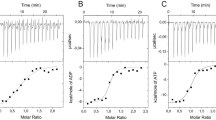
Abstract
Two interesting previously reported properties of mitochondrial F1 ATPase have been confirmed and have been examined by18O exchange measurements to assess if they are consistent with sequential participation of catalytic sites during ATP hydrolysis. These are the ability of HCO −3 to increase reaction rate with apparent loss of cooperative interaction between subunits and the ability of ITP to accelerate the hydrolysis of a low concentration of ATP. The effect of HCO −3 was tested at concentrations of ATP lower than previous measurements. The activation disappeared when ATP was reduced to 0.1 µM. The HCO −3 activation at higher ATP concentrations did not change the extent of reversal of the cleavage of tightly bound ATP at the catalytic site, as measured by the average number of water oxygens incorporated with each Pi formed when 5 or 10 µM ATP is hydrolyzed. The data are consistent with sequential site participation with HCO −3 acceleration of ADP departure after a binding change that stops18O exchange and loosens ADP binding.
When ITP concentration was lowered during net ITP hydrolysis by F1 ATPase an increase in water oxygen incorporation into Pi formed is observed, as noted previously for ATP hydrolysis. The acceleration of the cleavage of a constant low concentration of [γ-18O]ATP by concomitant hydrolysis of increasing concentrations of ITP was accompanied by a decrease in water oxygen incorporation with each Pi formed from the ATP. These results add to evidence for the binding change mechanism for F1 ATPase with sequential participation of catalytic sites.
Similar content being viewed by others
References
Baykov, A. A., and Avaeva, S. M. (1981).Anal. Biochem. 116 1–4.
Boyer, P. D., Kohlbrenner, W. E., McIntosh, D. B., Smith L. T., and O'Neal, C. C. (1982).Ann. N.Y. Acad. Sci. 402 65–83.
Bradford, M. M. (1976).Anal. Biochem. 72 248–254.
Choate, G. L., Hutton, R. L., and Boyer, P. D. (1979).J. Biol. Chem. 254 286–290.
Di Pietro, A., Godinot, C., and Gautheron, D. C. (1983).Biochemistry 22 785–792.
Ebel, R. E., and Lardy, H. A. (1975).J. Biol. Chem. 250 191–196.
Gresser, M. J., Myers, J. A., and Boyer, P. D. (1982).J. Biol. Chem. 257 12030–12038.
Grubmeyer, C., Cross, R. G., and Penefsky, H. S. (1982).J. Biol. Chem. 257 12092–12100.
Hackney, D. D., Stempel, K. E., and Boyer, P. D. (1979).Methods Enzymol. 64 60–83.
Harris, D. A. (1978).Biochim. Biophys. Acta 463 245–273.
Harris, D. A., Dal-Larsen, T., and Klungsoyr, L. (1981).Biochim. Biophys. Acta 635 412–428.
Kohlbrenner, W. E., and Boyer, P. D. (1983).J. Biol. Chem. 258 10887–10894.
Leimgruber, R. M., and Senior, A. E. (1976).J. Biol. Chem. 251 7110–7113.
O'Neal, C. C., and Boyer, P. D. (1984).J. Biol. Chem. 259 5761–5767.
Penefsky, H. S. (1977).J. Biol. Chem. 252 2891–2899.
Penefsky, H. S. (1979).Methods Enzymol. 55 304–308.
Recktenwald, D., and Hess, B. (1980).Biochim. Biophys. Acta 592 372–384.
Schuster, S. M., Gertschen, R. J., and Lardy, H. A. (1976).J. Biol. Chem. 251 6705–6710.
Senda, M., Kanazawa, H., Tsuchiya, T., and Futai, M. (1983).Arch. Biochem. Biophys. 220 398–404.
Sorgato, M. C., Galliazzo, F., Valente, M., Cavallini, L., and Ferguson, S. J. (1982).Biochim. Biophys. Acta 681 319–322.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kasho, V.N., Boyer, P.D. Relationships of inosine triphosphate and bicarbonate effects on F1 ATPase to the binding change mechanism. J Bioenerg Biomembr 16, 407–419 (1984). https://doi.org/10.1007/BF00743235
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00743235