Abstract
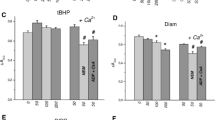
Effects of oxidative stress on isolated rat ventricular myocytes were studied. Myocyte viability was determined by the ability of these cells to retain rod-shaped morphology and to exclude trypan blue. The mean life time of myocytes was quantitated using the Weibull distribution function. Superfusion with 200 μM tert-butyl hydroperoxide (t-BHP) led to a time-dependent loss of cell viability, generation of the products of lipid peroxidation, oxidation of protein and non-protein thiols, a decrease in [ATP]i and in the cellular energy charge. Dithiothreitol (DTT, 5 mM) prolonged survival of myocytes exposed to t-BHP, attenuated oxidation of protein and non-protein thiols, and preserved the energy charge. Exposure to DTT did not affect the concentration of t-BHP-generated lipid peroxidation products. Promethazine (1 μM) prevented t-BHP-induced increase in the concentration of lipid peroxidation products, but did not prevent either loss of thiols or loss of cell viability. Superfusion with N-ethylmaleimide (NEM, 5 μM) also led to loss of cell viability, with accompanying decreases in protein and non-protein thiols, ATP and energy charge without the accumulation of the products of lipid peroxidation. Superfusion with FeSO4 (400 μM) and ascorbate (1 mM), (Fe-Asc) did not result in loss of cell viability or a decrease protein thiols or the energy charge. Superfusion with Fe-Asc, did, however, lead to a slight decrease in the concentration of non-protein thiols and ATP and a large increase in the concentration of lipid peroxidation products. Accumulation of lipid peroxidation products induced by Fe-Asc was prevented by promethazine. These results indicate that free radical-induced irreversible cell injury results from a loss of protein thiols. Changes in the cellular energy charge and lipid peroxidation do not bear a simple relationship to the survival of cardiac myocytes under oxidative stress.
Similar content being viewed by others
References
Meister A, Anderson ME: Glutathione. Ann Rev Biochem 52: 711–760, 1983
Halliwell B, Gutteridge JMC: Free Radicals in Biology and Medicine. Clarendon Press, Oxford, 1985
Halliwell B, Gutteridge JMC: Role of free radicals and catalytic metal ions in human disease: An overview. Methods Enzymol 186: 1–85, 1990
Downey JM: Free radicals and their involvement during long term ischemia and reperfusion. Ann Rev Physiol 52: 487–504, 1990
Harman D: The aging process: major risk factor for disease and death. Proc Natl Acad Sci 88: 5360–5363, 1991
Bregelius R: Mixed disulfides Biological function and increase in oxidative stress. In: H. Seis (ed). Oxidative Stress. Academic Press Inc., New York, pp 243–272, 1985
Kappus H: Lipid peroxidation: Mechanism, analysis, enzymology and biological relevance. In: H. Seis (ed.. Oxidative Stress. Academic Press Inc., New York, pp 273–310, 1985
Timerman AP, Altschuld RA, Hohl CM, Brierley GP, Merola AJ: Cellular glutathione and the response of adult rat heart myocytes to oxidative stress. J Mol Cell Cardiol 22: 565–575, 1990
Vlessis AA, Muller P, Bartos D, Trunkey D: Mechanism of peroxideinduced cellular injury in cultured adult cardiac myocytes. FASEB J 5: 2600–2605, 1991
Hyslop PA, Hinshaw DB, Halsey WA Jr, Scraufstatter IU, Sauerheber RD, Spragg RG, Jackson JH, Cochrane CG: Mechanisms of oxidant mediated cell injury: the glycolytic and mitochondrial pathways of ADP phosphorylation are major intracellular targets inactivated by hydrogen peroxide. J Biol Chem 263: 1665–1675, 1988
Isenberg G, Klockner U: Calcium tolerant ventricular myocytes prepared by preincubation in a ‘KB’ medium. Pflugers Arch 395: 6–18, 1982
Buege JA, Aust SD: Microsomal lipid peroxidation. Methods Enzymol 30: 303–310, 1977
Bradford MM: A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of dye binding. Anal Biochem 72: 248–254, 1976
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ: Protein measurement with the folin phenol reagent. J Biol Chem 193: 265–275, 1951
Bellomo G, Thor H, Orrenius S: Modulation of cellular glutathione and protein thiol status during quinone metabolism. Methods Enzymol 186: 627–635, 1990
