Abstract
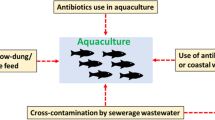
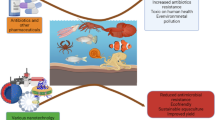
This study investigated the antibiotic resistance of enteric bacteria and their association with environmental factors in a coastal area of the Caribbean Sea. Seawater and oyster samples were collected during rainy and dry seasons. Faecal indicators of seawater fulfilled international standards, except for enterococci during dry season, while in oysters were above 800 MPN/g tissue. Different cultural methods were used to isolate enteric bacteria, further identified by biochemical tests for species of the generaEscherichia, Providencia, Kluyvera, Citrobacter, Morganella, Klebsiella andEnterococcus. A total of 21 isolates presented multiple antibiotic resistances at least to five antibiotics, with higher resistance forEnterococcus durans against 20 antibiotics tested (20/20), followed byEscherichia coli (9/20). Enteric bacteria isolated from any sample source during the rainy season presented the highest antimicrobial resistance to penicillins and cephalosporins. This is the first report of multiple antibiotic resistances in enteric environmental isolates at the Caribbean Sea. This coastal environment might serve as a reservoir of antibiotic resistant bacteria and might represent public health risks associated with the use of recreational waters and the consumption of raw seafood.
Similar content being viewed by others
References
APHA — American Public Health Association (1995). Standard Methods for the Examination of Water and Waste Water, 19th edn., APHA, Washington, DC.
Arvanitidou M., Katsouyannopoulos V., Tsakris A. (2001). Antibiotic resistance patterns of enterococci isolated from coastal bathing waters. J. Med. Microbiol., 50: 1001–1005.
Bauer A.W., Kirby W.M.M., Sherris J.C., Turck M. (1966). Antibiotic susceptibility testing by a standardized single disc method. Amer. J. Clin. Pathol., 45: 493–496.
Bustamante W., Alpízar A., Hernández S., Pacheco A., Vargas N., Herrera M.L., Vargas A., Caballero M., García F. (2003). Predominance ofvanA genotype among vancomycin-resistantEnterococcus isolates from poultry and swine in Costa Rica. Appl. Environ. Microbiol., 69: 7414–7419.
Biyela P.T., Lin J., Bezuidenhout C.C. (2004). The role of aquatic ecosystems as reservoirs of antibiotic resistant bacteria and antibiotic resistance genes. Water Sci. Technol., 50: 45–50.
Cardonha A.M.S., Vieira R.H., Rodrigues D.P., Macrae A., Peirano G., Teophilo G.N. (2004). Faecal pollution in water from storm sewers and adjacent seashores in Natal, Rio Grande do Norte, Brazil. Int. Microbiol., 7: 213–218.
Constanzo S.D., Murby J., Bates J. (2005). Ecosystem response to antibiotics entering in the aquatic environment. Mar. Pollut. Bull., 51: 218–223.
Dang H.Y., Song L., Chen M., Chang Y. (2006a). Concurrence ofcat andtet genes in multiple antibiotic-resistant bacteria isolated from a sea cucumber and sea urchin mariculture farm in China. Microb. Ecol., 52: 634–643.
Dang H.Y., Zhang X.X., Song L.S., Chang Y.Q., Yang G.P. (2006b). Molecular characterizations of oxytetracycline resistant bacteria and their resistance genes in mariculture waters of China. Mar. Pollut. Bull., 52: 1494–1503.
Dang H.Y., Zhang X.X., Song L.S., Chang Y.Q., Yang G.P. (2007). Molecular determination of oxytetracycline-resistant bacteria and their resistance genes from mariculture environments of China. J. Appl. Microbiol., 103: 2580–2592.
Dang H.Y., Ren J., Song L., Sun S., An L. (2008a). Diverse tetracycline resistant bacteria and resistance genes from coastal waters of Jiaozhou Bay. Microb. Ecol., 55: 237–246.
Dang H.Y., Ren J., Song L., Sun S., An L. (2008b). Dominant chloramphenicol-resistant bacteria and resistance genes in coastal waters of Jiaozhou Bay, China. World J. Microbiol. Biotechnol., 24: 209–217.
De Oliveira A.J., Pinhata J.M. (2008). Antimicrobial resistance and species composition ofEnterococcus spp. isolated from waters and sands of marine recreational beaches in Southeastern Brazil. Water Res., 42: 2242–2250.
Devriese L.A., Collins M.D., Wirth R. (1992). The genusEnterococcus. In: Balows A., Trüper H.G., Dworkin M., Harder W., Schleifer K., Eds, The Prokaryotes. A Handbook on the Biology of Bacteria: Ecophysiology, Isolation, Identification, Applications. Springer-Verlag, New York, USA, pp. 1465–1481.
