Abstract
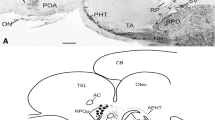
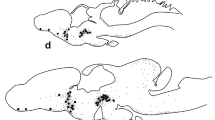
The brain of adult bullfrogs (Rana catesbeiana) contains six populations of cells which are immunoreactive for the neurohypophysial peptide arginine vasotocin (AVT). It is unknown when some of these cell populations first appear during development and when the sexual differences in AVT distribution first become apparent. We therefore used immunocytochemistry to examine development of AVT pathways in developing bullfrog tadpoles and in newly metamorphosed froglets of both sexes. AVT-immunoreactive (AVT-ir) cells were already present in the three diencephalic areas (magnocellular preoptic nucleus, suprachiasmatic nucleus and hypothalamus) at stage III (Taylor and Kollros stages), the earliest stage examined. Cell size in the magnocellular nucleus was not bimodally distributed in either tadpoles or froglets. AVT-ir cells in the telencephalic septal nucleus and amygdala did not appear until stage VI. There was no sexual difference in the density of AVT-ir cells or fibers in the amygdala of tadpoles or froglets. Finally, cells in the hindbrain pretrigeminal nucleus appeared much later-after stage XX. Thus, different populations of neurons begin to express AVT at unique times during development. The sexual dimorphism in AVT content observed in the amygdala of adult bullfrogs must appear during juvenile development or at adulthood.
Similar content being viewed by others
References
Alvarado RH, Johnson SR (1966) The effects of neurohypophsial hormones on water and sodium balance in larval and adult bullfrogs (Rana catesbeiana). Comp Biochem Physiol 18:549–561
Atwell WJ (1918) The development of the hypophysis of the Anura. Anat Rec 15:73–92
Bamshad M, Novak MA, De Vries GJ (1993) Sex and species differences in the vasopressin innervation of sexually naive and parental prairie voles, Microtus ochrogaster and meadow voles, Microtus pennsylvanicus. J Neuroendocrinol 5:247–255
Bentley PJ, Greenwald L (1970) Neurohypophysial function in bullfrog (Rana catesbeiana) tadpoles. Gen Comp Endocrinol 14:412–415
Boer GJ (1985) Vasopressin and brain development: studies using the Brattleboro rat. Peptides [Suppl 1] 6:49–62
Boer GJ (1987) Development of vasopressin systems and their functions. In: Gash DM, Boer GJ (eds) Vasopressin, principles and properties. Plenum Press, New York, pp 117–174
Boer GJ, Swaab DF, Uylings HBM, Boer K, Buijs RM, Velis DN (1980) Neuropeptides in rat brain development. Prog Brain Res 53:207–227
Boyd SK (1991) Effect of vasotocin on locomotor activity in bull-frogs varies with developmental stage and sex. Horm Behav 25:57–69
Boyd SK (1992) Sexual differences in hormonal control of release calls in bullfrogs. Horm Behav 26:522–535
Boyd SK, Moore FL (1992) Sexually dimorphic concentrations of arginine vasotocin in sensory regions of the amphibian brain. Brain Res 588:304–306
Boyd SK, Tyler CJ, De Vries GJ (1992) Sexual dimorphism in the vasotocin system of the bullfrog (Rana catesbeiana). J Comp Neurol 325:313–325
Buijs RM, Velis DN, Swaab DF (1980) Ontogeny of vasopressin and oxytocin in the fetal rat: early vasopressinergic innervation of the fetal brain. Peptides 1:315–324
Buijs RM, Pévet P, Masson-Pévet M, Pool CW, De Vries GJ, Canguilhem B, Vivien-Roels B (1986) Seasonal variation in vasopressin innervation in the brain of the European hamster (Cricetus cricetus). Brain Res 371:193–196
Carr JA, Norris DO (1990) Immunohistochemical localization of corticotropin-releasing factor and arginine vasotocin-like immunoreactivities in the brain and pituitary of the American bullfrog (Rana catesheiana) during development and metamorphosis. Gen Comp Endocrinol 78:180–188