Lee ET: Statistical Methods for Survival Data Analysis. Lifetime Learning Publications, Belmont, CA, 1980
Daniel WW: Biostatistics: A foundation for Analysis in the Health Sciences. John Wiley and Sons, New York, 1983
Cleland WW: Dithiothretol, a new protective reagent for SH groups. Biochemistry 3: 480–482, 1964
Beutler E, Srivastava SK, West C: The reversibility of N-ethylmaleimide (NEM) alkylation of red cell glutathione. Biochem Biophys Res Comm 38: 341–347, 1970
Seis H: Hydroperoxides and thiol oxidants in the study of oxidative stress in intact cells and organs. In: H. Seis (ed). Oxidative Stress. Academic Press, New York, 1985, pp 73–90
Kalyanaraman B, Mottley C, Mason RP: A direct spin resonance and spin trapping investigation of peroxyl free radical formation in hematin/hydroperoxide systems. J Biol Chem 258: 3855–3858, 1983
Castro GJ, Bhatnagar A: Effect of extracellular ions and modulators of calcium transport on survival of tert-butyl hydroperoxide exposed to cardiac myocytes. Cardiovas Res 27: 1873–1881, 1993
Donck LV, Reempts JV, Vandeplassche G, Borgers MJ: A new method to study activated oxygen species-induced damage in cardiac myocytes and protection by Ca2+-antagonists. J Mol Cell Cardiol 20: 811–823, 1988
Coetzee WA, Opie LH: Effects of oxygen free radicals on isolated cardiac myocytes from guinea pig ventricle: electrophysiological studies. J Mol Cell Cardiol 24: 651–663, 1992
Bhatnagar A, Srivastava SK, Szabo G: Oxidative stress alters specific membrane currents in isolated cardiac myocytes. Circ Res 67: 535–549, 1990
Josephson RA, Silverman HS, Lakatta EG, Stern MD, Zweier JL: Study of the mechanisms of hydrogen peroxide and hydroxyl free radical-induced cellular injury and calcium overload in cardiac myocytes. J Biol Chem 266: 2354–2361, 1991
Slater TF, Sawyer BC: The stimulatory effects of carbon tetrachloride on peroxidative reactions in rat liver fractionsin vitro. Biochem J 123: 823–828, 1971
Packer JE, Slater TF, Wilson RJ: Reactions of the carbon tetrachloride-related peroxy free radical with amino acids: pulse radiolysis evidence. Life Sci 23: 2617–2620, 1978
Poli G, Cheeseman K, Slater TF, Dianzani MU: The role of lipid peroxidation in CCl4-induced damage to liver microsomal enzymes: comparitive studiesin vitro using microsomes and isolated liver cells. Chem-Biol Interactions 37: 13–24, 1981
Marnett LJ: Peroxyl free radicals: potential mediators of tumor initiation and promotion. Carcinogenesis 8: 1365–1373, 1987
Srivastava SK, Ansari NH, Liu SQ, Izban A, Das B, Szabo G, Bhatnagar A: The effects of oxidants on biomembranes and cellular metabolism. Mol Cell Biochem 91: 149–157, 1989
Kunimoto M, Inoue K, Nojima S: Effect of ferrous ion and ascorbate-induced lipid peroxidation on liposomal membranes. Biochim Biophys Acta 646: 169–178, 1981
Beach DC, Giroux E: Inhibition of lipid peroxidation promoted by iron (III) and ascorbate. Arch Biochem Biophys 297: 258–264, 1992
Laudicina DC, Marnett LJ: Enhancement of hydroperoxide-dependent lipid peroxidation in rat liver microsomes by ascorbic acid. Arch Biochem Biophys 278: 73–80, 1990
Janero DR: Malonaldehyde and thiobarbituric-acid reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radic Biol Med 9: 515–540, 1990
Rush GF, Gorski JR, Ripple MG, Sowinski J, Bugelski P, Hewitt WR: Organic hydroperoxide-induced lipid peroxidation and cell death in isolated hepatocytes. Toxicol Appl Pharmacol 78: 473–483, 1985
Schnellmann RG: Mechanism of t-butyl hydroperoxide-induced toxicity to rabbit renal proximal tubules. Amer J Physiol 255: C28-C33, 1988
Rubin R, Farber JL: Mechanism of the killing of cultured hepatocytes by hydrogen peroxide. Arch Biochem Biophys 228: 450–459, 1984
Atkinson DE: Cellular energy metabolism and its regulation Academic Press, New York, 1977
Kristensen SR: Acritical appraisal of the association between energy charge and cell damage. Biochim Biophys Acta 1012: 272–278, 1989
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bhatnagar, A. Biochemical mechanism of irreversible cell injury caused by free radical-initiated reactions. Mol Cell Biochem 137, 9–16 (1994). https://doi.org/10.1007/BF00926034
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00926034