Abstract
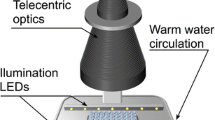
A new computer-controlled measurement system for assessing beat rates of spontaneously contracting cultured heart cells is presented. The system overcomes several disadvantages of established techniques such as: (i) lack of precise control of the environment (pH, temperature, humidity); (ii) restriction to the measurement of one culture at a time; (iii) inability to obtain long-term measurements. The beat rate is recorded by subjecting monolayer cultures to dark field illumination and recording contraction-related changes in light scattering. A maximum of environmental stability is achieved because measurements are performed in the incubator. Beat rates of up to 16 individual culture dishes can be assessed repeatedly during freely selectable time intervals. Control of the experiments, data acquisition and data analysis are carried out by a computer. The specific advantage of the method lies in the ability to measure the beat rate of several culture dishes continuously over time intervals limited only by the viability of the cultures, i.e. up to several weeks.
Similar content being viewed by others
References
Acosta D, Sorensen EMB, Anuforo DC, Mitchell DB, Ramos K, Santone KS, Smith AM (1985) An in vitro approach to the study of target organ toxicity of drugs and chemicals. In Vitro Cell Dev Biol 21: 495–504
Baum VC, Miletich DJ, Curran MJ (1987) Bupivacaine arrhythmogenicity in heart cell culture: effect of progesterone. Anesthesiology 67: A439
Boder GB, Harley RJ, Johnson IS (1971) Recording system for monitoring automaticity of heart cells in culture. Nature 231: 531–532
Bucher OM (1957) A photoelectric recording set for pulsation curves of heart muscle cultures in vitro. Exp Cell Res 13: 109–115
Csete K, Auclair MC, Lechat P (1979) Chronotropic effect of histamine on cultured neonatal rat heart cells. Eur J Pharmacol 58: 117–123
Fayet G, Couraud F, Miranda F, Lissitzky S (1974) Electro-optical system for monitoring activity of heart cells in culture: application to the study of several drugs and scorpion toxins. Eur J Pharmacol 27: 165–174
Harary I, Wallace G, Bristol G (1983) A video-computer for the chronotropic and inotropic measurements of the beating of cultured heart cells. Cytometry 3: 367–374
Kaufmann R, Tritthart H, Rodenroth S, Rost B (1969) The mechanical and electrical activity of embryonic chick heart cells cultured in vitro. Pflügers Arch 311: 25–49
Kaufmann AJ, Wittmann R, Birnbaumer L, Hoppe BH (1977) Activation of myocardial β-adrenoceptors by the nitrogen-free low affinity ligand 3′,4′-dihydroxy-alpha-methylpropiophenone (U-0521). Naunyn Schmiedebergs Arch Pharmacol 296: 217–228
Koidl B, Tritthart HA, Erkinger S (1980) Cultured embryonic chick heart cells: photometric measurement of the cell pulsation and the effect of calcium ions, electrical stimulation and temperature. J Mol Cell Cardiol 12: 165–178
Mark GE, Strasser FF (1966) Pacemaker activity and mitosis in cultures of newborn rat heart ventricle cells. Exp Cell Res 44: 217–233
Mashimo K (1985) Chronotropic responses to ethanol of mouse atrial and ventricular myocardial cells in culture. Jpn J Physiol 35: 659–671
Miletich DJ, Khan A, Albrecht RF, Jozefiak A (1983) Use of heart cell cultures as a tool for the evaluation of halothane arrhythmia. Toxicol Appl Pharmacol 70: 181–187
Mohamed SNW, Holmes R, Hartzell CR (1983) A serum-free, chemically-defined medium for function and growth of primary neonatal rat heart cell cultures. In Vitro 19: 471–478
Okarma TB, Kalman SM (1971) Photoelectric monitoring of single beating heart cells in culture. Exp Cell Res 69: 128–134
Rohr S (1987) Continuous measurement of beat frequency of monolayer myocyte cultures under incubation conditions. Experientia 43: 718
Rohr S (1989) Serum transiently increases the beat rate of cultured neonatal rat heart cells. Experientia 45: A72
Schanne OF (1970) Recording of contractile activity of cells in culture. J Appl Physiol 29: 892–893
Speicher DW, McCarl RL (1974) Pancreatic enzyme requirements for the dissociation of rat hearts for culture. In Vitro 10: 30–41
Stong LJ, Hartzell CR, McCarl RL (1975) Halothane and the beating response and ATP turnover rate of heart cells in tissue culture. Anesthesiology 42: 123–132
Van Brussel E, Freyss-Beguin M, Simonin G, Mathieu-Levy N, Lechat P (1979) Concentration optimale de trypsine pour les cultures de cellules cardiaques de rat. C R Soc Biol (Paris) 173: 1015–1022
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rohr, S. A computerized device for long-term measurements of the contraction frequency of cultured rat heart cells under stable incubating conditions. Pflügers Arch 416, 201–206 (1990). https://doi.org/10.1007/BF00370243
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00370243