Summary
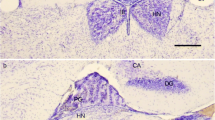
Synaptophysin (protein p38), a major integral membrane glycoprotein of small presynaptic vesicles, was localized immunohistochemically in semithin sections of the superficial pineal gland of the Mongolian gerbil (Meriones unguiculatus). Synaptophysin immunoreactivity could be detected in all pinealocytes, which were visualized with antibodies directed against neuron-specific enolase (NSE) in adjacent sections. No p38 immunoreactivity was discernible in the interstitial glial cells, which showed a heterogeneous pattern of immunostaining for the intermediate filament proteins glial fibrillary acidic protein (GFAP) and vimentin. Pinealocytes exhibited considerable intercellular differences in the densities of immunostaining. The various degrees of synaptophysin immunoreactivities in pinealocytes were not correlated with the densities of NSE immunostaining. Nerve terminals and varicosities displayed stronger immunoreactivities than pinealocytes. They were particularly numerous in the perivascular spaces. It is not clear whether this distribution indicates an innervation of pineal capillaries in addition to the functionally important innervation of pinealocytes. Several highly p38-positive dots of variable size were a conspicuous feature throughout the gland. By the consecutive semithin-thin section technique, they could be identified as processes of pinealocytes, filled with accumulations of small clear vesicles. Obviously, these vesicles represent the major site of synaptophysin immunoreactivity in pinealocytes. In the gerbil, similar vesicles have been ascribed a role in the secretory activity of the gland, and/or in the transport of calcium. The intercellular differences in the degrees of p38 immunostaining may, therefore, reflect different states of a specific cellular activity. The presence of synaptophysin in pinealocytes of the normal pineal, including the deep portions of the gland, emphasizes the paraneuronal character of these cells.
Similar content being viewed by others
References
Bishop AE, Carlei F, Lee V, Trojanowski J, Marangos PJ, Dahl D, Polak JM (1985) Combined immunostaining of neurofilaments, neuron specific enolase, GFAP and S-100. A possible means for assessing the morphological and functional status of the enteric nervous system. Histochemistry 82:93–97
Boeckmann D (1980) Morphological investigation of the deep pineal of the rat. Cell Tissue Res 210:283–294
Calvo J, Boya J, Borregon A, Garcia-Maurino JE (1988) Presence of glial cells in the rat pineal gland: a light and electron microscopic immunohistochemical study. Anat Rec 220:424–428
Collins VP (1987) Pineocytoma with neuronal differentiation demonstrated immunocytochemically. Acta Pathol Microbiol Immunol Scand [A] 95:113–117
Ehrhart M, Jörns A, Grube D, Gratzl M (1988) Cellular distribution and amount of chromogranin A in bovine endocrine pancreas. J Histochem Cytochem 36:467–472
Fujita T, Kanno T, Kobayashi S (1988) The paraneuron. Springer, Tokyo
Gould VE, Lee I, Wiedenmann B, Moll R, Chejfec G, Franke WW (1986) Synaptophysin: a novel marker for neurons, certain neuroendocrine cells, and their neoplasms. Hum Pathol 17:979–983
Grube D (1980) Immunoreactivities of gastrin (G-) cells. II. Nonspecific binding of immunoglobulins to G-cells by ionic interactions. Histochemistry 66:149–167
Grube D, Kusumoto Y (1986) Serial semithin sections in immunohistochemistry: Techniques and applications. Arch Histol Jpn 49:391–410
Haimoto H, Takahashi Y, Koshikawa T, Nagura H, Kato K (1985) Immunohistochemical localization of γ-enolase in normal human tissues other than nervous and neuroendocrine tissues. Lab Invest 52:257–263
Higley HR, McNulty JA, Rowden G (1984) Glial fibrillary acidic protein and S-100 protein in pineal supportive cells: an electron microscopic study. Brain Res 304:117–120
Hoog A, Gould VE, Grimelius L, Franke WW, Falkmer S, Chejfec G (1988) Tissue fixation methods alter the immunohistochemical demonstrability of synaptophysin. Ultrastruct Pathol 12:673–678
Hsu SM, Raine L, Fanger H (1981) Use of avidin-biotin-peroxidase complex (ABC) in immunoperoxidase techniques. A comparison between ABC and unlabeled antibody (PAP) procedures. J Histochem Cytochem 29:577–580
Jahn R, Schiebler W, Ouimet C, Greengard P (1985) A 38, 000-dalton membrane protein (p38) present in synaptic vesicles. Proc Natl Acad Sci USA 82:4137–4141
Japha JL, Eder TJ, Goldsmith ED (1974) Morphological and histochemical features of the gerbil pineal system. Anat Rec 178:381–382
Japha JL, Eder TJ, Goldsmith ED (1976) Calcified inclusions in the superficial pineal gland of the Mongolian gerbil,Meriones unguiculatus. Acta Anat 94:533–544
Juillard MT, Collin JP (1980) Pools of serotonin in the pineal gland of the mouse: the mammalian pinealocyte as a component of the diffuse neuroendocrine system. Cell Tissue Res 213:273–291
