Summary
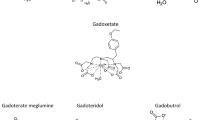
The stability and excretion of [14C]-gadodiamide (GdDTPA-BMA) was studied in male rats after i.v. injection of 0.3 mmol/kg [14C]-GdDTPA-BMA (Gd-diethylenetriaminepentaacetic-acid-bis-methylamide) formulated as gadodiamide injection (OMNISCAN®, Nycomed Imaging AS. Oslo, Norway). Samples of blood and urine obtained within 60 min and 6 h postdosing, respectively, were analyzed for radiolabeled compounds. Analysis of GdDTPA-BMA in high and low molecular weight fractions of serum indicated no protein binding. HPLC analyses of urine samples obtained 0–2 h, 2–4 h and 4–6 h after injection revealed no detectable amounts of biotransformation products of GdDTPA-BMA. Serum samples obtained 30 min and 60 min after injection contained 9–13 μM of an unidentified compound which had a retention time different from all conceivable metabolites of gadodiamide. A similar concentration of this unknown compound was found in spiked predose serum samples. The total amount of the unknown compound in serum was less than 1% of the injected dose of [14C]-gadodiamide injection. It is concluded that gadodiamide, when administered i.v. as gadodiamide injection at a dosage of 0.3 mmol/kg, is stable in vivo and that the very major part of the dose (> 99%) is excreted in urine as an unchanged complex.
Similar content being viewed by others
References
Harpur S.E., Worah D., Hals P-A., Holtz E., Furuhama K., Nomura H. (1993): Preclinical assessment of pharmacokinetics of gadodiamide injection, a new magnetic resonance imaging contrast agent. Invest. Radiol., 28 (Suppl. 1), S28-S43.
Øksendal A.N., Hals P-A. (1993): Biodistribution and toxicity of MR imaging contrast media. J. Magn. Reson. Imaging, 3, 157–165.
Bamhart J.L., Kuhnert N., Bakan D.A., Berk R.N. (1987): Biodistribution of GdCl3 and Gd-DTPA and their influence on proton magnetic relaxation in rat tissues. Magn. Reson. Imaging, 5, 221–231.
Wedeking P., Tweedle M. (1988): Comparison of the biodistribution of153Gd-labeled Gd(DTPA)2−, Gd(DOTA)−, and Gd(acetate)n in mice. Nucl. Med. Biol., 15, 395–402.
Durbin P.W., Williams M.H., Ge M., Newman R.H., Hamilton J.G. (1956): Metabolism of lanthanons in the rat. Proc. Soc. Exp. Biol. Med., 91, 78–85.
VanWagoner M., O’Toole M., Worah D., Leese P.T., Quay S.C. (1991): A phase I clinical trial with gadodiamide injection, a nonionic magnetic resonance imaging enhancement agent. Invest. Radiol., 26, 980–986.
Hvattum E., Normann P.T., Jamieson G.C., Lai J-J., Skotland T. (1995): Detection and quantitation of gadolinium-chelates in human serum and urine by high-performance liquid chromatography and post-column derivatization of gadolinium with Arsenazo III. J. Pharmaceut. Biomed. Anal., 13, 927–952.
Wedeking P., Kumar K., Tweedle M.F. (1992): Dissociation of gadolinium chelates in mice: relationship to chemical characteristics. Magn. Reson. Imaging, 10, 641–648.
Harrison A., Walker C.A., Pereira K.A., et al. (1993): Hepato-biliary and renal excretion in mice of charged and neutral gadolinium complexes of cyclic tetra-aza-phosphinic and carboxylic acids. Magn. Reson. Imaging, 11, 761–770.
Weinmann H.J., Brasch R.C., Press W.R., Westbey G.E. (1984): Characteristics of gadolinium-DTPA complex: a potential NMR contrast agent. Am. J. Radiol., 142, 619–624.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Normann, P.T., Hals, P.A. In vivo stability and excretion of gadodiamide (GdDTPA-BMA), a hydrophilic gadolinium complex used as a contrast enhancing agent for magnetic resonance imaging. European Journal of Drug Metabolism and Pharmacokinetics 20, 307–313 (1995). https://doi.org/10.1007/BF03190250
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03190250