Abstract
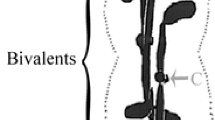
Evidence from several cell types indicates that chromatin can induce microtubule assembly in its vicinity. To determine whether this activity is present in sperm chromatin, whose biochemical composition differs from somatic chromatin, mouse oocytes that were undergoing meiotic maturation were inseminated. Maturing oocytes are not activated by sperm penetration but remain arrested at metaphase. The sperm chromatin within the oocyte cytoplasm initially became dispersed and later, under the influence of oocyte cytoplasmic factors, recondensed into a small mass of individual chromosomes. When inseminated oocytes were processed for immunofluorescence using an anti-α-tubulin antibody, microtubules were never associated with dispersed sperm chromatin, although the chromosomes of the oocyte were arranged on a spindle. In contrast, microtubules were associated with the majority of sperm nuclei that had become recondensed, and were frequently arranged into a spindle-like structure. When oocytes had been penetrated by more than three sperm, most sperm nuclei remained at the dispersed chromatin stage and these were never associated with microtubules. Exposure of polyspermic oocytes to taxol, which promotes microtubule assembly, failed to induce microtubule assembly around dispersed sperm chromatin. Exposure of monospermic oocytes to nocodazole, which inhibits tubulin polymerization, prevented resolution of the recondensed sperm chromatin into individual chromosomes. These results suggest that sperm chromatin lacks an activity that can induce local microtubule assembly, and that it acquires this activity once modified by oocyte cytoplasmic factors.
Similar content being viewed by others
References
AlexandreH, CauwenbergeAV, MulnardJ (1989) Involvement of microtubules and microfilaments in the control of the nuclear movement during maturation of mouse oocytes. Dev Biol 136:311–320
BalhornR (1982) A model for the structure of chromatin in mammalian sperm. J Cell Biol 93:298–305
ChurchK, NicklasRB, LinHPP (1986) Micromanipulated bivalents can trigger minispindle formation in Drosophila melanogaster spermatocyte cytoplasm. J Cell Biol 103:2765–2773
ClarkeHJ, MasuiY (1986) Transformation of sperm nuclei to metaphase chromosomes in the cytoplasm of maturing oocytes of the mouse. J Cell Biol 102:1039–1046
ClarkeHJ, MasuiY (1987) Dose-dependent relationship between oocyte cytoplasmic volume and transformation of sperm nuclei to metaphase chromosomes. J Cell Biol 104:831–840
De BrabanderM, GevensG, NuydensR, WillebrordsR, AertsF, De MeyJ (1986) Microtubule dynamics during the cell cycle: The effects of taxol and nocodazole on the microtubule system of PTK2 cells at different stages of the mitotic cycle. Int Rev Cytol 101:215–275
EcklundPS, LevineL (1975) Mouse sperm basic nuclear protein: Electrophoretic characterization and fate after fertilization. J Cell Biol 66:251–262
ElinsonRP (1975) Fertilization of immature frog eggs: cleavage and development following subsequent activation. J Embryol Exp Morphol 37:187–201
HoulistonE, PickeringSJ, MaroB (1987) Redistribution of microtubules and pericentriolar material during the development of polarity in mouse blastomeres. J Cell Biol 104:1299–1308
InouéM, WolfDP (1974) Comparative solubility properties of the zona pellucidae of unfertilized mouse ova. Biol Reprod 11:558–565
IwamatsuT, ChangMC (1972) Sperm penetration in vitro of mouse oocytes at various times during maturation. J Reprod Fertil 31:237–247
KarsentiE, NewportJ, HubbleR, KirschnerMW (1984) Interconversion of metaphase and interphase microtubule arrays, as studied by the injection of centrosomes and nuclei into Xenopus eggs. J Cell Biol 98:1730–1745
KirschnerMW, MitchisonT (1986) Beyond self assembly: From microtubules to morphogenesis. Cell 45:329–342
KubiakJZ (1989) Mouse oocytes gradually develop the capacity for activation during the metaphase II arrest. Dev Biol 136:537–545
KubiakJC, WeberM, GéraudG, MaroB (1992) Cell cycle modifications during the transitions between meiotic M-phases in mouse oocytes. J Cell Sci 102:457–467
LongoFJ (1973) Fertilization: A comparative ultrastructural review. Biol Reprod 9:149–215
MaroB, HowlettSK, WebbM (1985) Non-spindle microtubule organizing centers in metaphase II-arrested mouse oocytes. J Cell Biol 101:1665–1672
MaroB, JohnsonMH, WebbM, FlachG (1986) Mechanism of polar body formation in the mouse oocyte: an interaction between the chromosomes, the cytoskeleton and the plasma membrane. J Embryol Exp Morphol 92:11–32
McIntoshJR, KoonceMP (1989) Mitosis. Science 246:622–628
MillerMA, MasuiY (1982) Changes in the stainability and sulfhydryl level in the sperm nucleus during sperm-oocyte interaction in mice. Gamete Res 5:167–179
NicklasRB, GordonGW (1985) The total number of spindle microtubules depends on the number of chromosomes present. J Cell Biol 100:1–7
NonchevS, TsanevR (1990) Protamine-histone replacement and DNA replication in the male mouse pronucleus. Mol Repord Dev 25:72–76
RodmanTC, PruslinFH, HoffmanHP, AllfreyVG (1981) Turnover of basic chromosomal proteins in fertilized eggs: A cytoimmunochemical study of events in vivo. J Cell Biol 90:351–364
SawinKE, MitchisonTJ (1991) Mitotic spindle assembly by two different pathways in vitro. J Cell Biol 112:925–940
SchattenG, SimerlyC, SchattenH (1985) Microtubule configurations during fertilization, mitosis, and early development in the mouse and the requirement for egg microtubule-mediated motility during mammalian fertilization. Proc Natl Acad Sci USA 82:4152–4156
SchroederAC, EppigJJ (1984) The developmental capacity of mouse oocytes that matured spontaneously in vitro is normal. Dev Biol 104:831–840
SzollosiDP, CalarcoP, DonahueRP (1972) Absence of centrioles in the second meiotic spindles of mouse oocytes. J Cell Sci 11:521–541
TarkowskiAK (1966) An air-drying method for chromosome preparations for mouse eggs. Cytogenetics 5:394–400
TheurkaufWE, HawleyRS (1992) Meiotic spindle assembly in Drosophila females: Behavior of nonexchange chromosomes and the effects of mutations in the nod kinesin-like protein. J Cell Biol 116:1167–1180
Van BlerkomJ (1991) Microtubule mediation of cytoplasmic and nuclear maturation during the early stages of resumed meiosis in cultured mouse oocytes. Proc Natl Acad Sci USA 88:5031–5035
Author information
Authors and Affiliations
Additional information
Communicated by: J.B. Rattner
Rights and permissions
About this article
Cite this article
Harrouk, W., Clarke, H.J. Sperm chromatin acquires an activity that induces microtubule assembly during residence in the cytoplasm of metaphase oocytes of the mouse. Chromosoma 102, 279–286 (1993). https://doi.org/10.1007/BF00352402
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00352402