Abstract
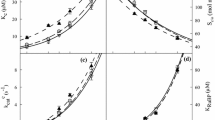
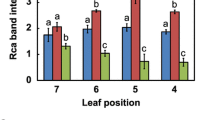
Decrease in rubisco activation at high CO2 concentration was caused by decrease in carbamylation of rubisco (Rohet al., 1996). However, it is unclear whether decrease in carbamylation rate at high CO2 concentration is due to decrease in activity itself or content of rubisco activase. To clarify this ambiguity, investigation was performed to determine effects of CO2 concentration on rubisco activase with kidney bean (Phaseolus vulgaris L.) leaves grown at normal CO2 (350 ppm) and high CO2 (650 ppm) concentration. The analysis of Western blotting showed that the 50 and 14.5 kl) polypeptides were identified immunochemically as the large and small subunits of rubisco in the preparation, respectively. For the 14.5 kD small subunit, the degree of intensity at high CO2 concentration was similar to that at normal CO2 concentration. For the 50 kD large sububit, however, the intensity of a band at high CO, concentration was significantly higher than that at normal CO2 concentration, indicating that only the large subunit is affected by high CO2 concentration. The analysis of Western immunoblotting showed two major polypeptides at 46 and 42 kD which were identified as rubisco activase subunits. The intensities of two bands were shown to be higher at normal CO2 than high CO2 concentration. These data indicate that decrease of carbamylation resulting from increase of CO2 concentration was caused by rubisco activase. Finally, by employing ATP hydrolysis assay and ELISA, we also observed a significant decrease in both activity and content of rubisco activase as CO2 concentration was raised from normal to high CO2 concentration. These results suggest that decrease in rubisco carbamylation at high CO2 concentration is caused by activity itself and/or content of rubisco activase.
Similar content being viewed by others
Literature Cited
Andrews, T.J. and G.H. Lorimer. 1987. Rubisco: Structure, mechanisms, and prospects for improvements.In Biochemistry of Plants. M.D. Hatch and N.K. Boardmaan (ed.s.), Vol. 10, Academic Press, New York pp. 131–218.
Bowes, G. 1991. Growth at elavated CO2: Photosynthetic responses mediated through rubisco.Plant Cell Environ. 14: 759–806.
Bradford, M.M. 1976. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding.Anal. Biochem. 72: 248–254.
Campbell, W.J. and W.L. Ogren. 1990. Electron transport through photosystem I stimulates light activation of ribulose bisphosphate carboxylase/oxygenase (rubisco) by rubisco activasc.Plant Physiol. 94: 479–484.
Campbell, W.J. and W.L. Ogren. 1992. Light activation of rubisco by rubisco activase and thylakoid membranes.Plant Cell Physiol. 33: 751–756.
Downton, W.J.S., O. Bjorkman and C.S. Pike. 1980 Consequences of increased atmospheric concentrations of carbon dioxide for growth and photosynthesis of higher plant.In Carbon Dioxide and Climate: Australian Research. G.I. Pearman (ed.). Australian Academy of Science, Canberra, pp. 143–151.
Harlow, E. and D. Lane. 1988. Antibodies. A Laboratory manual. Cold Spring Harbor Laboratory.
Hartman, F.C. and M.R. Harpel. 1994. Structure, function, regulation and assembly of D-ribulose-l,5-bisphosphate carboxylase/oxygenase.Ann. Rev. Biochem. 63: 197–234.
Laemmli, U.K. 1970. Cleavage of structural proteins during the assembly of the head of the hacteriophage T4.Nature 227: 680–685.
Lilley, R.McC. and A.R. Portis, Jr. 1990. Activation of ribulose-1,5-bisphosphate carboxylase/oxygenase (rubisco) by rubisco activase. Effects of some sugar phosphates.Plant Physiol. 94: 245–250.
Lorimer, G.H. andH.M. Miziorko. 1980. Carbamate formation on the ɛ-amino group of a lysyl residue as the basis for the activation of ribulose bisphosphate carboxylase by CO2 and Mg2+.Biochemistry 19: 5321–5328.
Majeau, N. and J.R. Coleman. 1996. Effect CO2 of concentration on carbonic anhydrase and ribulose-1,5-bisphosphate carboxylase/oxygenase expression in pea.Plant Physiol. 112: 569–574.
Micallef, B.J., K.A. Haskins, P.J. Vanderveer, K.S. Roh, C.K. Shewmaker and T.D. Sharkey. 1995 Altered photosynthesis, flowering, and fruiting in transgenic tomato plants that have an increased capacity for sucrose synthesis.Planta 196: 327–334.
Miziorko, H.M. and G.H. Lorimer. 1983. Ribulose-1,5-bisphosphate carboxylase-oxygenase.Ann. Rev. Biochem. 52: 507–535.
Morin, F., M. Andre and T. Betsche. 1992. Growth kinetics, carbohydrate and leaf phosphate content of clover(Trifolium subterraneum L.) after transfer to a high CO2 atmosphere or to high light and ambient air. CO2 and ribulose bisphosphate concentrations by rubisco activase.Plant Physiol. 99: 89–95.
