Summary
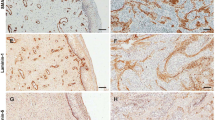
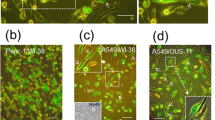
A primary culture of serous cystadenocarcinoma of the ovary was used to study the expression of intermediate filament proteins and the deposition of basal lamina proteins. It was found that cells grown on type I and IV collagens or in collagen gels failed to express vimentin, which was readily demonstrable in cultures of the same cells grown on plastic or glass. Furthermore cells grown in collagen gels formed colonies demonstrating a cystic architecture Unlike what is commonly observed on glass or plastic where laminin and fibronectin are deposited as disorganized fibrils in the extracellular space, in or on collagen these proteins appear solely at the interface between the epithelial cells and matrix. The results suggest that the extracellular matrix influences the cytoskeletal organization of the intermediate filaments and determines cell polarity. They confirm that collagen substrates permit epithelial cell cultures to progress toward a more differentiated state.
Similar content being viewed by others
References
Berg, R. A. Determination of 3- and 4-hydroxyproline. In: Cunningham, L. W.; Frederiksen, D. W., eds. Methods in enzymology, vol. 82. London: Academic Press; 1982:372–380.
Ehrmann, R. L.; Gey, G. D. The growth of cells on transparent gel of reconstituted rat-tail collagen. JNCI 16:1375–1390; 1956.
Emerman, J. T.; Pitelka, D. R. Maintenance and induction of morphological differentiation in dissociated mammary epithelium on floating collagen membranes. In Vitro 13:316–328; 1977.
Foidart, J. M. Distribution and immunoelectron-microscopic localization of laminin, a noncollagenous basement membrane glycoprotein. Lab. Invest. 42:336–342; 1980.
Franke, W. W.; Schmidt, E.; Breitkreutz, D., et al. Simultaneous expression of two different types of intermediate sized filaments in mouse keratinocytes proliferating in vitro. Differentiation. 14:35–50; 1979.
Hsu, S. M.; Raine, L.; Fanger, H. Use of avidin-biotinperoxidase complex (ABC) in immunoperoxidase techniques: a comparison between ABC and unlabelled antibody (PAP) procedures. J. Histochem. Cytochem. 29:577–580; 1981.
Iversen, L. P.; Lester, M. P.; Stensaas, L. J., et al. Characterization of a variety of standard collagen substrate: ultrastructure, uniformity and capacity to bind and promote growth of neurons. In Vitro 17:540–552; 1981.
Lazarides, E. Intermediate filaments—A chemical heterogeneous developmentally regulated class of proteins. Ann. Rev. Biochem. 51:219–250; 1982.
Michalopoulos, G.; Pitot, H. C. Primary culture of parenchymal liver cells on collagen membranes. Exp. Cell. Res. 94:70–78; 1975.
Miller, E. J.; Rhodes, R. K. Preparation and characterization of the different types of collagen. In: Cunningham, L. W.; Frederksen, D. W., eds. Methods in enzymology, vol. 82. London: Academic Press; 1982:33–65.
Sage, H.; Woodbury, R. G.; Bornstein, P. Structural studies on human type IV collagen. J. Biol. Chem. 254:9893–9900; 1979.
Simons, K.; Fuller, S. D. Cell surface polarity in epithelia. Ann. Rev. Cell. Biol. 1:243–288; 1985.
Sternberger, L. A.; Hardy, P. H.; Cuculis, J. J., et al. The unlabeled antibody enzyme method of immunohistochemistry: preparation and properties of soluble antigen-antibody complex (horseradish peroxidase anti-horseradish peroxidase) and its use in identification of spirochetes. J. Histochem. Cytochem. 18:315–333; 1970.
Strom, S. C.; Michalopoulos, G. Collagen as a substrate for cell growth and differentiation. In: Cunningham, L. W.; Frederksen, D. W., eds. Methods in enzymology. vol. 82. London: Academic Press; 1982:544–555.
Sugrue, S. P.; Hay, E. D. Response of basal epithelial cell surface and cytoskeleton to solubilized extracellular matrix molecules. J. Cell Biol. 91:45–54; 1981.
Summerhayes, I. C.; Cheng, Y. S.; Sun, T. T., et al. Expression of keratin and vimentin intermediate filaments in rabbit bladder epithelial cells at different stages of benzopyrene-induced neoplastic progression. J. Cell Biol. 90:63–69; 1981.
Uldfelder, H. Chairman. Staging system for cancer at gynecologic sites. In: Manual for staging cancer. Philadelphia: J. B. Lippincott; 1978:94–97.
Von Der Mark, R.; Mollenhauer, J.; Kuhl, K., et al. A new class of membrane proteins involved in cell-matrix interactions. In: Trelstad, R. L., ed. The role of extracellular matrix in development. New York: Alan Liss; 1984:67–87.
Wicha, M. S.; Lowrie, G.; Kohn, E., et al. Extracellular matrix promotes mammary epithelial growth and differentiation in vitro. Proc. Natl. Acad. Sci. USA 79:3213–3217; 1982.
Yang, J.; Richards, J.; Bowman, P., et al. Sustained growth and three-dimensional organization of primary mammary tumor epithelial cells embedded in collagen gels. Proc. Natl. Acad. Sci. USA 76:3401–3405; 1979.
Yang, J.; Richards, J.; Guzman, R., et al. Sustained growth in primary culture of normal mammary epithelial cells embedded in collagen gels. Proc. Natl. Acad. Sci. USA 77:2088–2092; 1980.
Yang, J.; Elias, J. J.; Petrakis, N. L. et al. Effects of hormones and growth factors on human mammary epithelial cells in collagen gel culture. Cancer Res. 41:1021–1027; 1981.
Author information
Authors and Affiliations
Additional information
Supported by grants from the Italian Assciation for Cancer Research (AIRC).
Rights and permissions
About this article
Cite this article
Liscia, D.S., Bernardi, A., Griselli, B. et al. Different substrates influence the expression of intermediate filaments and the deposition of basement membrane proteins. In Vitro Cell Dev Biol 24, 183–187 (1988). https://doi.org/10.1007/BF02623544
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02623544