Summary
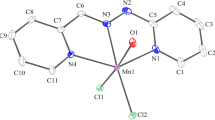
New complexes of the general formulae [M(LH2)2Cl2] (M = Mn, Fe, Co, Ni, Cu, Zn), [Mn(LH2)2X2] (X = Br, I), [Cu(LH2)2Br2], [Ni(LH2)2X2] (X = Br, NCS, ONO2), [Cu(LH2)X2]n (X = Cl, Br), K2[NiL2]·2H2O and K2-[CuL2]·H2O, where LH2 = malonamide, were isolated. The complexes were characterized by elemental analyses, X-ray powder patterns, magnetic susceptibilities and spectroscopic (variable-temperature 57Fe-Mössbauer, e.s.r., u.v.-vis., i.r., far-i.r., Raman) studies. Monomeric trans pseudo-octahedral stereochemistries for the neutral 1∶2 complexes and square planar structures of D 2h symmetry for the two ionic complexes are assigned in the solid state. Dimeric or polymeric five-coordinate structures are proposed for the 1∶1 copper(II) compounds. LH2 and L2− behave as bidentate chelating ligands binding through oxygen and deprotonated nitrogen atoms, respectively. A detailed comparison of the studied complexes with the corresponding oxamide complexes is also presented.
Similar content being viewed by others
References
J. Soto, R. Martinez-Màñez, J. Payà, F. Lloret and M. Julve, Transition Met. Chem., 18, 69 (1993) and references therein.
K. E. Gilmore and G. K. Pagenkoph, Inorg. Chem., 24, 2436 (1985).
O. Kahn, Y. Pei, K. Nakatani and Y. Journaux, New J. Chem., 16, 269 (1992) and references therein.
J. A. Real, M. Mollar, R. Ruiz, J. Faus, F. Lloret, M. Julve and M. Philoche-Levisalles, J. Chem. Soc., Dalton Trans., 1483 (1993) and references therein.
Y. Pei, O. Kahn, K. Nakatani, E. Codjovi, C. Mathoniére and J. Sletten, J. Am. Chem. Soc., 113, 6558 (1991) and references therein.
J. J. Bour, P. J. M. W. L. Birker and J. J. Steggerda, Inorg. Chem., 10, 1202 (1971).
H. O. Desseyn, B. J. Van der Veken and M. A. Herman, Spectrochim. Acta, 33A, 633 (1977).
H. O. Desseyn, W. J. Van Riel and B. J. Van der Veken, Can. J. Spectrosc., 24, 98 (1979).
F. Quaeyhaegens, H. O. Desseyn, B. Bracke and A. T. H. Lenstra, J. Mol. Struct., 238, 139 (1990).
H. O. Desseyn in S. Patai (Ed.), The Chemistry of Acid Derivatives, Wiley, New York, 1992, Vol. 2, Chap. 7, pp. 271–303.
H. O. Desseyn, Pure Appl. Chem., 61, 867 (1989).
H. O. Desseyn, W. Van Riel, L. Van Haverbeke and A. Goeminne, Transition Met. Chem., 5, 88 (1980).
G. Schoeters and H. O. Desseyn, Transition Met. Chem., 6, 305 (1981).
H. O. Desseyn and G. Schoeters, Bull. Soc. Chim. Belg., 95, 13 (1986).
F. Quaeyhaegens, S. P. Perlepes and H. O. Desseyn, Spectrochim. Acta, 43A, 917 (1987).
F. J. Quaeyhaegens, S. P. Perlepes and H. O. Desseyn, Spectrochim. Acta, 45A, 809 (1989).
S. P. Perlepes, F. Quaeyhaegens and H. O. Desseyn, Bull. Soc. Chim. Belg., 98, 153 (1989).
S. P. Perlepes, F. J. Quaeyhaegens and H. O. Desseyn, Transition Met. Chem., 15, 132 (1990).
F. J. Quaeyhaegens, H. O. Desseyn, S. P. Perlepes, J. C. Plakatouras, B. Bracke and A. T. H. Lenstra, Transition Met. Chem., 16, 92 (1991).
I. I. Kalinichenko, N. M. Titov and L. A. Pecherskikh, Russ. J. Inorg. Chem., 34, 1280 (1989) and references therein.
L. S. Tikhonova and A. I. Stetsenko, Russ. J. Inorg. Chem., 38, 90 (1993).
J. C. Plakatouras, S. P. Perlepes, D. Mentzafos, A. Terzis, T. Bakas and V. Papaefthymiou, Polyhedron, 11, 2657 (1992).
H. Sigel and R. B. Martin, Chem. Rev., 82, 385 (1982).
F. A. Cotton and G. Wilkinson, Advanced Inorganic Chemistry, 5th Edit., Wiley, New York, 1988, pp. 702, 716, 729–731, 744, 745, 766–772.
S. De Beukeleer, H. O. Desseyn and S. P. Perlepes, submitted for publication in Thermochim. Acta.
B. F. Little and G. J. Long, Inorg. Chem., 17, 3401 (1978) and references therein.
P. N. Hawker and M. V. Twigg in G. Wilkinson, R. D. Gillard and J. A. McCleverty (Eds), Comprehensive Coordination Chemistry, Pergamon Press, Oxford, 1987, Chap. 44, pp. 1210–1211.
N. N. Greenwood and T. C. Gibb, Mössbauer Spectroscopy, Chapman and Hall, London, 1971.
R. Ingalls, Phys. Rev., 133A, 787 (1964).
P. Illiopoulos and K. S. Murray, J. Chem. Soc., Dalton Trans., 433 (1988).
B. J. Hathaway, Coord. Chem. Rev., 35, 211 (1981).
B. J. Hathaway, J. Chem. Soc., Dalton Trans., 1196 (1972).
D. W. Smith, J. Chem. Soc. (A), 3108 (1970).
W. E. Estes, D. P. Gavel, W. E. Hatfield and D. J. Hodgson, Inorg. Chem., 17, 1415 (1978).
A. B. P. Lever, Inorganic Electronic Spectroscopy, 2nd Edit., Elsevier, Amsterdam, 1984, pp. 355, 356, 448–451, 458–72, 480–505, 507–544, 554–572.
R. S. Drago, Physical Methods in Chemistry, Saunders, Philadelphia, 1977, pp. 359–395, 647–649.
S. De Beukeleer and H. O. Desseyn, submitted for publication in Spectrochim. Acta.
P. X. Armendarez and K. Nakamoto, Inorg. Chem., 5, 796 (1966).
K. Nakamoto, Infrared and Raman Spectra of Inorganic and Coordination Compounds, 3rd Edit., Wiley, New York, 1977, pp. 231, 244–247, 270–274, 317–324.
L. S. Gelfand, F. J. Iaconianni, L. L. Pytlewski, A. N. Speca, C. M. Mikulski and N. M. Karayannis, J. Inorg. Nucl. Chem., 42, 377 (1980).
S. P. Perlepes, P. Jacobs, H. O. Desseyn and J. M. Tsangaris, Spectrochim. Acta, 43A, 1007 (1987).
G. J. Kleywegt, W. G. R. Wiesmeijer, G. Van Driel, W. L. Driessen, J. Reedijk and J. H. Noordik, J. Chem. Soc., Dalton Trans., 2177 (1985).
U. Casellato, P. A. Vigato and M. Vidali, Coord. Chem. Rev., 36, 183 (1981).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
De Beukeleer, S., Desseyn, H.O., Perlepes, S.P. et al. Transition metal complexes with neutral and deprotonated malonamide. Transition Met Chem 19, 468–476 (1994). https://doi.org/10.1007/BF00139331
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00139331