Abstract
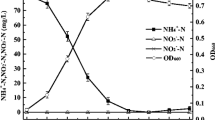
Nitrate and nitrite was reduced by Escherichia coli E4 in a l-lactate (5 mM) limited culture in a chemostat operated at dissolved oxygen concentrations corresponding to 90–100% air saturation. Nitrate reductase and nitrite reductase activity was regulated by the growth rate, and oxygen and nitrate concentrations. At a low growth rate (0.11 h−1) nitrate and nitrite reductase activities of 200 nmol · mg−1 protein · min−1 and 250 nmol · mg−1 protein · min−1 were measured, respectively. At a high growth rate (0.55 h−1) both enzyme activities were considerably lower (25 and 12 nmol mg−1 · protein · min−1). The steady state nitrite concentration in the chemostat was controlled by the combined action of the nitrate and nitrite reductase. Both nitrate and nitrite reductase activity were inversely proportional to the growth rate. The nitrite reductase activity decreased faster with growth rate than the nitrate reductase. The chemostat biomass concentration of E. coli E4, with ammonium either solely or combined with nitrate as a source of nitrogen, remained constant throughout all growth rates and was not affected by nitrite concentrations. Contrary to batch, E. coli E4 was able to grow in continuous cultures on nitrate as the sole source of nitrogen. When cultivated with nitrate as the sole source of nitrogen the chemostat biomass concentration is related to the activity of nitrate and nitrite reductase and hence, inversely proportional to growth rate.
Similar content being viewed by others
References
Abou-JaoudéA, PascalMC, ChippauxM (1979) Formate-nitrite reduction in Escherichia coli K-12. 2. Identification of components involved in electron transfer. Eur J Biochem 95:315–321
BucttnerMJ, SpitzE, RickenbergHV (1973) Cyclic adenosine 3′,5′-monophosphate in Escherichia coli. J Bacteriol 114:1068–1073
Deutsche Einheitsverfahren zur Wasseruntersuchung (1989) Band I. VCH, Weinheim, FRG
ColeJA (1968) Cytochrome c 552 and nitrite reduction in Escherichia coli. Biochim Biophys Acta 162:356–368
ColeJA, BrowneCM (1980) Nitrite reduction to ammonia by fermentative bacteria: a short circuit in the biological nitrogen cycle. FEMS Microbiol Lett 7:65–72
DeMossRD, BardRC (1957) Manual of microbiological methods. MacGraw-Hill, New York, pp 169–198
DunnGM, HerbertRA, BrownCM (1979) Influence of oxygen tension on nitrate reduction by a Klebsiella sp. growing in chemostat culture. J Gen Microbiol 112:379–383
GiordanoG, RossetR, AzoulayE (1977) Isolation and study of mutants of Escherichia coli K12 that are sensitive to chlorate and derepressed for nitrate reductase. FEMS Microbiol Lett 2:21–26
GriffithsL, ColeJA (1987) Lack of redox control of the anaero-bically-induced nirB + gene of Escherichia coli K-12. Arch Microbiol 147:364–369
HackettCS, MacGregorCH (1981) Synthesis and degradation of nitrate reductase in Escherichia coli. J Bacteriol 146:352–359
HarderW, DijkhuizenL (1983) Physiological responses to nutrient limitation. Annu Rev Microbiol 37:1–23
IngledewWJ, PooleRK (1984) The respiratory chains of Escherichia coli. Microbiol Rev 48:222–271
JacksonRH, Cornish-BowdenA, ColeJA (1981) Prosthetic groups of the NADH-dependent nitrite reductase from Escherichia coli K-12. Biochem J 193:861–867
JohnP (1977) Aerobic and anaerobic bacterial respiration monitored by electrodes. J Gen Microbiol 98:231–238
KobayashiM, IshimotoM (1973) Aerobic inhibition of nitrate assimilation in Escherichia coli. Z Allg Microbiol 13:405–413
KraprálekF, JechováE, OtavováM (1982) Two sites of oxygen control in induced synthesis of respiratory nitrate reductase in Escherichia coli. J Bacteriol 149:1142–1145
