Summary
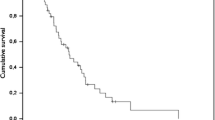
The object of this study is to evaluate the efficacy of a high dose of carboplatin in 20 patients operated on for high grade glioma (Group A) compared with a matched control (Group B) treated with BCNU administered after radiotherapy. The toxicity profile has been evaluated during the therapy. The survival of patients entering this study was measured in terms of months: the mean survival time was 10.45 months and the median 11.0 months in the — group treated with carboplatin (8 patients are still alive); the 18-month survival rate was 10%. The mean survival time of the control group was 9.85 months and the median 10.5 months; no patients are still alive and the 18-month survival rate was 0%. On the basis of our phase II clinical study, we could conclude that i.v. administration of high-doses of carboplatin in high grade gliomas is generally well tolerated and the results are better than those of a matched control treated with 1–2 courses of BCNU (low-dose). The adjuvant treatment and the role of carboplatin in the therapy of high grade gliomas is discussed.
Similar content being viewed by others
References
Aida T, Bodell WJ (1987) Cellular resistance to chlorethylnitro-soureas, nitrogen mustard and cis-diamrninedichloroplatinum II) in human glial-derived cell lines. Cancer Res 47: 1361–1366
Brufman G, Halpern J, Sukes R, Catane R, Biran S (1984) Procarbazine, CCNU, and vincristine combination chemotherapy for brain tumours. Oncology 41: 239–241
Cianfriglia F, Pompili A, Riccio A, Grassi A (1980) CCNU-chemotherapy of hemispheric supratentorial glioblastoma multiforme. Cancer 45: 1289–1299
Doy F, Berens ME, Dougherty DV, Rosenblum ML (1991) Comparison of the cytotoxic activities of cisplatin and carboplatin against gliomas cell lines at pharmalogically relevant drug exposure. J Neurooncol 11: 27–35
EORTC Brain Tumor Group (1978) Effect of CCNU on survival rate of objective remission and duration of free interval in patients with malignant brain glioma-final evaluation. Eur J Cancer 14: 851–856
EORTC Brain Tumor Group (1985) Evaluation of AZQ (1,4-cyclohexadiene-1,4-diacarbamic acid-2,5-bis(l-aziridinyl)-3,6-dioxodiethylester) in recurring supratentorial malignant brain gliomas — a phase II study. Eur J Cancer Clin Oncol 21: 143–146
EORTC Brain Tumor Group (1981) Evaluation of CCNU, VM26, plus CCNU, and procarbazine in supratentorial brain gliomas. Final evaluation of a randomized study. J Neurosurg 55: 27–31
Hochberg FD, Pruitt AA, Beck DO, DeBrun G, Davis K (1985) The rationale and the methodology for intra-arterial chemotherapy with BCNU as a treatment for glioblastoma. J Neurosurg 63: 876–880
Kaplan EL, Meier P (1958) Nonparametric estimation from incomplete observation. J Am Stat Assoc 53: 457–481
Kornblith P, Walker M (1988) Chemotherapy for malignant gliomas. J Neurosurg 68: 1–17
Kumar ARV, Renaudin J, Wilson CB, Bodrey EB, Enot KJ, Levin VA (1974) Procarbazine hydrochloridein the treatment of brain tumors. A phase II study. J Neurosurg 40: 365–371
Levin A, Wilson CB, Davis R (1979) A phase III comparison of BCNU, hydroxyurea, and radiation therapy to BCNU and radiation therapy for treatment of primary malignant gliomas. J Neurosurg 51: 526–532
Mahaley MS, Hipp SW, Dropcho EJ, Bertsch L, Cush S, Tirey T, Gillespie GY (1989) Intracarotid cisplatin chemotherapy for recurrent glioma. J Neurosurg 70: 371–378
Miller AB, Hoogstraten B, Staquet M, Winkler A (1981) Reporting results of cancer treatment. Cancer 47: 207–214
Muggia FM (1989) Overview of carboplatin. Replasing, complementing and extending the therapeutic horizons of cisplatin. Semin Oncol 16 [Suppl 5]: 7–13
Papavero L, Loew F, Jakche H (1987) Intracarotid infusion of ACNU and BCNU as adjuvant therapy of malignant gliomas. Acta Neurochir (Wien) 85: 128–137
Poisson M, Pereon Y, Chiras J, Delattre JY (1991) Treatment of recurrent malignant supratentorial gliomas with carboplatin (CBDCA). J Neurooncol 10: 139–144
Spence AM, Berger MS, Livingston RB (1992) Phase II evaluation of high dose intravenous cisplatin for the treatment of adult malignant gliomas recurrent after chloroethylnitrosurea failure. I Neurooncol 12: 187–191
Stewart DJ, O'Bryan RM, Al-Sarraf M (1983) Phase II study of cisplatin in recurrent astrocytomas in adults. A Southwest Oncology Group Study. J Neurooncol 1: 145–147
Twelves CJ, Ash CM, Miles DW, Thomas DG, Souhami RL (1991) Activity and toxicity of carboplatin and iproplatin in relapsed high-grade glioma. Cancer Chemother Pharmacol 27: 481–483
Wagstaff A, Ward A, Benfield P, Heel RC (1989) Carboplatin. A preliminary review of its pharmacodynamic and pharmacokinetic properties and therapeutic efficacy in the treatment of cancer. Drugs 37: 162–190
Walker MD, Green SB, Byar DP, Alexander E Jr, Batzdorf U, Hunt WE, MacCarty CS, Mahaley SM Jr, Mealey J Jr, Owens G, Ransohoff J II, Robertson JT, Shapiro WR, Smith KR, Wilson CB, Strike TA (1980) Randomized comparisons of radiotherapy and nitrosureas for the treatment of malignant glioma after surgery. N Engl J Med 303: 1323–1329
Walker WK, Mechtler L, Gleason MJ (1987) Intravenous carboplatin for recurrent glioma with carboplatin (CBDCA). Proc Am Soc Clin Oncol 6: 72
Warnick REet al (1994) A phase II study of intravenous carboplatin for the treatment of recurrent gliomas. J Neurooncol 19: 69–74
World Health Organization (1979) Handbook for Reporting Results of Cancer Treatment. WHO Offset publication No. 48, Geneva
Yung WK, Mechtler L, Gleason MJ (1991) Intravenous carboplatin for recurrent malignant glioma. A phase II study. J Clin Oncol 9: 860–864
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lunardi, P., Osman Farah, J., Mastronardi, L. et al. Intravenous administration of high doses of carboplatin in multimodal treatment of high grade gliomas: a phase II study. Acta neurochir 138, 215–220 (1996). https://doi.org/10.1007/BF01411364
Issue Date:
DOI: https://doi.org/10.1007/BF01411364