Abstract
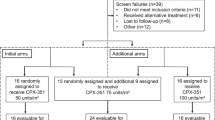
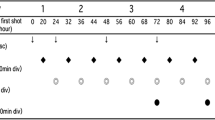
Aggressive adult T-cell leukemia-lymphoma (ATL) generally has a very poor prognosis. Deoxycoformycin (DCF, pentostatin), an inhibitor of adenosine deaminase, has shown promising therapeutic efficacy for ATL. To develop a new effective therapy against aggressive ATI., we carried out a multicenter phase II study of DCF-containing combination chemotherapy. Sixty-two previously untreated patients with ATL (34, 21, and 7 patients with diseases of the acute, lymphoma, and unfavorable chronic types, respectively) were enrolled, but 2 were ineligible because they were judged to be favorable chronic types. A regimen of 1 mg/m2 vincristine intravenously on days 1 and 8, 40 mg/m2 doxorubicin intravenously on day 1,100 mg/m2 etoposide intravenously on days 1 through 3, 40 mg/m2 prednisolone orally on days 1 and 2, and 5 mg/m2 DCF intravenously on days 8,15, and 22 was administered every 28 days for 10 cycles unless disease progression or toxic complications occurred. Fifty-two percent of 60 eligible patients responded (95% confidence interval [CI], 38%-65%), with 17 patients (28%) achieving a complete response (CR) (95% CI, 17%-41%) and 14 achieving a partial response. The CR rate was inferior to those of both the previous Japan Clinical Oncology Group (JCOG) study (JCOG8701, 43%), a 9-drug combination chemotherapy of the second generation, and the subsequent JCOG9303 study (35%), a granulocyte colony-stimulating factor-supported, dose-intensified, 9-drug regimen. The median survival time of the 60 eligible patients in JCOG9109 was 7.4 months, and the estimated 2-year survival rate was 15.5%; these results were identical with those of JCOG8701 but inferior to those of JCOG9303. Grade 4 neutropenia and infection of grade 3 or greater were frequent (67% and 22%, respectively), and treatment-related death was observed in 4 patients (7%), scpticcmia in 2, and cytomcgalovirus pneumonia in 2. We conclude that DCF-containing combination chemotherapy is not a promising regimen against aggressive ATL.
Similar content being viewed by others
References
Uchiyama T, Yodoi J, Sagawa K, Takatsuki K, Uchino H. Adult T-cell leukemia: clinical and hématologie features of 16 cases.Blood. 1977;50:481–492.
Yoshida M, Miyoshi I, Hinuma Y. Isolation and characterization of retrovirus from cell lines of human adult T cell leukemia and its implication in the disease.Proc Natl Acad Sei 11 S A. 1982;79:2031–2035.
Shimoyama M. Diagnostic criteria and classification of clinical sub-types of adult T-cell leukaemia-lymphoma: a report from the Lymphoma Study Group (1984–87).Br J Haematol. 1991;79:428–437.
Pawson R, Mufti GJ, Pagliuca A. Management of adult T-cell lcukacmia/lyraphoraa.Br J Haematol. 1998;100:453–458.
Tobinai K. Adult T-cell lcukcmia-lymphoma. In: Abeloff MD, Arraitagc JO, Lichter AS, Nicdcrhubcr JE. eds.Clinical Oncology. 2nd ed. New York, NY: Churchill Livingstone; 2000:2748–2768.
Shimoyama M. Chemotherapy of ATL. In: Takatsuki K, ed.Adult T-cell Leukemia. Oxford, UK: Oxford University Press; 1994:221–237.
Kuwazuru Y, Hanada S, Furukawa T, et al. Expression of P-glycoprotein in adult T-cell leukemia cells.Blood. 1990;76:2065–2071.
Tkeda K, Oka M, Yamada Y, et al. Adult T-celI leukemia cells overexpress the multidrug-resistance-protein (MRP) and lung-resistance-protein (LRP) genes.M J Cancer. 1999;82:599–604.
Tsukasaki K, Koeffler P, Tomonaga M. Human T-lymphotropic virus type 1 infection.Baillieres Best Pract Res Clin Haematol. 2000;13:231–243.
