Abstract
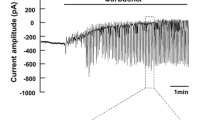
We have performed whole-cell patch-clamp studies on dispersed seccretory cells of the rat mandibular gland to determine how β-adrenergic stimulation causes fluid secretion. When the pipette contained a high K+ solution, the resting membrane potential averaged −33 mV±1.1 (SEM,n=34) and the clamped cell showed strong outward rectification. We monitored K+ and Cl− currents for periods of 15 min by recording the currents needed to clamp the cell potential at 0 and −80 mV, respectively. Isoproterenol (1–2 μmol/l) caused increases in the clamp current at 0 mV (the K+ current) and at −80 mV (the Cl− current) in about 80% of cases, although the responses were variable in size and time-course; the responses were indistinguishable from those induced by acetylcholine or the Ca2+ ionophore, A23187. The α-adrenergic antagonist, phentolamine (1–2 μmol/l), had no effect on the response, but the β-adrenergic antagonist, propranolol (10 μmol/l), blocked it completely. The isoproterenol response could not be mimicked by application to either surface of the cell membrane, of cyclic AMP (100 μmol/l), forskolin (1 or 20 μmol/l) or cholera toxin (2.5 μg/ml). However, increasing the Ca2+-chelating capacity of the pipette solution by raising its EGTA concentration from the customary 0.5 to 20 mmol/l, blocked the response to isoproterenol, suggesting that β-adrenergic agonists activate Cl− and K+ channels by raising cytosolic Ca2+. Since neomycin, which blocks phospholipase C, blocked the action of isoproterenol without impairing the cell responsiveness to A23187, it appears that isoproterenol, like muscarinic agonists, increased cytosolic Ca2+ via the phosphatidylinositol cycle.
Similar content being viewed by others
References
Alexander JH, Lennep EW van, Young JA (1972) Water and electrolyte secretion by the exorbital lacrimal gland of the rat studied by micropuncture and catheterization techniques. Pflügers Arch 337:299–309
Argent BE, Arkle S (1985) Mechanism of action of extracellular calcium on isoprenaline-evoked amylase secretion from isolated rat parotid glands. J Physiol (Lond) 369:337–353
Aub DL, Putney JW (1985) Properties of receptor-controlled inositol trisphosphate formation in parotid acinar cells. Biochem J 225:263–266
Butcher FR (1978) Calcium and cyclic nucleotides in the regulation of secretion from the rat parotid by autonomic agonists. In: George WJ, Ignarro LJ, Greengard P, Robison GA (eds) Third international conference on cyclic nucleotides (Advances in cyclic nucleotide research, vol 9). Raven Press, New York, pp 707–722
Butcher FR (1980) Regulation of calcium efflux from isolated rat parotid cells. Biochim Biophys Acta 630:254–260
Butcher FR, Putney JW (1980) Regulation of parotid gland function by cyclic nucleotides and calcium. In: Greengard P, Robison GA (eds) Advances in cyclic nucleotide research, vol 13. Raven Press, New York, pp 215–249
Cook DI, Gard GB, Champion M, Young JA (1988) Patch-clamp studies of the electrolyte secretory mechanism of rat mandibular gland cells stimulated with acetylcholine or isoproterenol. In: Thorn NA, Treiman M, Petersen OH, Thaysen JH (eds) Molecular mechanisms in secretion (25th Alfred Benzon Symposium, 1987). Munksgaard, Copenhagen, pp 133–151
Doughney C, Dormer RL, McPherson MA (1987) Adrenergic regulation of formation of inositol phosphate in rat submandibular acini. Biochem J 241:705–709
Dreux C, Imhoff V, Huleux C, Busson S, Rossignol B (1986) Forskolin, a tool for rat parotid secretion studies:45Ca efflux is not related to cAMP. Am J Physiol 251:C754-C762
Evans MG, Marty A (1986) Calcium-dependent chloride currents in isolated cells from rat lacrimal glands. J Physiol (Lond) 378:437–460
Findlay I, Petersen OH (1985) Acetylcholine stimulates a Ca2+-dependent Cl− conductance in mouse lacrimal acinar cells. Pflügers Arch 403:328–330
Gallacher DV, Morris AP (1986) A patch-clamp study of potassium currents in resting and acetylcholine-stimulated mouse submandibular acinar cells. J Physiol (Lond) 373:379–395
Greger R, Schlatter E, Gögelein H (1987) Chloride channels in the apical membrane of the rectal gland of the dogfish (Squalus acanthias). Properties in the “larger” conductance channel. Pflügers Arch 409:114–121
Helman J, Ambudkar IS, Baum BJ (1987) Adrenoceptor mobilization of calcium in rat submandibular cells. Eur J Pharmacol 143:65–72
Hunter M, Smith PA, Case RM (1983) The dependence of fluid secretion by mandibular salivary gland and pancreas on extracellular calcium. Cell Calcium 4:307–317