Chetverukhin VK, Polenov AL (1993) Ultrastructural radioautographic analysis of neurogenesis in the hypothalamus of the adult frog, Rama temporaria, with special reference to physiological regeneration of the preoptic nucleus. I. Ventricular zone cell proliferation. Cell Tissue Res 271:341–350
Choy VJ, Watkins WB (1979) Maturation of the hypothalamo-neurohypophysial system. I. Localization of neurophysin, oxytocin and vasopressin in the hypothalamus and neural lobe of the developing rat brain. Cell Tissue Res 197:325–336
Clairambault P (1976) Development of the prosencephalon. In: Llinas R, Precht W (eds) Frog neurobiology. A handbook. Springer, New York, pp 924–945
Conway KM, Gainer H (1987) Immunocytochemical studies of vasotocin, mesotocin, and neurophysins in the Xenopus hypothalamo-neurohypophysial system. J Comp Neurol 64:494–508
Crenshaw BL, De Vries GJ, Yahr PI (1992) Vasopressin innervation of sexually dimorphic structures of the gerbil brain under various hormonal conditions. J Comp Neurol 322:589–598
De Vries GJ, Al-Shamma HA (1990) Sex differences in hormone sensitivity of vasopressin pathways in the rat brain. J Neurobiol 21:686–693
De Vries GJ, Buijs RM, Swaab DF (1981) Ontogeny of the vasopressinergic neurons of the suprachiasmatic nucleus and their extrahypothalamic projections in the rat brain — presence of a sex difference in the lateral septum. Brain Res 218:67–78
De Vries GJ, Best W, Sluiter AA (1983) The influence of androgens on the development of a sex difference in the vasopressinergic innervation of the rat lateral septum. Dev Brain Res 8:377–380
De Vries GJ, Duetz W, Buijs RM, Van Heerikhuize J, Vreeburg JTM (1986) Effects of androgens and estrogens on the vasopressin and oxytocin innervation of the adult rat brain. Brain Res 399:296–302
Di Meglio M, Morrell JI, Pfaff DW (1987) Localization of steroid-concentrating cells in the central nervous system of the frog Rana esculenta. Gen Comp Endocrinol 67:149–154
Diakow C, Nemiroff A (1981) Vasotocin, prostaglandin, and female reproductive behavior in the frog, Rana pipiens. Horm Behav 15:86–93
Dubois-Dauphin M, Tribollet E, Dreifuss JJ (1989) Distribution of neurohypophysial peptides in the guinea pig brain. An immunocytochemical study of the vasopressin-related glycopeptide. Brain Res 496:45–65
Gash D, Sladek C, Scott D (1980) Cytodifferentiation of the supraoptic nucleus correlated with vasopressin synthesis in the rat. Brain Res 181:345–355
Gonzalez A, Smeets WJAJ (1992) Comparative analysis of the vasotocinergic and mesotocinergic cells and fibers in the brain of two amphibians, the anuran Rana ridibunda and the urodele Pleurodeles waltlii. J Comp Neurol 315:53–73
Gorlick DL, Kelley DB (1986) The ontogeny of androgen receptors in the CNS of Xenopus laevis frogs. Dev Brain Res 26:193–200
Gorlick DL, Kelley DB (1987) Neurogenesis in the vocalization pathway of Xenopus laevis. J Comp Neurol 257:614–627
Hofmann MH, Michler A, Meyer DL (1989) Thyroxine influences neuronal connectivity in the adult frog brain. Brain Res 492:389–391
Hsu C-Y, Liang H-M (1970) Sex races of Rana catesbeiana in Taiwan. Herpetologica 26:214–221
Hsu C-Y, Chang L-T, Ku H-H, Lu M-H (1985) In vitro estradiol synthesis and secretion by tadpole ovaries of different developmental stages. Gen Comp Endocrinol 57:393–396
Humason GL (1972) Animal tissue techniques. Freeman, San Francisco
Jokura Y, Urano A (1987) Extrahypothalamic projection of immunoreactive vasotocin fibers in the brain of the toad, Bufo japonicus. Zool Sci 4:675–681
