Abstract
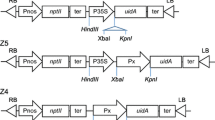
We monitored, for the first time, the activity of two model heterologous promoters, the Agrobacterium rhizogenes rolC and the cauliflower mosaic virus (CaMV) 35S, throughout the annual cycle of growth and dormancy in a perennial species, hybrid aspen. Each promoter was fused to the uidA β-glucuronidase (GUS) reporter gene and the constructs were introduced into the hybrid aspen genome by Agrobacterium-mediated transformation. Both wildtype and transgenic plants were cultivated under different regimes of photoperiod and temperature to induce passage through one growth-dormancy-reactivation cycle, and at intervals GUS staining was assessed in stem sections. In rolC::uidA transformants, GUS activity in rapidly growing current-year shoots was not only tissue-specific, being localized to the phloem, but also cell-specific at the shoot base, where it was present only in the companion cells. However, during the onset of dormancy induced by short photoperiod, GUS activity shifted laterally from the phloem to include the cortex and pith. After subsequent exposure to chilling temperatures to induce the transition between the dormancy stages of rest and quiescence, GUS activity almost disappeared from all stem tissues, but regained its original phloem specificity and intensity after the shoots were reactivated by exposing them to long photoperiod and high temperatures. In contrast, GUS activity in the stem of 35S::uidA transformants was strong in all tissues except for the vascular cambium and xylem, and did not vary in intensity during the growth-dormancy-reactivation cycle. The lateral shift and increased intensity of GUS activity in the stem of rolC::uidA transformants during dormancy induction was shown to be associated with the accumulation of starch, and to be mimicked by incubating stem sections in sucrose, as well as glucose and fructose, but not sorbitol, prior to the GUS assay. Our results demonstrate that the activities of the rolC and 35S promoters varied in very different, unpredictable ways during the annual cycle of growth and dormancy in a perennial species, and indicate that the spatial and temporal variation in rolC promoter activity that we observed in the stem of transgenic hybrid aspen plants is attributable to cellular and seasonal changes in sucrose content.
Similar content being viewed by others
References
Battraw MJ, Hall TC: Histochemical analysis of CaMV 35S promoter-β-glucuronidase gene expression in transgenic rice plants. Plant Mol Biol 15: 527–538 (1990).
Benfey PN, Chua N-H: Regulated genes in transgenic plants. Science 244: 174–181 (1989).
Benfey PN, Ren L, Chua N-H: The CaMV 35S enhancer contains at least two domains which can confer different developmental and tissue-specific expression patterns. EMBO J 8: 2195–2202 (1989).
Catesson A-M: Cambial ultrastructure and biochemistry: changes in relation to vascular tissue differentiation and the seasonal cycle. Int J Plant Sci 155: 251–261 (1994).
Essau K: Anatomy of Seed Plants. John Wiley, New York (1977).
Geigenberger P, Langenberger S, Wilke I, Heineke D, Heldt HW, Stitt M: Sucrose is metabolised by sucrose synthase and glycolysis within the phloem complex of Ricinus communis L. seedlings. Planta 190: 446–453 (1993).
Ingestad T: A definition of optimum nutrient requirements in birch seedlings. I. Physiol Plant 23: 1127–1138 (1970).
Jefferson RA, Kavanagh TA, Bevan MW: GUS fusions: β-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 6: 3901–3907 (1987).
Jouanin L, Brasileiro ACM, Leplé JC, Pilate G, Cornu D: Genetic transformation: a short review of methods and their applications, results and perspectives for forest trees. Ann Sci For 50: 325–336 (1993).
Kozlowski TT: Carbohydrate sources and sinks in woody plants. Bot Rev 58: 107–222 (1992).
Lang GA, Early JD, Arroyave NJ, Darnell RL, Martin GC, Stutte GW: Dormancy: toward a reduced, universal terminology. HortScience 20: 809–812 (1985).
Lavender DP: Measuring phenology and dormancy. In: Lassoie JP, Hinckley TM (eds) Techniques and Approaches in Forest Tree Ecophysiology, pp. 403–422. CRC Press, Boca Raton, FL (1991).