Perchorowicz, J.T., D.A. Raynes and R.G. Jensen. 1981. Light limitation of photosynthesis and activation of ribulose bisphosphate carboxylase in wheat seedings.Proc. Natl. Acad. Sci. USA 78: 2985–2989.
Portis, A.R. Jr. 1990. Rubisco activase.Biochim. Biophys. Acta 1015: 15–28.
Portis, A.R. Jr. 1992, Regulation of ribulose 1,5-bisphosphate carboxylase/oxygenase activity.Annu. Rev. Plant Physiol. Plant Mol. Biol. 43: 415–437.
Portis, A.R. Jr.,M.E. Salvucci and W.L. Ogren. 1986 Activation of ribulose bisphosphate carboxylase/oxygenase at physiological CO2 and ribulose bisphosphale concentrations by Rubisco activase.Plant Physiol. 82: 967–971.
Robinson, S.P. and A.R. Portis, Jr. 1989a. Adenosine triphosphate hydrolysis by purified rubisco activase.Arch. Biochem. Biophys. 268: 93–99.
Robinson, S.P. and A.R. Portis, Jr. 1989b. Ribulose-1,5-bisphosphate carboxylase/oxygenase activase protein prevents thein vitro decline in activity of ribulose-1,5-bisphosphate carboxylase/oxygenase.Plant Physiol. 90: 968–971.
Robinson, S.P., V.J. Streusand, J.M. Chatrield and A.R. Portis, Jr. 1988. Purification and assay of rubisco activase from leaves.Plant Physiol. 88: 1008–1014.
Roh, K.S., J.K. Kim, S.D. Song, H.S. Chung and J.S. Song. 1996. Decrease of the activation and carbamylation of rubisco by high CO2 in kidney bean.Korean J. Biotechnol. Bioeng. 11: 295–302.
Salvucci, M.E. 1989. Regulation of rubisco activityin vivo.Physiol. Plant 77: 164–171.
Salvucci, M.E. 1992. Subunit infractions of rubisco activase: Polyethylene glycol promotes self-association, stimulates ATPase and activation activities, and enhances interactions with rubisco.Arch. Biochem. Biophy. 298: 688–696.
Salvucci, M.E., A.R. Portis, Jr.and W.L. Ogren. 1985 A soluble chloroplast protein catalyzes ribulose-bisphosphate carboxylase/oxygenase activationin vivo.Photosynth. Res. 7: 191–203.
Salvucci, M.E., J.M. Werneke, W.L. Ogren and A.R. Portis, Jr. 1987. Purification of species distribution of rubisco activase.Plant Physiol. 84: 930–936.
Somerville, C.R., A.R. Portis, Jr.and W.L. Ogren. 1982. A mutant ofArabidopsis thaliana which lacks activation of RuBP carboxylasein vivo.Plant Physiol. 70:381–387.
Stitt, M. 1991. Rising CO2 levels and their potential significance for carbon flow in photosynthetic cells.Plant Cell Environ. 14: 741–762.
Streusand, V.J. and A.R. Portis, Jr. 1987 Rubisco activase mediates ATP-dependent activation of ribulose bisphosphate carboxylase.Plant Physiol. 85: 152–154.
Vu, C.V., L.H. Allen, Jr.and G. Bowes. 1983. Effect of light and elevated atmosperic CO2 on the ribulose bisphosphate carboxylase activity and ribulose bisphosphate level soybean leavesPlant Physiol. 73: 729–734.
Wang, Z.V., G.W. Snyder, B.D. Esau, A.R. Portis, Jr.and W.L. Ogren. 1992. Species-dependent variation in the interaction of substrate-bound ribulose-1,5-bisphosphate carboxylase/oxygenase (rubisco) and rubisco activase.Plant Physiol. 100: 1858–1862.
Wang, Z.V. and A.R. Portis, Jr. 1991. A fluorometric study with 1-anilinonaphthalene-8-sulfonie acid (ANS) of the interactions of ATP and ADP with rubisco activase.Biochim. Biophys. Acta 1079: 263–267.
Wang, Z.V. and A.R. Portis, Jr. 1992. Dissociation of ribulose-1,5-bisphosphate bound to ribulose-1,5-bisphosphate carboxylase/oxygenase and its enhancement by ribulose-1,5-bisphosphate carboxylase/oxygenase activase-mediated hydrolysis of ATP.Plant Physiol. 99: 1348–1353.
Woodrow, I.W. 1994. Control of steady-state photosynthesis in sunflowers growing in enhanced CO2.Plant Cell Environ. 17: 277–286.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Roh, K.S., Kim, I.S., Kim, B.W. et al. Decrease in carbamylation of rubisco by high CO2 concentration is due to decrease of rubisco activase in kidney bean. J. Plant Biol. 40, 73–79 (1997). https://doi.org/10.1007/BF03030237
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03030237