Kalimo H (1975) Ultrastructural studies on the hypothalamic neurosecretory neurons of the rat. III. Paraventricular and supraoptic neurons during lactation and dehydration. Cell Tissue Res 163:151–158
Kar S, Naik DR (1987) Ontogeny of the hypothalamo-neurohypophysial system in the toad, Bufo melanostictus: an immunohistochemical study. Gen Comp Endocrinol 65:184–188
Kicliter E, Ebbesson SOE (1976) Organization of the “nonolfactory” telencephalon. In: Llinas R, Precht W (eds) Frog neurobiology. A Handbook. Springer, New York, pp 946–972
Kollros JJ (1984) Growth and death of cells of the mesencephalic fifth nucleus in Rana pipiens larvae. J Comp Neurol 224:386–394
Krug EC, Honn KV, Battista J, Nicoll CS (1983) Corticosteroids in serum of Rana catesbeiana during development and metamorphosis. Gen Comp Endocrinol 52:232–241
Lopek K (1989) Sex differentiation and early gonadal development in Bombina orientalis (Anura: Discoglossidae). J Morphol 199:299–311
Mayes CR, Watts AG, McQueen JK, Fink G, Charlton HM (1988) Gonadal steroids influence neurophysin II distribution in the forebrain of normal and mutant mice. Neuroscience 25:1013–1022
CcCreery BR, Licht P (1984) The role of androgen in the development of sexual differences in pituitary responsiveness to gonadotropin releasing hormone (GnRH) agonist in the bullfrog, Rana catesbeiana. Gen Comp Endocrinol 54:350–359
Miller MA, Urban JA, Dorsa DM (1989) Steroid dependency of vasopressin neurons in the bed nucleus of the stria terminalis by in situ hybridization. Endocrinology 125:2335–2340
Moore FL (1987) Behavioral actions of neurohypophysial peptides. In: Crews D (ed) Psychobiology of reproductive behavior. An evolutionary perspective. Prentice-Hall, Englewood Cliffs, New Jersey, pp 61–87
Moore FL, Zoeller RT (1979) Endocrine control of amphibian sexual behavior: evidence for a neurohormone-androgen interaction. Horm Behav 13:207–213
Moore FL, Wood RE, Boyd SK (1992) Sex steroids and vasotocin interact in a female amphibian (Taricha granulosa) to elicit female-like egg-laying behavior of male-like courtship. Horm Behav 26:156–166
Neary TJ, Northcutt RG (1983) Nuclear organization of the bullfrog diencephalon. J Comp Neurol 213:262–278
Nieuwkoop PD, Faber J (1956) Normal table of Xenopus laevis (Daudin). North Holland, Amsterdam
Northcutt RG, Kicliter E (1980) Organization of the amphibian telencephalon. In: Ebbesson SOE (ed) Comparative neurology of the telencephalon. Plenum Press, New York, pp 203–255
Panzica G, Viglietti-Panzica C, Sanchez F, Sante P, Balthazart J (1991) Effects of testosterone on a selected neuronal population within the preoptic sexually dimorphic nucleus of the Japanese quail. J Comp Neurol 303:443–456
Penna M, Capranica RR, Somers J (1992) Hormone-induced vocal behavior and midbrain auditory sensitivity in the green treefrog, Hyla cinerea. J Comp Physiol [A] 170:73–82
Polenov AL, Chetverukhin VK (1993) Ultrastructural radioautographic analyysis of neurogenesis in the hypothalamus of the adult frog, Rana temporaria, with special reference to physiological regeneration of the preoptic nucleus. II. Types of neuronal cells produced. Cell Tissue Res 271:351–362
Schmidt RS (1985) Prostaglandin-induced mating call phonotaxis in female American toad: facilitation by progesterone and arginine vasotocin. J Comp Physiol [A] 156:823–829
Shipley MT, Luna J, McLean JH (1989) Processing and analysis of neuroanatomical images. In: Heimer L, Zaborszky I (eds) Neuroanatomical tract tracing methods 2. Plenum Press, New York, pp 331–390