Grever M, Kopecky K, Foucar MK, et al. Randomized comparison of pentostatin versus interferon alfa-2a in previously untreated patients with hairy cell leukemia: an intergroup study.J Clin Oncol. 1995;13:974–982.
Flinn IW, Kopecky KJ, Foucar MK, et al. Long-term follow-up of remission duration, mortality, and second malignancies in hairy cell leukemia patients treated with pentostatin.Blood. 2000;96:2981–2986.
Mercieca J, Matutes E, Dearden C, MacLenn an K, Catovsky D. The role of pentostatin in the treatment of T-cell malignancies: analysis of response rate in 145 patients according to disease subtype.J. Clin Oncol. 1994;12:2588–2593.
Ho AD, Suciu S, Stryckmans P, et al. Pentostatin in T-cell malignancies: a phase II trial of the EORTC Leukemia Cooperative Group.Ann Oncol. 1999;10:1493–1498.
Kurzrock R, Pilat S, Duvic M. Pentostatin therapy of T-cell lymphomas with cutaneous manifestations.J. Clin Oncol. 1999;17:3117–321.
Grever MR, Siaw ME, Jacob WE, et al. The biochemical and clinical consequences of 2’-deoxycoformycin in refractory lymphoproliferative malignancy.Blood. 1981;57:406–417.
de Körte D, Haverkort WA, van Leeuwen EF, Roos D, van Gennip AH. Biochemical consequences of 2’-deoxycoformycin treatment in a patient with T-cell lymphoma: some unusual findings.Cancer. 1987;60:750–755.
Major PP, Agarwal RP, Kufe DW. Clinical pharmacology of deoxycoformycin.Blood. 1981;58:91–96.
Daenen S, Rojer RA, Smit JW, Halie MR, Nieweg HO. Successful chemotherapy with deoxycoformycin in adult T-cell lymphomaleukaemia.Br.I Haematol 1984;58:723–727.
Yamaguchi K, Yul LS, Oda T, et al. Clinical consequences of 2’-deoxycoformycin treatment in patients with refractory adult T-cell leukaemia.Leuk Res. 1986;10:989–993.
Lofters W, Campbell M, Gibbs WN, Cheson BD. 2’-Deoxycoformycin therapy in adult T-cell leukemia/lymphoma.Cancer. 1987;60:2605–2608.
Tobinai K, Shimoyama M, Inoue S, et al. Phase I study of YK-176 (2’-deoxycoformycin) in patients with adult T-cell leukemia-lymphoma: the DCF Study Group.Jpn J Clin Oncol. 1992;22:164–171.
Kodama K, Kusakabe H, Machida H, et al. Isolation of 2’-deoxycoformycin and cordyceptin from wheat bran culture ofAspergillus nidulans Y 176-2.Agric Biol Chem. 1979;43:2375–2377.
Gryn J, Gordon R, Bapat A, Goldman N, Goldberg J. Pentostatin increases the acute toxicity of high dose cyclophosphamide.Bone Marrow Transplant. 1993;12:217–220.
Tobinai K, Shimoyama M, Minato K, et al. Japan Clinical Oncology Group phase II trial of second-generation “LSG4 protocol” in aggressive T- and B-lymphoma: a new predictive model forT-and B~lymphoma [abstract].Proc Am Soc Clin Oncol. 1994;13:378.
Wilson WH, Bryant G, Bates S, et al. EPOCH chemotherapy: toxicity and efficacy in relapsed and refractory non-Hodgkin’s lymphoma.J. Clin Oncol. 1993;11:1573–1582.
Velasquez WS, McLaughlin P,Tucker S, et al. ESHAP—an effective chemotherapy regimen in refractory and relapsing lymphoma: a 4-year follow-up study.Clin Oncol. 1994;12:1169–1176.
Tanaka K, Rgami K, Nakamura E, et al. A case of ATI, presenting a huge intrahepatic tumor, abated with etoposide (NK-171) therapy alone [in Japanese].Rinsho Kelsueki. 1987;28:392–397.
Wakui A, YokoyamaM.Takahashi H, et al. A phase I study of VP-16-213 (VP, etoposide) by single and 5-day intravenous administration [in Japanese],Can To Kagaku Ryoho. 1986;13:319–329.
