Abstract
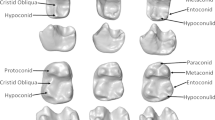
Marmosets have reduced second molars of which size and shape are varied in different species. Mesiodistal and buccolingual diameter of the first and second upper molars in 16 species were measured, and molar area (molar rectangle) and M2 reduction index were calculated by the equation,R=(M2 area/M1 area) × 100. This index ranged from 36.1 inSaguinus oedipus geoffroyi to 70.6 which was the largest found inCebuella pygmaea. Species ofSaguinus showed relatively wide variation as well as a consistantly smaller index. The index forCallithrix registered around 60 andLeontopithecus rosalia was positioned within this genus. There was no relationship between this index and body size of each form. The sizes of the first molar and second molar may not significantly correlate either with body size across species, becauseLeontopithecus rosalia had exceptionally large molars for its body size and contrarily genusSaguinus had relatively small molars. When the shape of the mandible was expressed as length/width ratio, the reduction index significantly correlate with this ratio in genusSaguinus andCallithrix, indicating that longer jaw in shape had relatively large M2. The reduction indices of two possible subspecies,S. oedipus geoffroyi andS. o. oedipus, were 36.1 and 47.3, respectively. This difference suggested that there was a difference in diet or function of jaw apparatus beyond subspecies level.
Similar content being viewed by others
References
Du Brul, E. L., 1965. The skull of the lion marmosetLeontideus rosalia Linnaeus: A study in biomechanical adaptation.Amer. J. Phys. Anthropol., 23: 261–276.
Eaglen, R. H., 1986. Morphometrics of the anterior dentition in strepsirhine primates.Amer. J. Phys. Anthropol., 71: 185–201.
Gingerich, P. D., B. H. Smith, &K. Rosenberg, 1982. Allometric scaling in the dentition of primates and prediction of body weight from tooth size in fossils.Amer. J. Phys. Anthropol., 58: 81–100.
Hanihara, T. &M. Natori, 1987. A preliminary analysis for numerical taxonomy of the genusSaguinus.Primates, 28: 517–523.
Hershkovitz, P., 1977.Living New World Monkeys (Platyrrhini) with an Introduction to Primates, Vol. 1. Univ. of Chicago Press, Chicago.
Hylander, W. L., 1985. Mandibular function and biomechanical stress and scaling.Amer. Zool., 25: 315–330.
Inoue, N., 1980. Tooth to denture base discrepancy in human evolution.J. Anthropol. Soc. Nippon, 88: 69–82. (in Japanese with English summary)
Kanazawa, E. & A. L. Rosenberger, 1987. Interspecific allometry of the mandible, dental arch and molar area in primates. (unpublished data)
Kay, R. F., 1975. The functional adaptations of primate molar teeth.Amer. J. Phys. Anthropol., 43: 195–216.
Kurtén, B., 1967. Some quantitative approaches to dental microevolution.J. Dent. Res., 46: 817–828.
Mizoguchi, Y., 1983. Influences of the earlier developing teeth upon the later developing teeth.Bull. Nation. Sci. Mus., Tokyo, Ser. D, 9: 33–45.
Mucci, R. J., 1982. The role of attrition in the etiology of third molar impactions: confirming the Begg hypothesis. Thesis, Univ. of Illinois, Chicago.
Ozaki, T., 1960. The reduction index of the Japanese teeth.Acta Anat. Nippon, 35: 563–577. (in Japanese)
Robinson, J. T., 1954. Prehominid dentition and hominid evolution.Evolution, 8: 324–334.
Rosenberger, A. L. &A. F. Coimbra-Filho, 1984. Morphology, taxonomic status and affinities of the lion tamarins,Leontopithecus (Callitrichinae, Cebidae).Folia Primatol., 42: 149–179.
Setoguchi, T. &A. L. Rosenberger, 1987. A fossil owl monkey from La Venta, Columbia.Nature, 326: 692–694.
Sofaer, J. A., 1973. A model relating developmental interaction and differential evolutionary reduction of tooth size.Evolution, 27: 427–434.
Author information
Authors and Affiliations
About this article
Cite this article
Kanazawa, E., Rosenberger, A.L. Reduction index of the upper M2 in marmosets. Primates 29, 525–533 (1988). https://doi.org/10.1007/BF02381139
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02381139