradiol and progesterone. J. Steroid Biochem. 24:747–751; 1986.
Cooke, P. S.; Uchima, F.-D. A.; Fujii, D. K., et al. Restoration of normal morphology and estrogen responsiveness in cultured vaginal and uterine epithelia transplanted with stroma. Proc. Natl. Acad. Sci. USA 83:2109–2113; 1986.
Cunha, G. R.; Fujii, H. Stromal-parenchymal interactions in normal and abnormal development of the genital tract. In: Herbst, A. L.; Bern, H. A., eds. Developmental effects of diethylstilbestrol (DES) in pregnancy. New York: Thieme-Stratton; 1981:179–193.
Eiger, S.; Mills, K. T.; Bern, H. A. Steroid binding alterations in tissue compartments of the vagina of control and neonatally diethylstilbestrol-treated adult mice. J. Steroid Biochem. 35:617–621; 1990.
Galand, P.; Rodesch, F.; Leroy, F., et al. Radioautographic evaluation of the estrogen-dependent proliferative pool in the stem cell compartment of the mouse uterine and vaginal epithelia. Exp. Cell Res. 48:595–604; 1967.
Galand, P.; Leroy, F.; Chretien, J. Effect of oestradiol on cell proliferation and histological changes in the uterus and vagina of mice. J. Endocrinol. 49:243–252; 1971.
Gilchrest, B. A.; Karassik, R. L.; Wilkins, L. M., et al. Autocrine and paracrine growth stimulation of cells derived from human skin. J. Cell. Physiol. 117:235–240; 1983.
Hinegardner, R. T. An improved fluorometric assay for DNA. Anal. Biochem. 39:197–201; 1971.
Iguchi, T.; Uchima, F.-D. A.; Ostrander, P. L., et al. Growth of normal mouse vaginal epithelial cells in and on collagen gels. Proc. Natl. Acad. Sci. USA 80:3743–3747; 1983.
Iguchi, T.; Uchima, F.-D. A.; Bern, H. A. Growth of mouse vaginal epithelial cells in culture: effect of sera and supplemented serum-free media. In Vitro Cell. Dev. Biol. 23:535–539; 1987.
Ikeda, T.; Sirbasku, D. A. Purification and properties of a mammary-uterine-pituitary tumor cell growth factor from pregnant sheep uterus. J. Biol. Chem. 259:4049–4061; 1984.
Kirkland, J. L.; Gardner, R. M.; Ireland, J. S., et al. The effect of hypophysectomy on the uterine response to estradiol. Endocrinology 101:403–410; 1977.
Knabbe, C.; Lippman, M. E.; Wakefield, L. M., et al. Evidence that transforming growth factor-β is a hormonally regulated negative growth factor in human breast cancer cells. Cell 48:417–428; 1987.
Kronenberg, M. S.; Clark, J. H. Changes in keratin expression during the estrogen-mediated differentiation of rat vaginal epithelium. Endocrinology 117:1480–1489; 1985.
Licht, P.; McCreery, B. R.; Barnes, R., et al. Seasonal and stress related changes in plasma gonadotropins, sex steroids and corticosterone in the bullfrog,Rana catesbeiana. Gen. Comp. Endocrinol. 50:124–145; 1983.
Lin, T.-H.; Kirkland, J. L.; Mukku, V. R., et al. Regulation of deoxyribonucleic acid polymerase activity in uterine luminal epithelium after multiple doses of estrogen. Endocrinology 122:1403–1409; 1988.
MacLusky, N. J.; Naftolon, F.; Krey, L. C., et al. The catechol estrogens. J. Steroid Biochem. 15:111–124; 1981.
Martin, L.; Finn, C. A.; Trinder, G. Hypertrophy and hyperplasia in the mouse uterus after oestrogen treatment: an autoradiographic study. J. Endocrinol. 56:133–144; 1973.
Seegers, J. C.; Aveling, M.-L.; van Aswegen, C. H., et al. The cytotoxic effects of estradiol-17β, catecholestradiols and methoxyestradiols on dividing MCF-7 and HeLa cells. J. Steroid Biochem. 32:797–809; 1989.
Sirbasku, D. A. Estrogen induction of growth factors specific for hormone-responsive mammary, pituitary, and kidney tumor cells. Proc. Natl. Acad. Sci. USA 75:3786–3790; 1978.
Snell, G. D. Reproduction. In: Snell, G. D., ed. Biology of the laboratory mouse. New York: Dover Publication; 1956:55–88.
Sonnenschein, C.; Pierce, G. B. Symposium on cell multiplication and cancer: positive or negative control. Cancer Res. 41:4742–4743; 1981.
Sonnenschein, C.; Olea, N.; Pasanen, M. E., et al. Negative controls of cell proliferation: human prostate cancer cells and androgens. Cancer Res. 49:3474–3481; 1989.
Soto, A. M.; Sonnenschein, C. The role of estrogens on the proliferation of human breast tumor cells (MCF-7). J. Steroid Biochem. 23:87–94; 1985.
Soto, A. M.; Sonnenschein, C. Cell proliferation of estrogen sensitive cells: the case for negative control. Endocrin. Rev. 8:44–52; 1987.
Soto, A. M.; Sonnenschein, C. Mechanism of estrogen action on cellular proliferation: evidence for indirect and negative control on cloned breast tumor cells. Biochem. Biophys. Res. Comm. 122:1097–1103; 1984.
Taguchi, O.; Bigsby, R. M.; Cunha, G. R. Estrogen responsiveness and the estrogen receptor during development of the murine female reproductive tract. Dev. Growth & Differ. 30:301–313; 1988.
Uchima, F.-D. A.; Edery, M.; Mills, K. T., et al. Estrogen and progestin receptors in mouse vaginal epithelium and fibromuscular wall. Biochim. Biophys. Acta 841:135–138; 1985.
Uchima, F.-D. A.; Edery, M.; Iguchi, T., et al. Growth of mouse vaginal epithelial cells in culture: functional integrity of the estrogen receptor system and failure of estrogen to induce proliferation. Cancer Lett. 17:115–123; 1987.
Vijayasaradhi, S.; Khar, A.; Gupta, P. D. Effect of steroid hormones on vaginal epithelial cells: an in vitro model for steroid hormone action. J. Biosci. 12:257–265; 1987.
Wood, E. J.; Raxworthy, M. J.; Holland, D. B. Retinoids and the epidermis. Biochem. Soc. Trans. 16:668–671; 1988.
Yang, J.; Flynn, D.; Larson, L., et al. Growth in primary culture of mouse submandibular epithelial cells embedded in collagen gels. In Vitro 18:435–442; 1982.
Author information
Authors and Affiliations
Additional information
This work was supported by grants CA-05388 and CA-09041 from the National Institutes of Health, Bethesda, MD.
Rights and permissions
About this article
Cite this article
Tsai, PS., Uchima, FD.A., Hamamoto, S.T. et al. Proliferation and differentiation of prepubertal mouse vaginal epithelial cells in vitro and the specificity of estrogen-induced growth retardation. In Vitro Cell Dev Biol - Animal 27, 461–468 (1991). https://doi.org/10.1007/BF02631145
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02631145