Iwatsuki N, Maruyama Y, Matsumoto O, Nishiyama A (1985) Activation of Ca2+-dependent Cl− and K+ conductances in rat and mouse parotid acinar cells. Jpn J Physiol 35:933–944
Kanagasuntheram P, Randle PJ (1976) Calcium metabolism and amylase release in rat parotid acinar cells. Biochem J 160:547–564
Llano I, Marty A (1987) Protein kinase C activators inhibit the inositol triphosphate mediated muscarinic current responses in rat lacrimal cells. J Physiol (Lond) 394:239–248
Llano I, Marty A, Tanguy J (1987) Dependence of intracellular effects of GTPγS and inositoltrisphosphate on cell membrane potential and on external Ca ions. Pflügers Arch 409:499–306
Martinez JR, Cassity N (1985)36Cl fluxes in dispersed rat submandibular acini: effects of acetylcholine and transport inhibitors. Pflügers Arch 403:50–54
Marty A, Tan YP, Trautmann A (1984) Three types of calcium-dependent channels in rat lacrimal glands. J Physiol (Lond) 357:293–325
Marty A, Horn R, Zimmerberg J (1987) Muscarinic response in rat lacrimal gland. In: Thorn NA, Trieman M, Petersen OH, Thaysen JH (eds) Molecular mechanisms in secretion, Abstracts (25th Alfred Benzon Symposium) Munksgaard, Copenhagen, p 25
McPherson MA, Dormer RL (1984) Mucin release and calcium fluxes in isolated rat submandibular acini. Biochem J 224: 473–481
Miller BE, Nelson DL (1977) Calcium fluxes in isolated acinar cells from rat parotid. Effect of adrenergic and cholinergic stimulation. J Biol Chem 252:3629–3636
Morris AP, Gallacher DV, Irvine RF, Petersen OH (1987) Synergism of inositol trisphosphate and tetrakisphosphate in activating Ca2+-dependent K+ channels. Nature 330:653–655
Nauntofte B, Poulsen JH (1986) Effects of Ca2+ and furosemide on Cl− transport and O2 uptake in rat parotid acini. Am J Physiol 251:C175-C185
Novak I, Young JA (1986) Two independent anion transport systems in rabbit mandibular salivary glands. Pflügers Arch 407:649–656
Petersen OH, Maruyama Y (1984) Calcium-activated potassium channels and their role in secretion. Nature 307:693–696
Putney JW (1978) On the role of cellular calcium in the response of the parotid to dibutyryl and monobutyryl cyclic AMP. Life Sci 22:631–638
Putney JW, Weiss SJ, Leslie BA, Marier SH (1977) Is calcium the final mediator of exocytosis in the rat parotid gland? J Pharmacol Exp Ther 203:144–155
Putney JW, Van de Walle, CM, Leslie BA (1978) Receptor control of calcium influx in parotid acinar cells. Mol Pharmacol 14:1046–1053
Quissell DO, Barzen KA, Lafferty JL (1981) Role of calcium and cAMP in the regulation of rat submandibular mucin secretion. Am J Physiol 241:C76-C85
Silva P, Stoff J, Field M, Fine L, Forrest JN, Epstein FH (1977) Mechanism of active chloride secretion by shark rectal gland: role of Na−K-ATPase in chloride transport. Am J Physiol 233:F298-F306
Suzuki K, Petersen OH (1985) The effect of Na+ and Cl− removal and of loop diuretics on acetylcholine-evoked membrane potential changes in mouse lacrimal acinar cells. Q J Exp Physiol 70:437–445
Takemura H (1985) Changes in cytosolic free calcium concentration in isolated rat parotid cells by cholinergic and β-adrenergic agonists. Biochem Biophys Res Commun 131:1048–1055
Trautmann A, Marty A (1984) Activation of Ca-dependent K channels by carbamylcholine in rat lacrimal glands. Proc Natl Acad Sci USA 81:611–615
Vergara J, Tsien RY, Delay M (1985) Inositol 1,4,5-triphosphate: a possible chemical link in excitation-contraction coupling in muscle. Proc Natl Acad Sci USA 82:6352–6356
Welsh MJ (1986) Single apical membrane anion channels in primary cultures of canine tracheal epithelium. Pflügers Arch 407 (Suppl 2):S116-S122
Young JA, Lennep EW van (1978) The morphology of salivary glands. Academic Press, London
Young JA, Martin CJ (1971) The effect of a sympatho- and a parasympathomimetic drug on the electrolyte concentrations of primary and final saliva of the rat submaxillary gland. Pflügers Arch 327:285–302
Author information
Authors and Affiliations
Additional information
This project was supported by the National Health and Medical Research Council of Australia
Rights and permissions
About this article
Cite this article
Cook, D.I., Day, M.L., Champion, M.P. et al. Ca2+ not cyclic AMP mediates the fluid secretory response to isoproterenol in the rat mandibular salivary gland: Whole-cell patch-clamp studies. Pflugers Arch. 413, 67–76 (1988). https://doi.org/10.1007/BF00581230
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00581230