Naito K, Kobayashi M, Oyama A, et al. Phase II study of carbo-platin in malignant lymphoma [in Japanese].Gan To Kagaku Ryoho. 1988;15:2145–2151.
Oken MM, Creech RII, Tormey DC, et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group.Am J Clin Oncol. 1982;5:649–655.
Shimoyama M, Fukuda H, Saijo N, Yamaguchi N. Japan Clinical Oncology Group (JCOG).Jpn J Clin Oncol. 1998;28:158–162.
World Health Organization.WHO Handbook for Reporting Results of Cancer Treatment. Geneva, Switzerland: World Health Organization; 1979. WHO Offset Publication No. 48.
Yamada Y,Tomonaga M, Fukuda H, et al. A new G-CSF-supported combination chemotherapy, LSG15, for adult T-cell leukaemia-lymphoma: Japan Clinical Oncology Group Study 9303.Br.1 Haematol. 2001;113:375–382.
Kaplan EL, Meier P. Nonparametric estimation from incomplete observations.J am Stat Assoc. 1958;53:157–181.
Shimoyama M, Ota K, Kikuchi M, et al. Chemotherapeutic results and prognostic factors of patients with advanced non-Hodgkin’s lymphoma treated with VEPA or VEPA-M.J. Clin Oncol. 1988;6:128–141.
Shimoyama M, Ota K, Kikuchi M, et al. Major prognostic factors of adult patients with advanced T-cell Iymphoma/leukemia.J. Clin Oncol. 1988;6:1088–1097.
Fisher RI, Gaynor ER, Dah Iberg S, et al. Comparison of a standard regimen (CHOP) with three intensive chemotherapy regimens for advanced non-Hodgkin’s lymphoma.N Engl J Med. 1993;328:1002–1006.
Urba WJ, Baseler MW, Kopp WC, et al. Deoxycoformycin-induced immunosuppression in patients with hairy cell leukemia.Blood. 1989;73:38–46.
Waldmann TA, White JD, Goldman CK, et al. The interleukin-2 receptor: a target for monoclonal antibody treatment of human T-cell lymphotropic virus I-induced adult T-cell leukemia.Blood. 1993;82:1701–1712.
Waldmann TA, White JD, Carrasquillo JA, et al. Radioim-munothcrapy of intcrlcukin-2R a-expressing adult T-cell leukemia with yttrium-90-labeled anti-Tac.Blood. 1995;86:4063–4075.
41. Sparano JA, Wiernik PH, Strack M, Leaf A, Becker N, Valentine ES. Infusional cyclophosphamide, doxorubicin, and etoposide in human immunodeficiency virus- and human T-cell leukemia virus type I-related non-Hodgkin’s lymphoma: a highly active regimen.Blood. 1993;81:2810–2815.
Taguchi H, Kinoshita K, Takatsuki K, et al. An intensive chemotherapy of adult T-cell leukemia/lymphoma: CHOP followed by etoposide, vindesine, ranimustine, and mitoxantrone with granulocyte colony-stimulating factor support.J Acquir Immune Defic Syndr Him Retroviral. 1996;12:182–186.
Utsunomiya A, Miyazaki Y,Takatsuka Y, et al. Improved outcome of adult T cell leukemia/lymphoma with allogeneic hematopoietic stem cell transplantation.Bone Marrow Transplant. 2001;27:15–20.
Abe Y, Yashiki S, Choi I, et al. Eradication of virus-infected T-cells in a case of adult T-cell leukemia/lymphoma by nonmyeloablative peripheral blood stem cell transplantation with conditioning consisting of low-dose total body irradiation and pentostatin.Int J Hematol. 2002;76:91–93.
Author information
Authors and Affiliations
Consortia
Corresponding author
About this article
Cite this article
Tsukasaki, K., Tobinai, K., Shimoyama, M. et al. Deoxycoformycin-Containing Combination Chemotherapy for Adult T-Cell Leukemia-Lymphoma: Japan Clinical Oncology Group Study (JCOG9109). Int J Hematol 77, 164–170 (2003). https://doi.org/10.1007/BF02983215
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02983215