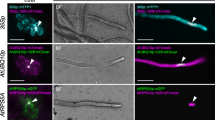
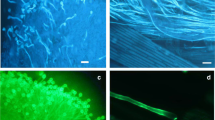
Abstract
The promoter of a rice pollen-specific gene, PS1, has been fused to the Bacillus amyloliquefaciens Barnase gene which encodes a secreted ribonuclease. The PS1-Barnase chimeric gene has been introduced into tobacco. These transgenic tobacco plants show normal vegetative and floral development, but they display a range of reproductive properties from slightly reduced in fertility to completely sterile. Barnase mRNAs are detectable in the pollen from transgenic plants which do not show an obvious fertility-related phenotype, and in a few plants which have a mildly reduced-fertile phenotype. However, transgenic plants with a severely reduced-fertile or sterile phenotype do not accumulate detectable amounts of Barnase mRNA in their pollen, and the quality of their RNA is poor, presumably because of extensive RNA degradation. Reciprocal crosses between these transgenic plants and wild-type controls showed that the reduced-fertile phenotype is associated only with the transgenic pollen. When used as the female parent, these PS1-Barnase transgenic plants are fully fertile. Anthers in the severely sterile transgenic plants develop normally, but the majority of their pollen grains have abnormal morphology and they fail to germinate. These results indicate that expression of a pollen-specific cytotoxic gene induces lethality in pollen and may lead to severely reduced male fertility.
Similar content being viewed by others
References
Block M de, Debrouwer D (1993) Engineered fertility control in transgenic Brassica napus L.: histochemical analysis of anther development. Planta 189:218–225
Cheung AY (1995) Pollen-pistil interactions in compatible pollination. Proc Natl Acad Sci USA 92:3077–3080
Church G, Gilbert W (1984) Genomic sequencing. Proc Natl Acad Sci USA 81:1991–1995
Delebrese R, Reynaert A, Hofte H, Hernalsteen J-P, Leemans J, Van Mantagu M (1986) Vectors for cloning in plant cells. Methods Enzymol 153:277–290
Dixon RA (1985) Plant cell culture, a practical approach. IRL Press, Washington DC
Gasser CS (1991) Molecular studies on the differentiation of floral organs. Annu Rev Plant Physiol Plant Mol Biol 42:621–649
Goldberg RB, Beals TP, Sanders PM (1993) Anther development: basic principles and practical applications. Plant Cell 5:1217–1229
Gu Q, Kawata EE, Morse MJ, Wu H-M, Cheung AY (1992) A flower-specific cDNA encoding a novel thionin in tobacco. Mol Gen Genet 234:89–96
Hanson MR, Conde MF (1985) Functioning and variation of cytoplasmic genomes: lessons from cytoplasmic-nuclear interactions affecting male fertility in plants. Int Rev Cytol 94:213–267
Hanson DD, Hamilton DA, Travis JL, Bashe DM, Mascarenhas JP (1989) Characterization of a pollen-specific cDNA clone from Zea mays and its expression. Plant Cell 1:173–179
Hartley RW (1988) Barnase and barstar. Expression of its cloned inhibitor permits expression of a cloned ribonuclease. J Mol Biol 202:913–915
Hartley RW (1989) Barnase and barstar: two small proteins to fold and fit together. Trends Biochem Sci 14:450–454
Jahnen W, Lush VM, Clarke AE (1989) Inhibition of in vitro pollen tube growth by isolated S-glycoproteins of Nicotiana alata. Plant Cell 1:501–510
Jefferies CJ, Belcher AR (1974) A fluorescent brightener used for pollen tube identification in vivo. Stain Technol 49:199–202
Kamalay JC, Goldberg RB (1980) Regulation of structural gene expression in tobacco. Cell 19:934–946
Kamalay JC, Goldberg RB (1984) Organ-specific nuclear RNAs in tobacco. Proc Natl Acad Sci USA 81:2801–2805
Knox RB (1984) Pollen-pistil interactions. Encycl Plant Physiol 17:508–608
Koltunow AM, Truettner J, Cox KH, Wallroh M, Goldberg RB (1990) Differential temporal and spatial gene expression patterns occur during anther development. Plant Cell 2:1201–1224
Lonsdale DM (1987) Cytoplasmic male sterility: a molecular perspective. Plant Physiol Biochem 25:265–271
Mariani C, De Beuckeleer M, Truettner J, Leemans J, Goldberg RB (1990) Induction of male sterility in plants by a chimaeric ribonuclease gene. Nature 347:737–741
Mariani C, Gossele V, Beuckeleer M, De Block M, Goldberg RB, Greef W, Leeman J (1992) A chimaeric ribonuclease inhibitor gene restores fertility to male sterile plants. Nature 357:384–387
Mascarenhas JP (1975) The biochemistry of angiosperm pollen development. Bot Rev 41:259–314
Mascarenhas JP (1989) The male gametophyte of flowering plants. Plant Cell 1:657–664
Mascarenhas JP (1990) Gene activity during pollen development. Annu Rev Plant Physiol 41:317–338
Mascarenhas JP (1993) Molecular mechanisms of pollen tube growth and differentiation. Plant Cell 5:1303–1314
McCormick S (1993) Male gametophyte development. Plant Cell 5:1265–1275
Muschietti J, Dircks L, Vancanneyt G, McCormick S (1994) LAT52 protein is essential for tomato pollen development: pollen expressing antisense LAT52 RNA hydrates and germinates abnormally and cannot achieve fertilization. Plant J 6:321–338
Mulcahy DL (1971) A correlation between gametophytic and sporophytic characteristics in Zea mays L. Science 171:1155–1156
Mulcahy DL (1974) Correlation between speed of pollen tube growth and seedling height in Zea mays L. Nature 249:491–492
Mulcahy DL, Mulcahy GB (1975) The influence of gametophytic competition on sporophytic quality in Dianthus chinensis. Theor Appl Genet 46:277–280
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY
Takegami MH, Yoshioka M, Tanaka I, Ito M (1981) Characteristics of isolated microsporocytes from liliaceous plants for studies of the meiotic cell cycle in vitro. Plant Cell Physiol 22:1–10
Tanaka I, Ito M (1980) Induction of typical cell division in isolated microspores of Lilium longiflorum. Plant Sci Lett 17:279–285
Tanaka I, Ito M (1981) Studies on microspore development in liliaceous plants. II. Pollen tube development in lily pollens cultured from the uninucleate microspore stage. Plant Cell Physiol 22:149–153
Twell D, Wing R, Yamaguchi J, McCormick S (1989) Isolation and expression of an anther-specific gene from tomato. Mol Gen Genet 217:240–245
Willing RP, Mascarenhas JP (1984) Analysis of the complexity and diversity of mRNAs from pollen and shoots of Tradescantia. Plant Physiol 75:865–868
Willing RP, Bashe D, Mascarenhas JP (1988) An analysis of the quantity and diversity of messenger RNAs from pollen and shoots of Zea mays. Theor Appl Genet 75:751–753
Xu H, Knox RB, Taylor PE, Singh MB (1995) Bcpl, a gene required for male fertility in Arabidopsis. Proc Natl Acad Sci USA 92:2106–2110
Zou J-T, Zhan X-Y, Wu H-M, Wang H, Cheung AY (1994) Characterization of a rice pollen-specific gene and its expression. Am J Bot 81:552–561
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhan, Xy., Wu, Hm. & Cheung, A.Y. Nuclear male sterility induced by pollen-specific expression of a ribonuclease. Sexual Plant Reprod 9, 35–43 (1996). https://doi.org/10.1007/BF00230364
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00230364