Abstract
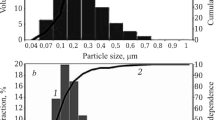
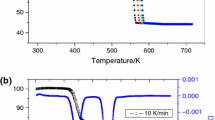
Particle evolution during and following the decomposition of cadmium carbonate was investigated using surface area and crystallite size analyses. Surface area results of decompositions at temperatures ranging from 523 to 775 K showed a maximum corresponding to the total decomposition of the carbonate. The maxima for vacuum and air decomposition occurred at 583 and 613 K, respectively. Crystallite size analyses showed that the sizes of the CdO resulting from the air decomposition of CdCO3 at low temperatures was significantly larger than the corresponding values obtained for vacuum decomposition. These observations suggest that CdCO3 sinters favourably in air before decomposition, leading to larger CdO crystallites after decomposition. After complete dissociation, the evolution of CdO particles is the same for vacuum and air decomposition, indicating that the sintering of CdO is essentially the same under these two conditions.
Similar content being viewed by others
References
B. J. Asirvatham and Z. A. Munir, J. Mater. Sci. 21 (1986) 1997.
R. S. Mikhail, D. Dollimore, A. M. Karnel and N. R. El-Nazer, J. Appl. Chem. Biotechnol. 23 (1973) 419.
Idem, ibid. 23 (1973) 431.
P. Dumas, N. Ea, J. C. Niepce and G. Watelle, J. Solid State Chem. 27 (1979) 317.
B. D. Cullity, “Elements of X-ray Diffraction”, 2nd Edn (Addison-Wesley, Reading, Massachusetts, 1978) p. 284.
Z. A. Munir and P. G. Coombs, Met. Trans. 148 (1983) 95.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Munir, Z.A., Asirvatham, B.J. Particle evolution during the decomposition of CdCO3 . J Mater Sci 21, 2002–2006 (1986). https://doi.org/10.1007/BF00547940
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00547940