KrulJM, VeeningenR (1977) The synthesis of the dissimilatory nitrate reductase under aerobic conditions in a number of denitrifying bacteria isolated from activated sludge and drinking water. Water Res 11:39–43
LinECC, KuritzkesDR (1987) Pathways for anaerobic electron transport. In: NeidhardtFC, IngrahamJL, Brooks LowK, MagasanikB, SchaechterM, UmbargerHE (eds) Escherichia coli and Salmonella typhimurium, vol 1. American Society for Microbiology, Washington, DC, pp 210–221
LiuMC, PeckHDJr, Abou-JaoudéA, ChippauxM, LeGallJ (1981) A reappraisal of the role of the low potential c-type cytochrome (cytochrome c 552) in NADH-dependent nitrite reduction and its relationship with a co-purified NADH-oxidase in Escherichia coli K-12. FEMS Microbiol Lett 10:333–337
MatinA (1981) Regulation of enzyme synthesis as studied in continuous culture. In: CalcottPH (ed) Continuous culture of cells, vol II. CRC Press, Boca Raton, Florida, pp 69–97
MeibergJBM, BruinenbergPM, HarderW (1980) Effect of dissolved oxygen tension on the metabolism of methylated amines in Hyphomicrobium X in the absence and presence of nitrate: evidence for “aerobic” denitrification. J Gen Microbiol 120: 453–463
MonodJ (1950) La technique de la culture continue: théorie et applications. Ann Inst Pasteur 79:390–410
NishimuraY, TanIKP, OhgamiY, KohgamiK, KamiharaT (1983) Induction of membrane-bound l-lactate dehydrogenase in Escherichia coli under conditions of nitrate respiration, fumarate reduction and trimethyl-N-oxide reduction. FEMS Microbiol Lett 17:283–286
NojiS, TaniguchiS (1987) Molecular oxygen controls nitrate transport of Escherichia coli nitrate-respiring cells. J Biol Chem 262:9441–9443
PecherA, ZinoniF, JatisatienrC, WirthR, HenneckeH, BöckA (1983) On the redox control of synthesis of anaerobically induced enzymes in enterobacteriaceae. Arch Microbiol 136: 131–136
PopeNR, ColeJA (1982) Generation of a membrane potential by one of the two independent pathways for nitrite reduction by Escherichia coli. J Gen Microbiol 128:219–222
RehrB, KlemmeJH (1986) Metabolic role and properties of nitrite reductase of nitrate-ammonifying marine Vibrio species. FEMS Microbiol Lett 35:325–328
RobertsonLA, KuenenJG (1984) Aerobic denitrification: a controversy revived. Arch Microbiol 139:351–354
RutgersM, Teixeira de MattosMJ, PostmaPW, VanDamK (1987) Establishment of the steady state in glucose-limited chemostat cultures of Klebsiella pneumoniae. J Gen Microbiol 133:445–451
ShoweMK, DeMossJA (1968) Localization and regulation of synthesis of nitrate reductase in Escherichia coli. J Bacteriol 95:1305–1313
StewartV (1988) Nitrate respiration in relation to facultative metabolism in enterobacteria. Microbiol Rev 52:190–232
StouthamerAH (1988) Dissimilatory reduction of oxidized nitrogen compounds. In: ZehnderAJB (ed) Biology of anaerobic microorganisms. John Wiley & Sons, New York, pp 245–303
StrandSE, McDonellAJ, UnzRF (1988) Oxygen and nitrate reduction kinetics of a nonflocculating strain of Zoogloea ramigera. Antonie Leeuwenhoek 54:245–255
UndenG, DucheneA (1987) On the role of cyclic AMP and the Fnr protein in Escherichia coli growing anaerobically. Arch Microbiol 147:195–200
ZehnderAJB, SvenssonBH (1986) Life without oxygen: what can and what cannot? Experientia 42:1197–1205
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Brons, H.J., Zehnder, A.J.B. Aerobic nitrate and nitrite reduction in continuous cultures of Escherichia coli E4. Arch. Microbiol. 153, 531–536 (1990). https://doi.org/10.1007/BF00245261
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00245261