Abstract
Protein kinase C (PKC) is a mediator of transmembrane signal transduction, important in cell growth and differentiation. Cell activation by extracellular signals is associated with a translocation of PKC from the cytosol to the membrane. We measured and compared PKC activity in cytosol and membrane fractions of normal and neoplastic colorectal tissue. Total and membrane-associated PKC activity in normal colorectal tissue was greater in patients (N=16) with colorectal cancer compared to that from patients with a normal colonoscopy (N=16),P<0.01. A similar trend was noted in PKC activity of normal colorectal tissue from patients with adenomas compared to patients with a normal colonoscopy. PKC activity (total, membrane-associated, percent membrane) was not different in neoplastic colorectal tissue compared to that of adjacent normal tissue. However, there was a considerable range of PKC activity noted in all groups, which would limit the utility of PKC activity as a marker for colorectal neoplasia.
Similar content being viewed by others
References
Nishizuka Y: Studies and perspectives of protein kinase C. Science 233:305–312, 1986
Ashendel CL: The phorbol ester receptor: A phospholipid regulated protein kinase. Biochem Biophys Acta 822:219–242, 1985
Craven PA, DeRubertis FR: Role of activation of protein kinase C in the stimulation of colonic epithelial proliferation by unsaturated fatty acids. Gastroenterology 95:676–685, 1988
Craven PA, Pfanstiel J, DeRubertis FR: Role of activation of protein kinase C in the stimulation of colonic epithelial proliferation and reactive oxygen formation by bile acids. J Clin Invest 79:532–541, 1987
Fitzer CJ, O'Brian CA, Guillem JG, Weinstein IB: The regulation of protein kinase C by chenodeoxycholate, deoxycholate and several structurally related bile acids. Carcinogenesis 8:217–220, 1987
Baum CL, Wali RK, Sitrin MD, Bolt MJG, Brasitus TA: 1,2-Dimethylhydrazine-induced alterations in protein kinase C activity in the rat preneoplastic colon. Cancer Res 50:3915–3920, 1990
Muto T, Bussey HJR, Morson BC: The evolution of cancer of the colon and rectum. Cancer 36:2251–2270, 1975
Boland CR, Montgomery CK, Kim YS: Alterations in colonic mucin occurring with cellular differentiation and malignant transformation. Proc Natl Acad Sci USA 79:2051, 1982
Luk GD, Baylin SB: Ornithine decarboxylase as a biologic marker in familial colonic polyposis. N Engl J Med 311:80–83, 1984
Maskens AP, Deschner EE: Tritiated thymidine incorporation into epithelial cells of normal-appearing colorectal mucosa of cancer patients. J Natl Cancer Inst 58:1221–1224, 1977
McGarrity TJ, Peiffer LP, Bartholomew MJ, Pegg AE: Colonic polyamine content and ornithine decarboxylase activity as markers for adenomas. Cancer 66:1539–1543, 1990
Bradford MA: A rapid and sensitive technique for the quantification of microgram amounts of protein utilizing the principle of protein dye binding. Anal Biochem 72:248–254, 1976
Kraft AS, Anderson WB, Cooper HL, Sando JJ: Decrease in cytosolic calcium/phospholipid-dependent protein kinase activity following phorbol ester treatment of EL4 thymoma cells. J Biol Chem 22:13193–13196, 1982
Craven PA, DeRubertis FR: Alterations in protein kinase C in 1,2-dimethylhydrazine induced colonic carcinogenesis. Cancer Res 52:2216–2221, 1992
Kopp R, Noelka B, Sauter G, Schildberg FW, Paumgartner G, Pfeiffer A: Altered protein kinase C activity in biopsies of human colonic adenomas and carcinomas. Cancer Res 51:205–210, 1991
Hashimoto Y, Chida K, Huang M, Katayama M, Nishihira T, Kuroki T: Levels of protein kinase C activity in human gastrointestinal cancers. Biochem Biophys Res Commun 163:406–411, 1989
Guillem JG, O'Brian CA, Fitzer CJ, Forde KA, LoGerfo P, Treat M, Weinstein IB: Altered levels of protein kinase C and Ca+2-dependent protein kinases in human colon carcinoma. Cancer Res 47:2036–2039, 1987
Kusunoki M, Sakanoue Y, Hatada T, Yanagi H, Yamamura T, Utsunomiya J: Protein kinase C activity in human colonic adenoma and colorectal carcinoma. Cancer 69:24–30, 1992
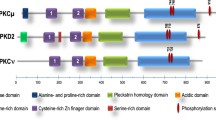
Parker PJ, Kour G, Marais RM, Mitchell F, Pears C, Schaap D, Stabel S, Webster C: Protein kinase C—a family affair. Mol Cell Endocrinol 65:1–11, 1989
Coussens L, Parker PJ, Rhee L, Yang-Feng TL, Chen E, Waterfield MD, Francke U, Ulrich A: Multiple distinct forms of bovine and human protein kinase C suggest diversity in cellular signaling pathways. Science 233:859–866, 1986
Pelosin J-M, Keramidas M, Souvignet C, Chambaz EM: Differential inhibition of protein kinase C subtypes. Biochem Biophys Res Commun 169:1040–1048, 1990
Kubo K, Ohno S, Suzuki K: Primary structures of human protein kinase C βI and βII differ only in their C-terminal sequences. FEBS Lett. 223:138–142, 1987
Clark EA, Leach KL, Trojanowski JQ, Lee VM-Y: Characterization and differential distribution of the three major human protein kinase C isozymes (PKCαm, PKCβ, and PKCγ) of the central nervous system in normal and Alzheimer's disease brains. Lab Invest 64:35–44, 1991
Sekiguchi K, Tsukuda M, Ogita K, Kikkawa U, Nishizuka Y: Three distinct forms of rat brain protein kinase C: Differential response to unsaturated fatty acids. Biochem Biophys Res Commun 145:797–802, 1987
Author information
Authors and Affiliations
Additional information
Supported by grant R29 CA45468 (T.J.M.) from the National Institutes of Health.
Rights and permissions
About this article
Cite this article
McGarrity, T.J., Peiffer, L.P. Protein kinase C activity as a potential marker for colorectal neoplasia. Digest Dis Sci 39, 458–463 (1994). https://doi.org/10.1007/BF02088328
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02088328