Abstract
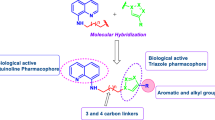
5,8-Quinazolinediones modified at positions6 and7 were synthesized and tested forin vitro antifungal activities againstCandida species andAspergillus niger. Most of 5,8-quinazolinediones 3-5 generally exhibited potent antifungal activity. 6-Arylamino-7-chloro-5,8-quinazolinediones (3) generally showed more potent antifungal activity than 7-arylthio-5,8-quinzolinediones (4) and 6,7-bis-(arylthio)-5,8-quinazolinediones (5).
Similar content being viewed by others
References
Giorgi-Renault, S., Renault, J., Gebel-Servolles, P., Baron, M., Paoletti, C., Cros, S., Bissery, M. C., Lavelle, F., and Atassi, G., Heterocyclic quinones 17. A newin vivo active antineoplastic drug.J. Med, Chem., 34, 38–46 (1991).
Jeschke, P., Under, W., Mueller, N., and Dehne, H. W., Fungicides based on amino-substituted quinolinequinones.Ger. Often., DE 4,208,874 (1993);Chem. Abstr., 118, 233893 (1993).
Mcginnis, M. R. and Rindali, M. G., Antifungal drug: In Lorian, V. (Eds.). Antibiotics in Laboratory Medicine (4th ed.). Williams and Wilkins, Baltimore, pp 176–211 (1996).
Melesani, G., Marcolin, F., and Rodighiero, G., Synthesis of 5,8- quinazolinedione.J. Med. Chem., 13, 161–162 (1970).
Middleton, R. W. and Parrick, J., Heterocyclic quinones: Patai, S., Rappoport, Z. (Eds),The Chemistry of The Quinonoid Compounds. John Wiley & Sons: London, vol 2, pp 1019–1066 (1988).
Park, H. J., Kim, Y.-S., Kim, J. S., Lee, H.-J., Lee, E.-J., Hwang, H. J., Suh, M.-E., Ryu, C.-K., and Lee, S. K., 6-Arylamino-7- chloro-quinazoline-5,8-diones as novel cytotoxic and DNA toposiomerase inhibitory agents.Bioorg. Med. Chem. Lett., 14, 3385–3388 (2004).
Renault, J., Giorgi-Renault, S., Baron, M., Mailliet, P., Paoletti, C., Cros, S., Viosin, E., and Renault, J., Heterocyclic quinone 4. A new highly cytotoxic drug: 6,7-bis(1-aziridinyl)-5,8-quinazolinedione.J. Med. Chem., 26, 1715–1719 (1983).
Roberts, H., Choo, W. M., Smith, S. C., Marzuki, S., Linnane, A. W., Porter, T. H., and Folkers, K., The site of inhibition of mitochondrial electron transfer by coenzyme Q analogs.Arch. Biochem. Biophys., 191, 306–315 (1978).
Ryu, C.-K. and Kim, H. J., The synthesis of 6-(N-arylamino)-7- chloro-5,8-quinolinedione derivatives for evaluation of antifungal activities.Arch. Pharm. Res., 17, 139–144 (1994).
Ryu, C.-K., Shin, K.H., Seo, J. H., and Kim, H. J., 6-Arylamino- 5,8-quinazolinediones as potent inhibitors of endothelium- dependent vasorelaxation.European J. Med. Chem., 37, 77–82 (2002a).
Ryu, C.-K., Sun, Y. J., Shim, J.Y., You, H. J., Choi, K.U., and Lee, H., Synthesis and antifungal activities of 6,7-bis-[S- (aryl)thio]-5,8-quinolinediones.Arch. Pharm. Res., 25, 784–789 (2002b).
Ryu, C.-K., Yi, Y.-J., Choi, I. H., Chae, M J., Han, J.-Y, Jung, O.-J., and Lee, C.-O., Synthesis and cytotoxic activity of 6-(substituted-phenyl)amino-5,8-quinazolinediones.Med. Chem. Res., 13 in press (2004).
Shaikh, I. A., Johnson, F., and Grollman, A. P., Structure-activity relationship among simple bicyclic analogues. Rate dependent of DNA degradation on quinone reduction potential.J. Med. Chem., 29, 1329–1340 (1986).
Skehan, P., Storeng, R., Scudiero, D., Monks, A., McMahon, J., Vistica, D., Warren, T. W., Bokesch, H., Kenney, S., and Boyd, M. R., New colorimetric cytotoxicity assay for anticancer-drug screening.J. Natl. Cancer Inst., 82, 1107–1112 (1990).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ryu, C.K., Shim, J.Y., Yi, Y.J. et al. Synthesis and antifungal activity of 5,8-quinazolinedione derivatives modified at positions 6 and 7. Arch Pharm Res 27, 990–996 (2004). https://doi.org/10.1007/BF02975419
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02975419