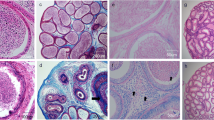
Abstract
Previous studies have shown that differential susceptibility to actively induced experimental allergic orchitis (EAO) exists among various BALB/c substrains. Of 13 substrains studied, BALB/cJ mice consistently exhibit greater resistance to disease induction. Such resistance is associated with a single recessive genotypic difference in an immunoregulatory locus which is unlinked to any of the known alleles distinguishing the BALB/cJ substrain. In this study, gene complementation protocols were used to study the genetics of susceptibility and resistance to EAO. The results indicate that resistance in BALB/cJ mice is not due to a mutation in theH-2D d linked gene which governs the phenotypic expression of autoimmune orchitis. The mechanistic basis for disease resistance was examined using reciprocal bone marrow radiation chimeras generated between the disease-susceptible BALB/ cByJ (ByJ) substrain and BALB/cJ (Jax) mice. All constructs, including Jax →- Jax and Jax →- ByJ, developed severe EAO following inoculation with mouse testicular homogenate (MTH) and adjuvants whereas control chimeras immunized with adjuvants alone did not. These results suggest that an active immunoregulatory mechanism rather than a passive one, such as the lack of T cells and/or B cells with receptors for the aspermatogenic autoantigens relevant in the induction of EAO, is responsible for disease resistance in BALB/cJ mice. The role of immunoregulatory cells was examined by pretreating BALB/cJ mice with either cyclophosphamide (20 mg/kg) or low-dose whole body or total lymphoid irradiation (350 rads) 2 days prior to inoculation. BALB/cJ mice immunized with MTH plus adjuvants generate immunoregulatory spleen cells (SpCs) that, when transferred to naive BALB/cByJ recipients, significantly reduce the severity of autoimmune orchitis observed during actively induced EAO. Treatment of such cells with either cytotoxic monoclonal anti-Thy-1.2 or anti-CD4 plus C' before transfer abrogates the ability of BALB/cJ spleen cells to inhibit disease. In contrast, neither SpCs from adjuvantimmunized BALB/cJ nor MTH plus adjuvant-primed BALB/cByJ donors significantly influenced the severity of disease observed in recipients. Taken together, these results suggest that genetically controlled resistance to EAO in BALB/cJ mice is associated with a mutation in an immunoregulatory locus whose effects appear to be mediated through a cyclophosphamide and low-dose radiation-sensitive CD4+ T-cell population.
Similar content being viewed by others
References
Arnon, R.: Experimental allergic encephalomyelitis-susceptibility and suppression.Immunol Rev 55: 5–30, 1981
Bernard, C. C. A.: Experimental autoimmune encephalomyelitis in mice: genetic control of susceptibility.J Immunogenet 3: 263–274, 1976
Bernard, C. C. A.: Suppressor T cells prevent experimental autoimmune encephalomyelitis in mice.Clin Exp Immunol 29: 100–109, 1977
Bruce, J., Symington, F. W., McKearn, T. J., and Sprent, J.: A monoclonal antibody discriminating between subsets of T and B cells.J Immunol 127: 2496–2501, 1981
Ceredig, R., Lowenthal, J. W., Nabholz, M., and MacDonald, H. R.: Expression of interleukin-2 receptors as a differentiation marker on intrathymic stem cells.Nature 314: 98–100, 1985
Chiorazzi, N., Fox, D. A., and Katz, D. H.: Hapten-specific IgE antibody responses in mice. VI. Selective enhancement of IgE antibody production by low doses of X-irradiation and by cyclophosphamide.J Immunol 117: 1629–1637, 1976
Chiorazzi, N., Fox, D. A., and Katz, D. H.: Hapten specific IgE antibody responses in mice. VII. Conversion of IgE “nonresponder” strains to IgE “responders” by elimination of suppressor T-cell activity.J Immunol 118: 48–54, 1977
Ellerman, K. E., Powers, J. M., and Brostoff, S. W.: A suppressor Tlymphocyte cell line for autoimmune encephalomyelitis.Nature 331: 265–267, 1988
Hall, B. M., Pearce, N. W., Gurley, K. E., and Dorsch, S. E.: Specific unresponsiveness in rats with prolonged cardiac allograft survival after treatment with cyclosporine. III. Further characterization of the CD4+ suppressor cell and its mechanisms of action.J Exp Med 171: 141–157, 1990
Hickey, W. F., Kirby, W. M., and Teuscher, C.: BALB/c substrain differences in susceptibility to experimental allergic encephalomyelitis (EAE).Ann NY Acad Sci 475: 331–333, 1986
Hiramine, C. and Hojo, K.: Adoptive transfer of suppression of experimental allergic orchitis with lymphoid cells from antigenpretreated guinea pigs.Clin Immunol Immunopathol 30: 202–213, 1984
Janeway, C. A., Jr., Carding, S., Jones, B., Murray, J., Portoles, P., Rasmussen, R., Rojo, J., Saizawa, K., West, J., and Bottomly, K.: CD4+ T cells: specificity and function.Immunol Rev 101: 39–80, 1988
Karpus, W. J. and Swanborg, R. H.: CD4+ suppressor cells differentially affect the production of IFN-γ by effector cells of experimental autoimmune encephalomyelitis.J Immunol 143: 3492–3497, 1989
Kohno, S., Munoz, J. J., Wiliams, T. M., Teuscher, C., Bernard, C. C. A., and Tung, K. S. K.: Immunopathology of murine experimental allergic orchitis.J Immunol 130: 2675–2682, 1983