Little CHA, Pharis RP: Hormonal control of radial and longitudinal growth in the tree stem. In: Gartner BL (ed) Plant Stems: Physiology and Functional Morphology, pp. 281–319. Academic Press, San Diego, CA (1995).
Lloyd AD, Mellerowicz EJ, Chow CH, Riding RT, Little CHA: Fluctuations in ribosomal RNA gene content and nucleolar activity in the cambial region of Abies balsamea (Pinaceae) shoots during reactivation. Am J Bot 81: 1384–1389 (1994).
Matsuki R, Onodera H, Yamauchi T, Uchimiya H: Tissuespecific expression of the rolC promoter of the Ri plasmid in transgenic rice plants. Mol Gen Genet 220: 12–16 (1989).
Nagata T, Okada K, Kawazu T, Takebe I: Cauliflower mosaic virus 35S promoter directs S phase specific expression in plant cells. Mol Gen Genet 207: 242–244 (1987).
Nelson EA, Dickson RE: Accumulation of food reserves in cootonwood stems during dormancy induction. Can J For Res 11: 145–154 (1981).
Nilsson O, Aldén T, Sitbon F, Little CHA, Chalupa V, Sandberg G, Olsson O: Spatial pattern of cauliflower mosaic virus 35S promoter-luciferase expression in transgenic hybrid aspen trees monitored by enzymatic assay and non-destructive imaging. Transgen Res 1: 209–220 (1992).
Pomponi M, Spanò L, Sabbadini MG, Costantino P: Restriction endonuclease mapping of the root-inducing plasmid of Agrobacterium rhizogenes 1855. Plasmid 10: 119–129 (1983).
Powell LE: Hormonal aspects of bud and seed dormancy in temperature-zone woody plants. HortScience 22: 845–850. (1987).
Sambrook J, Fritsch E, Maniatis T: Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY (1989).
Sauter JJ, vanCleve B. Biochemical and ultrastructural results during starch-sugar-conversion in ray parenchyma cells of Populus during cold adaptation. J Plant Physiol 139: 19–26 (1991).
Schell J: Transgenic plants as tools to study the molecular organization of plant genes. Science 237: 1176–1183 (1987).
Schmülling T, Schell J, Spena A: Promoters of the rolA, B and C genes of Agrobacterium rhizogenes are differentially regulated in transgenic plants. Plant Cell 1: 665–670 (1989).
Spanò L, Pomponi M, Costantino PSV, vanSlogteren GMS, Tempé J: Identification of T-DNA in the root-inducing plasmid of the agropine type Agrobacterium rhizogenes 1855. Plant Mol Biol 1: 291–300 (1982).
Sugaya S, Hayakawa K, Handa T, Uchimiya H: Cell-specific expression of the rolC gene of the TL-DNA of Ri plasmid in transgenic tobacco plants. Plant Cell Physiol 30: 649–653 (1989).
Walden R, Koncz C, Schell J: The use of gene vectors in plant molecular biology. Methods Mol Cell Biol 1: 175–194 (1990).
Whetten R, Sederoff R: Genetic engineering of wood. For Ecol Managem 43: 301–316 (1991).
Williamson JD, Hirsch-Wyncott ME, Larkins BA, Gelvin SB: Differential accumulation of a transcript driven by the CaMV 35S promoter in transgenic tobacco. Plant Physiol 90: 1570–1576 (1989).
Yokoyama R, Hirose T, Fujii N, Aspuria ET, Kato A, Uchimiya H: The rolC promoter of Agrobacterium rhizogenes Ri plasmid is activated by sucrose in transgenic tobacco plants. Mol Gen Genet 244: 15–22 (1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nilsson, O., Little, C.H.A., Sandberg, G. et al. Expression of two heterologous promoters, Agrobacterium rhizogenes rolC and cauliflower mosaic virus 35S, in the stem of transgenic hybrid aspen plants during the annual cycle of growth and dormancy. Plant Mol Biol 31, 887–895 (1996). https://doi.org/10.1007/BF00019475
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00019475