Abstract
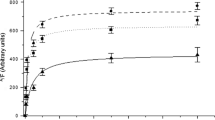
This comparative study investigates the relationship between sarcoplasmic reticulum (SR) calcium(Ca2+)-ATPase transport activity and phospholamban (PLB) phosphorylation in whole cardiac homogenates of spo`ntaneously hypertensive rats (SHR) and their parent, normotensive Wistar Kyoto (WKY) strain during early postnatal development at days 1, 3, 6, 12 and at day 40 to ascertain any difference in SR Ca2+ handling before the onset of hypertension. At day 1, the rate of homogenate oxalate-supported Ca2+ uptake was significantly higher in SHR than in WKY (0.25 ± 0.02 vs 0.12 ± 0.01 nmoles Ca2+/mg wet ventricular weight/min, respectively; p < 0.001). This interstrain difference disappeared with further developmental increase in SR Ca2+ transport. Western Blot analysis and a semiquantitative ELISA did not reveal any difference in the amount of immunoreactive PLB (per mg of total tissue protein) between strains at any of the ages studied. In addition, levels of phosphorylated PLB formed in vitro in the presence of radiolabelled ATP and catalytic (C) subunit of protein kinase A did not differ between SHR and WKY at days 1, 3, 6 and 12. At day 40, C subunit-catalyzed formation of 32P-PLB was reduced by 66% (p < 0.001) in SHR when compared to age-matched WKY In the early postnatal period between day 1 and 12 SR Ca2+-transport values were linearly related to the respective 32P-PLB levels of both SHR and WKY rats. The results indicate that cardiac SR of SHR can sequester Ca2+ at a much higher rate immediately after birth compared to WKY rats. The disappearance of this interstrain difference with further development suggests that some endogenous neuroendocrine or nutritional factor(s) from the hypertensive mother may exert an influence upon the developing heart in utero resulting in a transiently advanced maturation of the SR Ca2+ transport function in SHR pups at the time of birth.
Similar content being viewed by others
References
Fozzard H: Heart: Excitation-contraction coupling. Annu Rev Physiol 41:201–220, 1977
Maylie JG: Excitation-contraction coupling in neonatal and adult myocardium of cat. Am J Physiol 242: H834-H843, 1982
Nakanishi T, Seguchi M, Takao A: Development of the myocardial contractile system. Experientia 44: 936–944, 1988
Chemla D, Lecarpentier Y, Martin JL, Clergue M, Antonetti A, Hatt PY: Relationship between inotropy and relaxation in rat myocardium. Am J Physiol 250: H1008-H1016, 1986
Komuro I, Kurabayashi M, Shibazaki Y, Takaku F, Yazaki Y: Molecular cloning and characterization of a Ca2+ +Mg2+-dependent adenosine triphosphatase from rat cardiac sarcoplasmic reticulum. Regulation of its expression by pressure overload and developmental stage. J Clin Invest 83: 1102–1108, 1989
Lompre AM, Lambert F, Lakatta EG, Schwartz K: Expression of sarcoplasmic reticulum Ca2+-ATPase and calsequestrin genes in rat heart during ontogenic development and aging. Circ Res 69: 1380–1388, 1991
Nayler WG, Fassold E: Calcium accumulating and ATPase activity of cardiac sarcoplasmic reticulum before and after birth. Cardiovasc Res 11: 231–237, 1977
Fisher DJ, Tate CA, Phillips S: Developmental regulation of the sarcoplasmic reticulum calcium pump in the rabbit heart. Pediatr Res 31: 474–479, 1992
Vetter R, Kemsies C, Schulze W: Sarcolemmal Na+-Ca2+ exchange and sarcoplasmic reticulum Ca2+ uptake in several cardiac preparations. Biomed Biochim Acta 46: S375-S381, 1987
Katz AM, Nash-Adler P, Watras J, Messineo FC, Takenaka H, Louis CF: Fatty acid effects on calcium influx and efflux in sarcoplasmic reticulum vesicles from rabbit skeletal muscle. Biochim Biophys Acta 687: 17–26, 1982
Martonosi A, Donley J, Halpin RA: Sarcoplasmic reticulum. 3. The role of phospholipids in the adenosine triphosphatase activity and Ca2+ transport. J Biol Chem 243: 61–70, 1968
Colyer J: Control of the calcium pump of cardiac sarcoplasmic reticulum. A specific role for the pentameric structure of phospholamban? Cardiovasc Res 27: 1766–1771, 1993
Wegener AD, Simmerman HK, Lindemann JP, Jones LR: Phospholamban phosphorylation in intact ventricles. Phosphorylation of serme 16 and threonine 17 in response to beta-adrenergic stimulation. J Biol Chem 264: 11468–11474, 1989
Tada M, Katz AM: Phosphorylation of the sarcoplasmic reticulum and sarcolemma. Annu Rev Physiol 44: 401–423, 1982
Lindemann JP, Watanabe AM: Phosphorylation of phospholamban in intact myocardium. Role of Ca2+-calmodulin-dependent mechanisms. J Biol Chem 260: 4516–4525, 1985
Trippodo NC, Frohlich ED: Similarities of genetic (spontaneous) hypertension. Man and rat. Circ Res 48: 309–319, 1981
Clubb FJ, Jr., Bell PD, Kriseman JD, Bishop SP: Myocardial cell growth and blood pressure development in neonatal spontaneously hypertensive rats. Lab Invest 56: 189–197, 1987