Silverman AJ (1975) The hypothalamic magnocellular neurosecretory system of the guinea pig. II. Immunohistochemical localization of neurophysin and vasopressin in the fetus. Am J Anat 144:445–460
Sinding C, Robinson AG, Seif SM, Schmid PG (1980) Neurohypothyseal peptides in the developing rat fetus. Brain Res 195:177–186
Smoller CG (1966) Ultrastructural studies on the developing neurohypophysis of the Pacific treefrog, Hyla regilla. Gen Comp Endocrinol 7:44–73
Stoll CJ, Voorn P (1985) The distribution of hypothalamic and extrahypothalamic vasotocinergic cells and fibers in the brain of a lizard, Gekko gekko: presence of a sex difference. J Comp Neurol 239:193–204
Subhedar N, Krishna NSR (1990) The response of nucleus preopticus neurosecretory cells to ovarian pressure in the frog, Rana tigrina. Gen Comp Endocrinol 80:438–450
Swanson LW, Sawchenko PE (1983) Hypothalamic integration: organization of the paraventricular and supraoptic nuclei. Ann Rev Neurosci 6:269–324
Swingle WW (1926) The germ cells of anurans. II. An embryological study of sex differentiation in Rana catesbeiana. J Morphol Physiol 41:441–546
Taylor AC, Kollros JJ (1946) Stages in the normal development of Rana pipiens larvae. Anat Rec 94:7–23
Thepen Th, Voorn P, Stoll CJ, Sluiter AA, Pool CW, Lohman AHM (1987) Mesotocin and vasotocin in the brain of the lizard Gekko gekko: an immunocytochemical study. Cell Tissue Res 250:649–656
Tobet A, Zahniser DJ, Baum MJ (1986) Differentiation in male ferrets of a sexually dimorphic nucleus of the preoptic/anterior hypothalamic area requires prenatal estrogen. Neuroendocrinology 44:299–308
Tonon MC, Cuet P, Lamacz M, Jegou S, Cote J, Gouteux L, Ling N, Pelletier G, Vaudry H (1986) Comparative effects of corticotropin-releasing factor, arginine vasopressin, and related neuropeptides on the secretion of ACTH and α-MSH by frog anterior pituitary cells and neurointermediate lobes in vitro. Gen Comp Endocrinol 61:438–445
Van Leeuwen FW, Caffé AR, De Vries GJ (1985) Vasopressin cells in the bed nucleus of the stria terminalis of the rat: sex differences and the influence of androgens. Brain Res 325:391–394
Viglietti-Panzica C, Anselmetti GC, Balthazart J, Aste N, Panzica GC (1992) Vasotocinergic innervation of the septal region in the Japanese quail: sexual differences and the influence of testosterone. Cell Tissue Res 267:261–265
Voorhuis TAM, Kiss JZ, De Kloet ER, De Wied D (1988) Testosterone-sensitive vasotocin-immunoreactive cells and fibers in the canary brain. Brain Res 442:139–146
Wada M, Urano A, Gorbman A (1980) A stereotaxic atlas for diencephalic nuclei of the frog, Rana pipiens. Arch Histol Jap 43:157–173
Watkins WB, Choy VJ (1979) Maturation of the hypothalamo-neurohypophysial system. II. Neurophysin, vasopressin and oxytocin in the median eminence of the developing rat brain. Cell Tissue Res 197:337–346
White BA, Nicoll CS (1981) Hormonal control of amphibian metamorphosis. In: Gilbert LI, Frieden E (eds) Metamorphosis, 2nd edn. Plenum Press, New York, pp 363–396
Whitnall MH, Key S, Ben-Barak Y, Ozato K, Gainer H (1985) Neurophysin in the hypothalamo-neurohypophysial system. II. Immunocytochemical studies of the ontogeny of oxytocinergic and vasopressinergic neurons. J Neurosci 5:98–109
Wilczynski W, Northcutt RG (1983) Connections of the bullfrog striatum: efferent projections. J Comp Neurol 214:333–343
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Boyd, S.K. Development of vasotocin pathways in the bullfrog brain. Cell Tissue Res 276, 593–602 (1994). https://doi.org/10.1007/BF00343958
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00343958