Kivelä T, Tarkkanen A, Virtanen I (1989) Synaptophysin in the human retina and retinoblastoma. An immunohistochemical and western blotting study. Invest Ophthalmol Vis Sci 30:212–219
Kuwano R, Iwanaga T, Nakajima T, Masuda T, Takahashi Y (1983) Immunocytochemical demonstration of hydroxyindole O-methyltransferase (HIOMT), neuron-specific enolase (NSE) and S-100 protein in the bovine pineal. Brain Res 274:171–175
Lowe AW, Madeddu L, Kelly RB (1988) Endocrine secretory granules and neuronal synaptic vesicles have three integral membrane proteins in common. J Cell Biol 106:51–59
Møller M (1985) Non-sympathetic synaptic innervation of the pinealocyte of the Mongolian gerbil (Meriones unguiculatus): an electron microscopic study. J Neurocytol 14:541–550
Møller M, Ingild A, Bock E (1978) Immunohistochemical demonstration of S-100 protein and GFA protein in interstitial cells of rat pineal gland. Brain Res 140:1–13
Navone F, Jahn R, DiGioia G, Stukenbrok H, Greengard P, De Camilli P (1986) Protein p38: an integral membrane protein specific for small vesicles of neurons and neuroendocrine cells. J Cell Biol 103:2511–2527
Nielsen JT, Møller M (1978) Innervation of the pineal gland in the Mongolian gerbil (Meriones unguiculatus). A fluorescence microscopical study. Cell Tissue Res 187:235–250
Obendorf D, Schwarzenbrunner U, Fischer-Colbrie R, Laslop A, Winkler H (1988) In adrenal medulla synaptophysin (protein p38) is present in chromaffin granules and in a special vesicle population. J Neurochem 51:1573–1580
Osborn M, Debus E, Weber K (1984) Monoclonal antibodies specific for vimentin. Eur J Cell Biol 34:137–143
Pearse AGE, Takor Takor T (1979) Embryology of the diffuse neuroendocrine system and its relationship to the common peptides. Fed Proc Fed Am Soc Exp Biol 38:2288–2294
Radke R, Stach W (1986) Are the islets of Langerhans neuroparaneuronal control centers of the exocrine pancreas? Arch Histol Jpn 49:411–420
Redecker P (1987) Golgi-like immunostaining of pituicytes and tanycytes positive for glial fibrillary acidic protein in the neurohypophysis of the Mongolian gerbil (Meriones unguiculatus). Histochemistry 87:585–595
Rehm H, Wiedenmann B, Betz H (1986) Molecular characterization of synaptophysin, a major calcium-binding protein of the synaptic vesicle membrane. EMBO J 5:535–541
Schachner M, Huang SK, Ziegelmüller P, Bizzini B, Taugner R (1984) Glial cells in the pineal gland of mice and rats. A combined immunofluorescence and electron-microscopic study. Cell Tissue Res 237:245–252
Scheuermann DW, Stach W, Timmermans J-P, Adriaensen D, De Groodt-Lasseel MHA (1989) Neuron-specific enolase and S-100 protein immunohistochemistry for defining the structure and topographical relationship of the different enteric nerve plexuses in the small intestine of the pig. Cell Tissue Res 256:65–75
Schilling K, Gratzl M (1988) Quantification of p38/synaptophysin in highly purified adrenal medullary chromaffin vesicles. FEBS Lett 233:22–24
Schilling K, Blanco Barco E, Rhinehart D, Pilgrim Ch (1989) Expression of synaptophysin and neuron-specific enolase during neuronal differentiation in vitro: effects of dimethyl sulfoxide. J Neurosci Res 24:347–354
Schmechel D, Brightman M, Goodwin F (1978) Brain enolases as specific markers of neuronal and glial cells. Science 199:313–315
Sternberger LA (1986) Immunocytochemistry (3rd edn). Wiley, New York
Südhof TC, Lottspeich F, Greengard P, Mehl E, Jahn R (1987) A synaptic vesicle protein with a novel cytoplasmic domain and four transmembrane regions. Science 238:1142–1144
Vollrath L, Schröder H (1987) Neuronal properties of mammalian pinealocytes? In: Trentini GP, De Gaetani C, Pevet P (eds) Fundamentals and clinics in pineal research, vol 44. Raven Press, New York, pp 13–23
Welsh MG (1984) Cytochemical analysis of calcium distribution in the superficial pineal gland of the Mongolian gerbil. J Pineal Res 1:305–316
Welsh MG, Reiter RJ (1978) The pineal gland of the gerbil,Meriones unguiculatus. I. An ultrastructural study. Cell Tissue Res 193:323–336
Welsh MG, Cameron IL, Reiter RJ (1979) The pineal gland of the gerbil,Meriones unguiculatus. II. Morphometric analysis over a 24-hour period. Cell Tissue Res 204:95–109
Wiedenmann B, Franke WW (1985) Identification and localization of synaptophysin, an integral membrane glycoprotein of M 38, 000 characteristic of presynaptic vesicles. Cell 41:1017–1028
Wiedenmann B, Franke WW, Kuhn C, Moll R, Gould VE (1986) Synaptophysin: a marker protein for neuroendocrine cells and neoplasms. Proc Natl Acad Sci USA 83:3500–3504
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Redecker, P., Grube, D. & Jahn, R. Immunohistochemical localization of synaptophysin (p38) in the pineal gland of the Mongolian gerbil (Meriones unguiculatus). Anat Embryol 181, 433–440 (1990). https://doi.org/10.1007/BF02433790
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02433790