Gray SD: Spontaneous hypertension in the neonatal rat. A review. Clin Exp Hypertens A 6: 755–781, 1984
Bing OHL, Brooks WW, Conrad CH, Sen S, Perreault CL, Morgan JP: Intracellular calcium transients in myocardium from spontaneously hypertensive rats during the transition to heart failure. Circ Res 68: 1390–1400, 1991
Brooksby P, Levi AJ, Jones JV: Contractile properties of ventricular myocytes isolated from spontaneously hypertensive rat. J Hypertens 10: 521–527, 1992
Lammerich A, Günther J, Storch E, Pfitzer G, Vetter R: Alterations of cardiac contractile function are related to changes in membrane calcium transport in spontaneously hypertensive rats. J Hypertens 13: 1313–1224, 1995
Feher JJ, Briggs FN, Hess ML: Characterization of cardiac sarcoplasmic reticulum from ischemic myocardium: Comparison of isolated sarcoplasmic reticulum with unfractionated homogenates. J Mol Cell Cardiol 12: 427–432, 1980
Solaro RJ, Briggs FN: Estimating the functional capabilities of sarcoplasmic reticulum in cardiac muscle. Calcium binding. Circ Res 34: 531–540, 1974
Vetter R, Rupp H: CPT-1 inhibition by etomoxir has a chamber-related action on cardiac sarcoplasmic reticulum and isomyosins. Am J Physiol 267: H2091-H2099, 1994
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ: Protein measurement with the folin phenol reagent. J Biol Chem 193: 265–275, 1951
Tanaka H, Shigenobu K: Effect of ryanodine on neonatal and adult rat heart: developmental increase in sarcoplasmic reticulum function. J Mol Cell Cardiol 21: 1305–1313, 1989
Vetter R, Will H, Küttner I, Kemsies C, Will Shahab L: Developmental changes of Ca++ transport systems in chick heart. Biomed Biochim Acta 45: S219-S222, 1986
Vetter R, Studer R, Reinecke H, Kolar F, Ostadalova I, Drexler H: Reciprocal changes in the postnatal expression of the sarcolemma) Na+-Ca2+-exchanger and SERCA2 in rat heart. J Mol Cell Cardiol 27: 1689–1701, 1995
Arai M, Otsu K, MacLennan DH, Periasamy M: Regulation of sarcoplasmic reticulum gene expression during cardiac and skeletal muscle development. Am J Physiol 262: C614-C620, 1992
Karczewski P, Vetter R, Holtzhauer M, Krause EG: Indirect technique for the estimation of cAMP-dependent and Ca 2+/calmodulin-dependent phospholamban phosphorylation state in canine heart in vivo. Biomed Biochim Acta 45: S227-S231, 1986
Karczewski P, Bartel S, Krause EG: Differential sensitivity to isoprenaline of troponin I and phospholamban phosphorylation in isolated rat hearts. Biochim J 266: 115–122, 1990
Suzuki T, Lui P, Wang JH: The use of monoclonal antibodies for the species and tissues distribution of phospholamban. Cell Calcium 7: 41–47, 1986
Napolitano R, Vittone L, Mundina C, Chiappe de Cingolani G, Mattiazzi A: Phosphorylation of phospholamban in the intact heart. A study on the physiological role of the Ca2+-calmodulin-dependent protein kinase system. J Mol Cell Cardiol 24: 387–396, 1992
Hawkins C, Xu A, Narayanan N: Sarcoplasmic reticulum calcium pump in cardiac and slow twitch skeletal muscle but not fast twitch skeletal muscle undergoes phosphorylation by endogenous and exogenous Ca2+/calmodulin-dependent protein kinase. Characterization of optimal conditions for calcium pump phosphorylation. J Biol Chem 269: 31198–31206, 1994
Anversa P, Olivetti G, Loud AV: Morphometric study of early postnatal development in the left and right ventricular myocardium of the rat. I. Hypertrophy, hyperplasia and binucleation of myocytes. Circ Res 46: 495–502, 1980
Rakusan K: Assessment of cardiac growth In: R. Zak (ed.) Growth of the Heart in Health and Disease. Raven Press, New York, 1984, pp 25–40
Gupta MP, Gupta M, Jakovcic S, Zak R: Catecholamines and cardiac growth. Mol Cell Biochem (this volume)
Rohrer D, Dillmann WH: Thyroid hormone markedly increases the mRNA coding for sarcoplasmic reticulum Ca2+-ATPase in the rat heart. J Biol Chem 263: 6941–6944, 1988
Beekman RE, van Hardeveld C, Simonides WS: On the mechanism of the reduction by thyroid hormone of beta-adrenergic relaxation rate stimulation in rat heart. Biochem J 259: 229–236, 1989
Vetter R, Kott M, Rupp H: Differential influences of carnitine palmitoyltransferase-I inhibition and hyperthyroidism on cardiac growth and sarcoplasmic reticulum phosphorylation. Eur Heart J 15 (Suppl. D): 31–37, 1994
Kirby ML: Role of extracardiac factors in heart development. Experientia 44: 944–951, 1988
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Freestone, N., Singh, J., Krause, EG. et al. Early postnatal changes in sarcoplasmic reticulum calcium transport function in spontaneously hypertensive rats. Mol Cell Biochem 163, 57–66 (1996). https://doi.org/10.1007/BF00408641
Issue Date:
DOI: https://doi.org/10.1007/BF00408641