Abstract
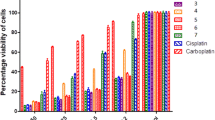
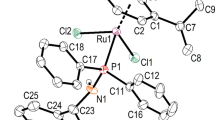
Mixed-ligand platinum complexescis-PtII(R6NH2)(NH3)X2 andcis-PtII(R5NH2)(NH3)X2 (R6 is 2,2,6,6-tetramethyl-4-piperidyl-1-oxyl and R5 is 2,2,5,5-tetramethyl-3-pyrrolidinyl-1-oxyl) were synthesized by either the reaction of aminonitroxides RNH2 with Na[PtII(NH3)Cl2I] generatedin situ (for X2=ClI) or by replacement of the iodo-chloro ligands incis-Pt11(RNH2)(NH3)ClI by dichloro and oxalato ligands. The complexes obtained were characterized by elemental analysis and by IR, UV, and ESR spectra. Forcis-Pt11(R5NH2)(NH3)Cl2, crystal and molecular structures were determined by X-ray diffraction analysis. Cisplatin accelerates autooxidation of methyl linoleate and the platinum nitroxide complexes synthesized exhibit antioxidant properties. The rate of isolated DNA binding with the new complexes is almost as high as that for cisplatin.cis-Pt11(R6NH2)(NH3)Cl2 exhibits the highest antitumor activity. The high antitumor activity of platinum nitroxide complexes shows that the possible “radical component” is not a crucial factor in the cytotoxic action of cisplatin.
Similar content being viewed by others
References
M. J. Bloemink and J. Reedijk, inMetal Ions in Biological Systems, Eds. H. Sigel and A. Sigel: M. Dekker, New York, 1996,32, p. 641.
D. L. Evans and C. Dive,Cancer Res., 1993,53, 2133.
A. Miyajima, J. Nakashima, K. Yoshioka, M. Tachibana, H. Tazaki, and M. Murai,Br. J. Cancer, 1997,76, 210.
K. Sugihara, S. Nakano, and M. Gemba,Jpn. J. Pharmacol., 1987,44, 71.
R. Ravi, S. M. Somani, and L. P. Rybak,Pharmacol. Toxicol., 1995,76, 386.
Y. Torri, M. Mutoh, H. Saito, and N. Matsuki,Eur. J. Pharmacol., 1993,248, 131.
V. D. Sen', V. A. Golubev, I. V. Kulyk, and E. G. Rozantsev,Izv. Akad. Nauk SSSR, Ser. Khim. 1976, 1745 [Bull. Acad. Sci. USSR, Div. Chem. Sci., 1976,25 (Engl. Transl.)].
S. I. Skuratova, Yu. N. Kozlov, N. V. Zakatova, and V. A. Sharpatyl,Zh. Fiz. Khim., 1971,45, 1821 [J. Phys. Chem. USSR, 1971,45 (Engl. Transl.)].
D. B. Korman, A. M. Garin, M. L. Gershanovich, V. M. Moiseenko, V. A. Snimchicov, S. A. Zharcov, P. Seminara, L. Gargano, and F. Franchi,J. Exp. Clin. Cancer Res., 1995,14, 65.
V. D. Sen', A. V. Kulikov, A. V. Shugalii, and N. P. Konovalova,Izv. Akad. Nauk, Ser. Khim., 1998, 1640 [Russ. Chem. Bull., 1998,47, 1598 (Engl. Transl.)].
E. G. Rozantsev,Svobodnye iminoksil'nye radikaly [Iminoxyl Free Radicals], Khimiya, Moscow, 1970, p. 186, p. 200 (in Russian).
C. M. Giandomenico, M. J. Abrams, B. A. Murrer, J. F. Vollano, M. I. Rheinheimer, S. B. Wyer, G. E. Bossard, and J. D. Higgins,Inorg. Chem., 1995,34, 1015.
S. U. Dunham and S. J. Lippard,J. Am. Chem. Soc., 1995,117, 10702.
USSR Pat. 582481;Byull. Izobret. [Invent Bull.], 1977, 44 (in Russian).
T. R. Brent,Cancer Res., 1984,44, 1887.
T. G. Talman, W. Bruning, J. Reedijk, A. L. Spek, and N. Veldman,Inorg. Chem., 1997,36, 854.
H. Ito, J. Fujita, and K. Saito,Bull. Chem. Soc. Jpn., 1967,40, 2584.
T. V. Hambley,Coord. Chem. Rev., 1997,166, 181.
A. V. Shugalii, A. V. Kulikov, M. V. Lichina, V. A. Golubev, and V. D. Sen',J. Inorg. Biochem., 1998,69, 67.
N. P. Konovalova, R. F. Diatchkovskaya, L. H. Ganieva, L. M. Volkova, and A. B. Shapiro,Neoplasma, 1991,38, 275.
G. Sosnovsky and S. W. Li,Drug Future, 1985,36, 1379.
N. P. Konovalova, R. F. Diatchkovskaya, L. M. Volkova, and V. N. Varfolomeev,Anti-Cancer Drugs, 1991,2, 591.
K. Takechi, H. Tamura, K. Yamaoka, and H. Sakurai,Free Rad. Res., 1997,26, 483.
H. Masuda, T. Tanaka, and S. Matsushima,Anticancer Res., 1998,18, 1473.
Author information
Authors and Affiliations
Additional information
Published inIzvestiya Akademii Nauk. Seriya Khimicheskaya, No. 9, pp. 1624–1630, September, 2000.
Rights and permissions
About this article
Cite this article
Sen, V.D., Rukina, N.A., Tkachev, V.V. et al. Synthesis, structure, and biological activity of mixed-ligand platinum(II) complexes with aminonitroxides. Russ Chem Bull 49, 1613–1619 (2000). https://doi.org/10.1007/BF02495168
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02495168