Abstract
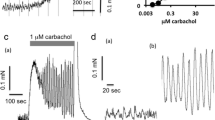
The alkylating agent isothiocyanatobenzyl imidazoline (IBI) was synthesized to investigate the unique receptor interacting properties of imidazolines. On the isolated rabbit iris sphincter, IBI produced concentration-dependent responses with an EC50 of 18 μmol/l, and at the highest concentration tested the maximum contraction of the tissue was 50% of the carbachol maximum. At equiactive concentrations with the similar washing procedure, the total duration of responses to IBI and carbachol was 24 and 3 min, respectively. After repeated washing, the sphincter relaxes to the control baseline of tone but, after reexposure to IBI for 6 h, failed to contract, indicating that desensitization or irreversible block has developed. Unlike with carbachol, the sphincter contraction to IBI was not affected by atropine I μmol/l, indomethacin 1 μmol/l, verapamil 10 μmol/l, or nifedipine 10 μmol/l. At a higher concentration of nifedipine and papaverine 100 μmol/l, the response to IBI was blocked. Furthermore, the contractile response to IBI was abolished by Ca++ removal from the medium. Under similar conditions, 26 ± 8% of the maximum response to carbachol was preserved. Thus influx of extracellular as well as rise in intracelluar Ca++ appears vital for the contractile response to IBI.
IBI did not contract the iris dilator, but shifted the concentration-response curve to the α-adrenoceptor activator, phenylephrine, to the right with a reduction in the maximum response. The tissue failed to regain the sensitivity to phenylephrine after 6 h of repeated washing. Phentolamine and nifedipine provided a small but significant protection of the response to phenylephrine against the irreversible block by IBI. Based on chemical and pharmacological properties of IBI, it is concluded that the molecule acts in the rabbit as an irreversible agonist on unidentified receptors of the iris sphincter and an irreversible antagonist of multiple receptors on the iris dilator. These molecular properties of IBI are clearly different from that of the parent imidazoline molecule tolazoline.
Similar content being viewed by others
References
Brasili L, Giannella M, Melchiorre C, Belleau B, Benfey BG (1981) Structural requirements for adrenergic α-receptor covalent occupancy by chiral tetramine disulfides. Eur J Med Chem 16: 115–118
Decker N, Leclerc G, Quennedey MC, Rouot B, Schwartz J (1983) Study of two alkylating derivatives: The p-isothiocyanato- and the p-methylisothiocyanato-clonidine. Eur J Pharmacol 93: 205–211
Furchgott RF (1966) The use of b-haloalkylamines in the differentiation of receptors and in the determination of dissociation constants of receptor- agonists complexes. In: Harper NJ, Simmonds AB (eds) Advances in drug research. Academic Press, London New York, pp 21–55
Furchgott RF (1967) The pharmacological differentiation of adrenergic receptors. Ann NY Acad Sci 139:553–570
Kern R (1970) Die adrenergischen Receptoren der intraoculären Muskeln des Menschen. Graefes Arch Klin Exp Ophthalmol 180:231–248
Konno F, Takayanagi I (1986) Characterization of postsynaptic alpha1-adrenoceptors in the rabbit iris dilator smooth muscles. Naunyn-Schmiedeberg's Arch Pharmacol 333:271–276
Leclerc G, Rouot B, Schwartz J, Velly J, Wermuth CG (1980) Studies on some para-substituted clonidine derivatives that exhibit an alpha-adrenoceptor stimulant activity. Br J Pharmacol 71:5–9
Lei L, Slavica M, Ernsberger P, Graves ME, Patil P, Miller D, Feller D (1995) Calcium channel-dependent and I1-imidazoline receptor binding properties of 2-(4′-isothiocyanatobenzyl) imidazoline analogs in vascular and brain tissues. Ann NY Acad Sci 763: 283–286
Lippert B, Belleau B (1973) Recent advances in the active site chemistry of the adrenergic alpha-receptor. In: Usdin E, Snyder SH (eds) Frontiers in catecholamine research. Pergamon Press, New York, pp 369–371
Nickerson M (1949) The pharmacology of adrenergic blockade. Pharmacol Rev 1:27–101
Patil PN, LaPidus JB, Tye A (1970) Steric aspects of adrenergic drugs. J Pharm Sci 59:1205–1234
Patil PN, Feller DR, Miller DD (1991) Molecular aspects of the receptor activation by imidazolines: an overview. J Neural Transm [Suppl] 34:187–194
Ruffolo RR Jr, Yaden EL, Waddell JE, Ward JS (1982a) Receptor interactions of imidazolines XI alpha-adrenergic and antihypertensive effects of clonidine and its methylene bridged analog, St. 913. Pharmacology 25:187–201
Ruffolo RR Jr, Yaden EL, Ward JS (1982b) Receptor interactions of imidazolines. Influence of ionization constant on the diffusion of clonidine and a series of structurally related imidazolines into and out of the central nervous system. Eur J Pharmacol 81: 367–375
Slavica M, Lei L, Patil PN, Kerezy A, Feller DR, Miller DD (1994) Synthesis and biological activities of a new set of irreversibly acting 2-(4′-isothiocyanatobenzyl) imidazoline analogs in rat thoracic aorta. J Med Chem 37:1874–1881
Stryer L (1981) Introduction to protein structure and function. In: Stryer L (ed) Biochemistry. Freeman, San Francisco, pp 11–38
Takayanagi I, Kaneko M, Hisayama T (1983) Mechanical responses and calcium movements in rabbit iris smooth muscles. Jpn J Ophthalmol 27:575–584
Triggle DJ (1965) 2-Halogenoethylamines and receptor analysis. In: Harper NJ, Simmonds AB (eds) Advances in drug research. Academic Press, London New York, pp 173–189
Venkataraman BV, Hamada A, Shams G, Miller DD, Feller DR, Patil PN (1989) Paradoxical effects of isothiocyanate analog of tolazoline on rat aorta and human platelets. Blood Vessels 26:335–346
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ishikawa, H., Miller, D.D. & Patil, P.N. Irreversible agonist and antagonist properties of isothiocyanatobenzyl imidazoline in albino rabbit iris muscles. Naunyn-Schmiedeberg's Arch Pharmacol 354, 362–368 (1996). https://doi.org/10.1007/BF00171069
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00171069