Kong, Y. M., Giraldo, A. A., Waldmanm, H., Cobbold, S. P., and Fuller, B. E.: Resistance to experimental autoimmune thyroiditis: L3T4- cells as mediators of both thyroglobulin-activated and TSH induced suppression.Clin Immunol Immunopathol 51: 38–54, 1989
Korngold, R., Feldman, A., Rorke, L. B., Lublin, F. D., and Doherty, P. C.: Acute experimental allergic encephalomyelitis in radiation bone marrow chimeras between high and low susceptible strains of mice.Immunogenetics 24: 309–315, 1986
Lando, Z., Teitelbaum, D., and Arnon, R.: Genetic control of susceptibility to experimental allergic encephalomyelitis in mice.Immunogenetics 9: 435–442, 1979a
Lando, Z., Teitelbaum, D., and Arnon, R.: Effect of cyclophosphamide on suppressor cell activity in mice unresponsive to EAE.J Immunol 123: 2156–2160, 1979b
Lando, Z., Teitelbaum, D., and Arnon, R.: Induction of experimental allergic encephalomyelitis in genetically resistant strains of mice.Nature 287: 551–552, 1980
Linthicum, D. S. and Frelinger, J. A.: Acute autoimmune encephalomyelitis in mice. II. Susceptibility is controlled by the combination of H-2 and histamine sensitization genes.J Exp Med 156: 31–40, 1982
Mahi-Brown, C. A. and Tung, K. S. K.: Susceptibility to experimental autoimmune orchitis (EAO) can be adoptively transferred from responder to non-responder substrains of BALB/c mice.Biol Reprod 38 (Suppl 1): 82, 1988
Mahi-Brown, C. A. and Tung, K. S. K.: Activation requirements of donor T cells and host T cell recruitment in adoptive transfer of murine experimental autoimmune orchitis (EAO).Cell Immunol 124: 368–379, 1989
Mahi-Brown, C. A., Yule, T. D., and Tung, K. S. K.: Adoptive transfer of marine autoimmune orchitis to naive recipients with immune lymphocytes.Cell Immunol 106: 408–419, 1987
McDonald, A. H. and Swanborg, R. H.: Antigen-specific inhibition of immune interferon production by suppressor cells of autoimmune encephalomyelitis.J Immunol 140: 1132–1138, 1988
Mossman, T. R. and Coffman, R. L.: TH1 and TH2 cells: different patterns of lymphokine secretion lead to different functional properties.Annu Rev Immunol 7: 145–173, 1989
Munoz, J. J. and Mackay, I. R.: Production of experimental allergic encephalomyelitis with the aid of pertussigen in mouse strains considered genetically resistant.J Neuroimmunol 7: 91–96, 1984
Padberg, W. M., Kupiec-Weglinski, J. W., Lord, R. H. H., Araneda, D. H., and Tilney, N. L.: W3/25+ T cells mediate the induction of immunologic unresponsiveness in enhanced rat recipients of cardiac allografts.J Immunol 138: 3669–3674, 1987
Potter, M.: History of the BALB/c family.Curr Top Microbiol Immunol 122: 1–5, 1985
Sarmiento, M., Glasebrook, A. L., and Fitch, F. W.: IgG or IgM monoclonal antibodies reactive with different determinants on the molecular complex bearing Lyt antigen block T-cell-mediated cytolysis in the absence of complement.J Immunol 125: 2665–2672, 1980
Sprent, J., Schaefer, M., Gao, E.-K., and Korngold, R.: Role of T cell subsets in lethal graft-versus-host disease (GVHD) directed to class I versus class II H-2 differences. I. L3T4÷ cells can either augment or retard GVHD elicited by Lyt-2+ cells in class I-different hosts.J Exp Med 167: 556–569, 1988
Teuscher, C.: Experimental allergic orchitis in mice. II. Association of disease susceptibility with the locus controllingBordetella pertussis-induced sensitivity to histamine.Immunogenetics 22: 417–425, 1985
Teuscher, C., Potter, M., and Tung, K. S. K.: Differential susceptibility to experimental autoimmune orchitis in BALB/c substrains.Curr Top Microbiol Immunol 122: 181–188, 1985a
Teuscher, C., Smith, S. M., Goldberg, E. H., Shearer, G. M., and Tung, K. S. K.: Experimental allergic orchitis in mice. I. Genetic control of susceptibility and resistance to induction of autoinunune orchitis.Immunogenetics 22: 323–333, 1985b
Teuscher, C., Smith, S. M., and Tung, K. S. K.: Experimental allergic orchitis in mice. III. Differential susceptibility and resistance among BALB/c sublines.J Reprod Immunol 10: 219–230, 1987a
Teuscher, C., Blankenhorn, E. P., and Hickey, W. F.: Differential susceptibility to actively induced experimental allergic encephalomyelitis and experimental allergic orchitis among BALB/c substrains.Cell Immunol 110: 294–304, 1987b
Teuscher, C., Blankenhorn, E. P., and Hickey, W. F.: Genetic analysis of BALB/cJ subline resistance to actively induced experimental allergic orchitis (EAO) and experimental allergic encephalomyelitis (EAE).Curr Top Microbiol Immunol 137: 233–239, 1988
Tite, J. P., Foellmer, H. G., Madri, J. A., and Janeway, C. A., Jr.: Inverse Ir gene control of antibody and T cell proliferative responses to human basement membrane collagen.J Immunol 139: 2892–2898, 1987
Yule, T. D., Montoya, G. D., Russell, L. D., Williams, T. M., and Tung, K. S. K.: Autoantigenic germ cells exist outside the blood testis barrier.J Immunol 141: 1161–1167, 1988
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Teuscher, C., Hickey, W.F. & Korngold, R. Experimental allergic orchitis in mice. V. Resistance to actively induced disease in BALB/cJ substrain mice is mediated by CD4 + T cells. Immunogenetics 32, 34–40 (1990). https://doi.org/10.1007/BF01787326
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01787326