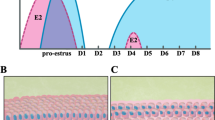
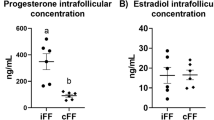
Summary
Angiotensin II (AII) is present in the mammalian ovary and has been correlated with atresia in follicles. Since the theca interna may be one site at which atresia is intiated, we wished to determine whether AII exerts an effect on theca interna from explanted ovarian follicles of hamsters. Hamsters were sacrified on the morning of proestrus, and ovaries were removed. Preovulatory follicles were excised from the ovaries, and cultured with one of the following components: medium alone (control); medium plus AII (1x10-6 M); the AII-receptor antagonist [Sar1, Ile8] AII (1x10-4 M); or AII plus antagonist. After 72 h, the follicles were processed for transmission electron microscopy (to determine quantities of theca interna organelles involved in the steroid synthetic pathway) or for protein determination (to normalize steroid production rates). The incubation medium was drawn off and analyzed by radioimmunoassay for progesterone, androstenedione, or estradiol-17β. There was a significant positive correlation (r=0.92, P<0.01) between follicular androstenedione secretion and area comprising theca interna smooth endoplasmic reticulum. In the theca interna, AII induced a two-fold and 1.6-fold increase in lipid droplet number and area comprising smooth endoplasmic reticulum, respectively (P<0.05). Excess antagonist negated the increase in cell or-ganelles and also reduced androstenedione secretion compared with AII alone (P<0.05). Most importantly, AII significantly augmented the ratio of androstenedione: estradiol-17β secretion by 44% over that of control. The ultrastructural changes observed in this study and the increase in the andostenedione: estradiol-17β production ratio are consistent with atresia-like changes in ovarian follicles. We believe, therefore, that AII is involved, possibly at its membrane receptor, in an aspect of the overall process of follicular atresia, operating in part at the level of the theca interna.
Similar content being viewed by others
References
Anderson B (1978) Regulation of water intake. Physiol Rev 58:582–603
Bejvan SM, Hutz RJ (1987) Effects of estradiol on follicular explants. In: Schwartz N (ed) Proceedings of the eighth annual minisymposium on reproductive biology. Northwestern University Press, Chicago, p 11
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principles of protein-dye binding. Anal Biochem 72:248–254
Bumpus FM, Pucell AG, Daud AI, Husain A (1988) AII: an intraovarian regulatory peptide. Am J Med Sci 31:406–408
Carson RS, Findlay JK, Clark JJ, Burger HG (1981) Estradiol-17β, testosterone, and androstenedione in ovine follicular fluid during growth and atresia of ovarian follicles. Biol Repord 24:105–113
Christensen AK, Gillium SW (1969) The correlation of fine structure and function in steroid-secreting cells, with emphasis on the gonads. In: McKerns KW (ed) The gonads. Appleton-Century-Crofts, New York, pp 415–488
Crisp TM, Denys FR (1975) The fine structure of the rat granulosa cell cultures correlated with progestin secretion. In: Hess M (ed) Electron microscopic concepts of secretion. Wiley and Sons, New York, pp 3–34
Daud AI, Bumpus FM, Husain A (1988) Evidence for selective expression of angiotensin II receptors on atretic follicles in the rat ovary: an autoradiographic study. Endocrinology 122:2727–2734
Everett JW (1947) Hormonal factors responsible for deposition of cholesterol in the corpus lutem of the rat. Endocrinology 41:364–377
Farookhi R (1980) Effects of androgen on induction of gonadotropin receptors and gonadotropin-stimulated adenosine 3′, 5′-monophosphate production in rat ovarian granulosa cells. Endocrinology 106:106–1216–1223
Glorioso N, Atlas SA, Laragh JH, Jewelewicz R, Sealy JE (1986) Prorenin in high concentrations in human follicular fluid. Science 233:1422–1424
Hillier SG, Zeleznik AJ, Knazek C, Ross GT (1980) Hormonal regulation of preovulatory follicle maturation in the rat. J Reprod Fert 60:219–229
Husain A, Bumpus FM, DeSilva P, Speth RC (1987) Localization of angiotensin II receptors in ovarian follicles and the identification of angiotensin II in rat ovaries. Proc Natl Acad USA 84:2489–2493
Hutz RJ, Schaller M, Kitzman PH, Bejvan SM (1991) Oestradiol-17β affects differentially viability, progesterone secretion, and apical surface morphology of hamster ovarian follicles in vitro. Zool Sci 8:81–88
Kramer RE, Gallant S, Brownie AC (1980) Actions of angiotensin II on aldosterone biosynthesis in the rat adrenal cortex. J Biol Chem 255:3442–3447
Laragh JH, Angers M, Kelly WG, Lieberman S (1960) Hypertensive agents and pressor substances: the effects of epinephrine, norepinephrine, angiotensin II, and others on the secretory rate of aldosterone in man. J Am Assoc 174:234–240
Millonig G (1961) Advantages of a phosphate buffer for OsO4 solutions in fixation. J Appl Phys 32:1637–1640
Moses HL, Davis WW, Rosenthal AS, Garren LD (1969) Adrenal cholesterol: localization by electronmicroscopic autoradiography. Science 163:1203–1205
Nussdorfer GG, Mazzochi G, Meneghelli V (1978) Cytophysiology of the adrenal zona fasciculata. Int Rev Cytol 55:291–365
Nussdorfer GG, Robba C, Mazzochi G, Rebuffat P (1981) Effects of angiotensin II on the zona fasciculata of the rat adrenal cortex: An ultrastructural stereologic study. J Anat 132:235–242
Pucell AG, Bumpus FM, Husain A (1987) Rat ovarian angiotensin II receptors. Characterization and coupling to estrogen secretion. J Biol Chem 262:7076–7080
Pucell A, Bumpus FM, Husain A (1988) Regulation of angiotensin II receptors in cultured rat ovarian granulosa cells by FSH and angiotensin II. J Biol Chem 263:11954–11961
Reynolds ES (1963) The use of lead citrate at high pH as an electron opaque stain in electron microscopy. J Cell Biol 17:208–211
Schultz D, Brunswig B, Mukhopadhyay A (1989) Renin and renin-like activities in bovine ovarian follicles. Endocrinology 124:1389–1398
Silavin SL, Greenwald GS (1984) Steroid production by isolated theca and granulosa cells after initiation of atresia in the hamster. J Reprod Fert 71:387–392
Speth RC, Husain A (1988) Distribution of angiotensin-converting enzyme and angiotensin II-receptor binding sites in the rat ovary. Biol Reprod 38:695–702
Spurr AR (1969) A low-viscosity epoxy resin embedding medium for electron microscopy. J Ultrastruct Res 26:31–43
Tamaoki B (1973) Steroidogenesis and cell structure: Biochemical pursuit of sites of steroid biosynthesis. J Steroid Biochem 4:89–118
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kitzman, P.H., Hutz, R.J. In-vitro effects of angiotensin II on steroid production by hamster ovarian follicles and on ultrastructure of the theca interna. Cell Tissue Res 268, 191–196 (1992). https://doi.org/10.1007/BF00338068
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00338068