Abstract
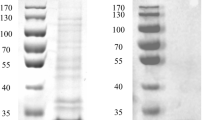
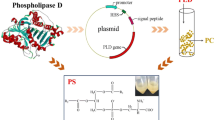
The phospholipase D (PLD) gene pld encoding the mature PLD enzyme fromStreptomyces chromofuscus was cloned and sequenced. The recombinant protein has been expressed inEscherichia coli and purified. The yield of aim protein was up to 27.5 mg/l culture medium. Because it was targeted with a 6 × his-tag, the recombinant protein could be much more easily purified by using Ni-NTA His·Bind Purification Kits. After concentration, the concentration of rPLD was 1.0 mg/ml buffer. With transphosphatidylation activity of rPLD, phosphatidylserine (PS) could be produced from phosphatidylcholine (PC). The optimum organic phase was chloroform, optimum pH was 7.5, optimum temperature and time of transphosphatidylation reaction that catalysed by rPSS were 30 °C and 6 h, respectively. The highest transphosphatidylation rate of rPSS was up to 31% and the specific enzyme activity was up to 39 U/mg.
Similar content being viewed by others
Reference
Bradford M.M. (1976). A rapid and sensitive for the quantitation of microgram quantitites of protein utilizing the principle of protein-dye binding. Anal. Biochem., 72: 248–254.
Comfurius P., Zwaal R.F. (1987). The enzymatic synthesis of phosphatidylserine and purification by CM-cellulose column chromatography. Biochim. Biophys. Acta, 488: 35–42.
Gottlin E.B., Rudoliph A.E., Zhao Y. (1998). Catalytic mechanism of the phospholipase D superfamily proceeds via a covalent phosphohistidine intermediate. Biochemistry, 95: 9202–9207.
Hanahan D.J., Chaikoff I.L. (1947). The phosphorus-containing lipids of the carrot. J. Biol. Chem., 168: 233–240.
Hatanaka T., Negishi T., Kubota-Akizawa M., Hagishita T. (2002). Purification, characterization, cloning and sequencing of phospholipase D fromStreptomyces septatus TH-2. Enzyme Microb. Technol., 31 (3): 233–241.
Leiros I., Secundo F., Zambonelli C., Servi S., Hough E. (2000). The first crystal structure of a phospholipase D. Structure, 8: 655–667.
Lekh R.J., Toru K., Naomi G. (1989). Conversion of phosphatidylcholine to phosphatidylserine by various phospholipase D in the presence of L-or D-serine. Biochim. Biophys. Acta, 1003: 277–283.
Lekh R.J., Eri T., Shoichi S. (1992). Increasing productivity by removing choine in conversion of phosphatidylcholine to phosphatidylserine by phospholipase D. Journal of fermentation and Bioengineering., 73:357–61.
Lowry O.H., Rosbrough N.J., Farr A.L., Randall R.J. (1951). Protein measurement with the Folin phenol reagent. J. Biol. Chem., 193: 265–275.
Masashi H., Tatuya S., Teruyuki K. (2000). Conversion to docosahexaenoic acid-containing phosphatidylserine from squid skin lecithin by phospholipase D-mediated transphosphatidylation. J. Agric. Food Chem., 48:4550–4554.
Ogino C., Kanemasu M., Hayashi Y., Kondo A., Shimizu N., Tokuyama S., Tahara Y., Kuroda S., Tanizawa K., Fukuda H. (2004). Over-expression system for secretory phospholipase D byStreptomyces lividans. Appl. Microbiol. Biotechnol., 64: 823–828.
Ponting C.P., Kerr I.D. (1996). A novel family of phospholipase D homologues that includes phospholipids synthases and putative endonucleases: Identification of duplicated repeats and potential active site residues. Protein Sci., 5: 914–922.
Satio M., Bourque E., Kanfer J. (1974). Phosphatidohydrolase and base-exchange activity of commercial phospholipase D. Arch. Biochem. Biophys., 164 (2): 420–428.
Shnigir V.M., Kisel M.A. (2004). Transformation of phospholipids by cabbage phospholipase D in mixed micelles containing 3-[(3-cholamidopropyl) dimethylammonio]-1-propanesulfonate. Appl. Biochem. Microbiol., 40 (3): 225–230.
Shuto S., Imamura S., Fukukawa K. (1987). A facile one-step synthesis of phosphatidylhomoserines by phospholipase D-catalyzed transphosphatidylation. Chem. Phar. Bull., 35: 447–449.
Soucek A., Michalec C., Souckova A. (1967). Enzymic hydrolysis of sphingomyelins by a toxin ofCorynebacterium ovis. Biochim. Biophys. Acta, 144 (1): 180–182.
Tadashi H., Tomofumi N., Megumi K.A. (2002). Study on thermostability of phospholipase D fromStreptomyces sp. Biochim. Biophys. Acta, 1598: 156–164.
Tairo H., Masanobu N., Tadashi H. (2000). Isolation of phospholipase D producing microorganisms with high transphosphatidylation activity. Biotechnol. Lett., 22: 1587–1590.
Uhm T.B., Li T., Bao J. (2005). Analysis of phospholipase D gene fromStreptoverticillium reticulum and the effect of biochemical properties of substrates on phospholipase D activity. 37: 641–647.
Yang H.Y., Roberts M.F. (2002). Cloning, overexpression, and characterization of a bacterial Ca2+-dependent phospholipase D. Protein Sci., 11: 2958–2968.
Yang S.F., Frees S., Benson A.A. (1967). Transphosphatidylation by phospholipase D. J. Biol. Chem., 242: 477–484.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, B., Lu, FP., Tian, L. et al. Cloning and expression of phospholipase D genepld fromStreptomyces chromofuscus . Ann. Microbiol. 58, 227–231 (2008). https://doi.org/10.1007/BF03175321
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03175321