Summary
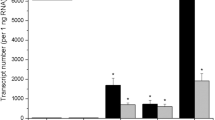
Nodal cuttings from micropropagated potato plantlets give rise to microtubers when placed on Murashige and Skoog medium containing 6% sucrose and 2.5 mg/liter kinetin and incubated in the dark at 19°C. Microtubers produced from the cultivar Superior were shown to contain the same characteristic group of proteins as field-grown tubers. As with field-grown tubers, the 40 000-dalton major tuber glycoprotein, patatin, accumulated to high levels in microtubers, reaching 3.7±0.2 mg/g fresh weight after 90 d. Also in agreement with field-grown plants, stems and leaves of micropropagated plantlets did not contain detectable levels of patatin, but small amounts of an electrophoretically distinct form accumulated transiently in roots. Patatin mRNA is readily detectable in developing microtubers 15 d after transfer of the cuttings to inductive medium. Patatin mRNA was also present in roots, but as with field-grown plants, was 50- to 100-fold less abundant and could be distinguished from that in tubers by primer extension. Microtuber development and patatin accumulation were inhibited by gibberellic acid.
Similar content being viewed by others
References
Barker, W. G. A method for the in vitro culturing of potato tubers. Science 118:383–385; 1953.
Focus. Formaldehyde gel conditions. Bethesda Research Laboratories 8(2): 14; 1986.
Forsline, P. L.; Langille, A. R. An assessment of the modifying effect of kinetin on in vitro tuberization of induced and noninduced tissues ofSolanum tubersosum. Can. J. Bot. 54:2513–2516; 1976.
Ghosh, P. K.; Reddy, V. B.; Swinscoe, J., et al. Heterogeneity and 5′-terminal structures of the late RNAs of simian virus 40. J. Mol. Biol. 126:813–846; 1978.
Hannapel, D. J.; Miller, J. C.; Park, W. D. Regulation of potato tuber protein accumulation by gibberellic acid. Plant Physiol. 78:700–703; 1985.
Hussey, G.; Stacey, N. S. Factors affecting the formation of in vitro tubers of potato (Solanum tuberosum L.). Ann. Bot. 53:565–578; 1984.
Jones, J. D. G.; Dunsmuir, P.; Bedbrook, J. High level expression of introduced chimaeric genes in regenerated transformed plants. EMBO J. 4:2411–2418; 1985.
Maniatis, T.; Fritsch, E. F.; Sambrook, J. Molecular cloning. A laboratory manual. New York: Cold Spring Harbor Laboratory Press; 1982: 187–209.
Mauk, C. S.; Langille, A. R. Physiology of tuberization inSolanum tuberosum L. Plant Physiol. 62:438–442; 1978.
Menzel, C. M. Tuberization in potato at high temperatures: responses to gibberellin and growth inhibitors. Ann. Bot. 46:259–265; 1980.
Mignery, G. A.; Piklaard, C. S.; Hannapel, D. J., et al. Isolation and sequence analysis of cDNAs for the major potato tuber protein, patatin. Nucleic Acids Res. 12:7987–8000; 1984.
Mingo-Castel, A. M.; Negm, F. B.; Smith, O. E. Effect of carbon dioxide and ethylene on tuberization of isolated potato stolons cultured in vitro. Plant Physiol. 53:798–801; 1974.
Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol. Plant. 15:473–496; 1962.
Paiva, E.; Lister, R. M.; Park, W. D. Induction and accumulation of major tuber proteins of potato in stems and petioles. Plant Physiol. 71:161–168; 1983.
Paiva, E.; Lister, R. M.; Park, W. D. Comparison of the protein in axillary bud tubers and underground stolon tubers in potato. Am. Pot. J. 59:425–433; 1982.
Palmer, C. E.; Smith, O. E. Effect of kinetin on tuber formation in isolated stems ofSolanum tuberosum L. cultured in vitro. Plant Cell Physiol. 11:303–314; 1970.
Racuson, D.; Foote, M. A major soluble glycoprotein of potato. J. Food Biochem. 4:45–52; 1980.
Rosahl, S.; Eckes, P.; Schell, J., Willmitzer: Organ-specific gene expression in potato: isolation and characterization of tuberspecific cDNA sequences. Mol. Gen. Genet. 202: 368–373; 1986.
Smith, O. E.; Palmer, C. E. Cytokinin-induced tuber formation on stolons ofSolanum tuberosum. Physiol. Plant. 23:599–606; 1970.
Stallknecht, G. F.; Farnsworth, S. General characteristics of coumarin-induced tuberization of axillary shots ofSolanum tuberosum L. cultured in vitro. Am. Pot. J. 59:17–32; 1982.
Towbin, H.; Staehelin, T.; Gordon, J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedures and some applications. Proc. Natl. Acad. Sci. USA 76(9):4350–4354; 1979.
Went, P.; Hu, C. In vitro mass tuberization and virus-free seedpotato production in Taiwan. Am. Pot. J. 59:33–37; 1982.
Wescott, R. J.; Grout, B. W. W.; Henshaw, G. G. Rapid clonal propagation ofSolanum curtilobum cv. Mallku by aseptic shoot meristem culture. Linn. Soc. Symp. Ser.; 1979.
Author information
Authors and Affiliations
Additional information
This work was supported by grants 83-CRCR-1-1348 and 85-CRCR-I-1792 from the U.S. Department of Agriculture Competitive Grants program and with funds from the Texas Agricultural Experiment Station.
Rights and permissions
About this article
Cite this article
Bourque, J.E., Miller, J.C. & Park, W.D. Use of an in vitro tuberization system to study tuber protein gene expression. In Vitro Cell Dev Biol 23, 381–386 (1987). https://doi.org/10.1007/BF02620996
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02620996