Summary
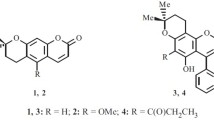
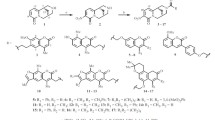
The behaviour of 7-hydroxy-coumarin indicates that there exists a double bond between 7 and 8 positions. But 7-hydroxy-4: 8-dimethyl-coumarin couples with diazocompounds and undergoes mercuration in the benzene nucleus. Its acetyl derivative undergoes the Fries migration and its allyl ether the Claisen transformation. It is therefore concluded that the nuclear double bonds are not fixed and are capable of assuming alternative positions.
Similar content being viewed by others
References
H. B. Mills and I. G. Nixon,J. C. S., 1930, 2510.
L. F. Fieser and W. C. Lothrop,J. Am. C. S., 1935, 1459.
W. Baker and (Miss) Lothian,J. C. S., 1935, 628.
D. B. Limaye and D. D. Gangal,Rasayanam, 1936,1, 15.
S. Rangaswami and T. R. Seshadri,Proc. Ind. Acad. Sci., 1937,6A, 112.
L. F. Fieser and W. C. Lothrop,J. Am. C. S., 1936, 2050.
T. R. Seshadri and P. Suryaprakasarao,Proc. Ind. Acad. Sci., 1936,4A, 162.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rangaswami, S., Seshadri, T.R. Fixation of the aromatic double bonds in the coumarin ring system. Proc. Indian Acad. Sci. (Math. Sci.) 7, 8–12 (1938). https://doi.org/10.1007/BF03045375
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03045375