Fernández-Delgado M., Contreras M., García-Amado M.A., Gueneau P., Suárez P. (2007). Occurrence ofProteus mirabilis associated with two species of Venezuelan oysters. Rev. Inst. Med. trop. S. Paulo, 49: 355–359.
Fernández-Delgado M., García-Amado M.A., Contreras M., Edgcomb V., J., Gueneau P., Suárez P. (2009).Vibrio cholerae non-O1, non-O139 associated with seawater and plankton from coastal marine areas of the Caribbean Sea. Int. J. Env. Health Res., 19: 279–289.
Goñi-Urriza M., Capdepuy M., Arpin C., Raymond N., Caumette P., Quentin C. (2000). Impact of an urban effluent on antibiotic resistance of riverine Enterobactericeae andAeromonas spp. Appl. Environ. Microbiol., 66: 125–132.
Herwig R.P., Gray J.P., Weston D.P. (1997). Antibacterial resistant bacteria in superficial sediments near salmon net-cage farms in Puget Sound, Washington. Aquaculture, 149: 263–283.
Kühn S., Iversen A., Burman L.G., Olsson-Liljequist B., Franklin A., Finn M., Aarestrup F., Seyfarth A., Blanch A.R., Taylor H., Caplin J., Moreno M.A., Dominguez L., Möllby R. (2000). Epidemiology and ecology of enterococci, with special reference to antibiotic resistant strains, in animals, humans and the environment. Int. J. Antimicrob. Agents, 14: 337–342.
Kumar H.S., Parvathi A., Karunasagar I., Karunasagar I. (2005). Prevalence and antibiotic resistance ofEscherichia coli in tropical seafood. World J. Microbiol. Biotechnol., 21: 619–623.
Kummerer K. (2004). Resistance in the environment. J. Antimicrob. Chemother., 54: 311–320.
Lima-Bittencourt C.I., Cursino L., Gonçalves-Dornelas H., Pontes D.S., Nardi R.M.D., Callisto M., Chartone-Souza E., Nascimiento A.M.A. (2007). Multiple antimicrobial resistance in Enterobacteriaceae isolates from pristine freshwater. Genet. Mol. Res., 6: 510–521.
Lukášová J., Šustácková A. (2003). Enterococci and Antibiotic Resistance. Acta Vet. Brno., 72: 315–323.
OPS — Organización Panamericana de la Salud (2004). Legislación sobre antibióticos en América Latina. Available at: http://www.paho.org/spanish/ad/dpc/cd/amr-legis.pdf. Accessed July 24, 2008.
Reinthaler F.F., Posh J., Feieri G., Wust G., Haas D., Ruckenbauer G., Mascher F., Marth E. (2003). Antibiotic resistance ofE. coli in sewage and sluge. Water Res., 37: 1685–1690.
Rice E.W., Messer J.W., Johnson C.H., Reasoner D.J. (1995). Occurrence of high-level aminoglycoside resistance in environmental isolates of enterococci. Appl. Environ. Microbiol., 61: 374–376.
Salyers A.A., Gupta A., Wang Y. (2004). Human intestinal bacteria as reservoirs for antibiotic resistance genes. Trends Microbiol., 12: 412–416.
Schwartz T., Kohnen W., Janses B., Obst U. (2003). Detection of antibiotic-resistant bacteria and their resistance genes in wastewater, surface water, and drinking water biofilms. FEMS Microbiol. Ecol., 43: 325–335.
Van T.T.H., Chin J., Chapman T., Tran L.T., Coloe P.J. (2008). Safety of raw meat and shellfish in Vietnam: An analysis ofEscherichia coli isolations for antibiotic resistance and virulence genes. Int. J. Food Microbiol., 124: 217–223.
Wang C., Dang H., Ding Y. (2008). Incidence of diverse integrons and β-lactamase genes in environmentalEnterobacteriaceae isolates from Jiaozhou Bay, China. World J. Microbiol. Biotechnol., 24: 2889–2896.
Watkinson A.J. (2007). Antibiotic-resistanceEscherichia coli in wastewaters, surface waters, and oysters from an urban riverine system. Appl. Environ. Microbiol., 73: 5667–5670.
Young H.K. (1993).Antimicrobial resistance spread in aquatic environments. J. Antimicrob. Chemother., 31: 627–635.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fernández-Delgado, M., Suárez, P. Multiple antibiotic resistances of enteric bacteria isolated from recreational coastal waters and oysters of the Caribbean Sea. Ann. Microbiol. 59, 409–414 (2009). https://doi.org/10.1007/BF03175123
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03175123