Summary
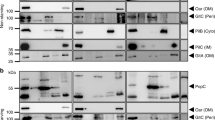
The TonB protein is involved in energy-coupled receptor-dependent transport processes across the outer membrane. The TonB protein is anchored in the cytoplasmic membrane but exposed to the periplasmic space. To fulfill its function, it has to couple the energy-providing metabolism in the cytoplasmic membrane with regulation of outer membrane receptor activity. Ferrichrome and albomycin transport, uptake of colicin M, and infection by the phages T1 andϕ80 occur via the same receptor, the FhuA protein in the outer membrane. Therefore, this receptor is particularly suitable for the study of energy-coupled TonB-dependent transport across the outer membrane. Ferrichrome, albomycin and colicin M bind to the FhuA receptor but are not released into the periplasmic space of unenergized cells, ortonB mutants. In vivo interaction between FhuA and TonB is suggested by the restoration of activity of inactive FhuA proteins, bearing amino acid replacements in the TonB box, by TonB derivatives with single amino acid substitutions. Point mutations in thefhuA gene are suppressed by point mutations in thetonB gene. In addition, naturally occurring degradation of the TonB protein and its derivatives is preferentially prevented in vivo by FhuA and FhuA derivatives where functional interaction takes place. It is proposed that in the energized state, TonB induces a conformation in FhuA which leads to the release of the FhuA-bound compounds into the periplasmic space. Activation of FhuA by TonB depends on the ExbBD proteins in the cytoplasmic membrane. They can be partially replaced by the TolQR proteins which show strong sequence similarity to the ExbBD proteins. A physical interaction of these proteins with the TonB protein is suggested by TonB stabilization through ExbB and TolQR. We propose a permanent or reversible complex in the cytoplasmic membrane composed of the TonB protein and the ExbBD/TolQR proteins through which TonB is energized.
Similar content being viewed by others
References
Bassford PJ Jr, Kadner RJ (1977) Genetic analysis of components involved in vitamin B12 uptake intonB mutants ofEscherichia coli. J Bacteriol 132:796–805
Bell PE, Nau CD, Brown JT, Konisky J, Kadner RJ (1990) Genetic suppression demonstrates interaction of TonB protein with outer membrane transport proteins inEscherichia coli. J Bacteriol 172:3826–3829
Bourdineaud JP, Howard SP, Lazdunski C (1989) Localization and assembly into theEscherichia coli envelope of a protein required for entry of colicin A. J Bacteriol 171:2458–2465
Braun V (1989) The structurally relatedexbB andtolQ genes are interchangeable in conferringtonB-dependent colicin, bacteriophage, and albomycin sensitivity. J Bacteriol 171:6387–6390
Braun V, Frenz S, Hantke K, Schaller K (1980) Penetration of colicin M into cells ofEscherichia coli. J Bacteriol 142:162–168
Curtis NAC, Eisenstadt RL, East SJ, Comford RJ, Walker LA, White AJ (1988) Iron-regulated outer membrane proteins ofEscherichia coli K12 and mechanism of action of catechol-substituted cephalosporins. Antimicrob Agents Chemother 32:1879–1886
Eick-Helmerich K, Braun V (1989) Import of biopolymers intoEscherichia coli: nucleotide sequences of theexbB andexbD genes are homologous to those of thetolQ andtolR genes, respectively. J Bacteriol 171:5117–5126
Fischer E, Günter K, Braun V (1989) Involvement of ExbB and TonB in transport across the outer membrane ofEscherichia coli: phenotypic complementation ofexbB mutants by overexpressedtonB and physical stabilization of TonB by ExbB. J Bacteriol 171:5127–5134
Günter K, Braun V (1990) In vivo evidence for FhuA outer membrane receptor interaction with the TonB inner membrane protein ofEscherichia coli. FEBS Lett. 274:85–88
Hancock REW, Braun V (1976) Nature of the energy requirement for the irreversible adsorption of bacteriophages T1 andϕ80 toEscherichia coli. J Bacteriol 125:309–415
Hantke K (1990) Dihydroxybenzolserine — a siderophore forE. coli. FEMS Microbiol Lett 67:5–8
Hantke K, Braun V (1978) Functional interaction of thetonA/tonB receptor system inEscherichia coli. J Bacteriol 135:190–197
Hantke K, Zimmermann L (1981) The importance of theexbB gene for vitamin B12 and ferric iron transport. FEMS Microbiol Lett 12:31–35
Hartmann A, Braun V (1980) Iron transport inEscherichia coli: uptake and modification of ferrichrome. J Bacteriol 143:246–255
Heller K, Kadner RJ, Günter K (1988) Suppression of thebtuB451 mutation by mutations in thetonB gene suggests a direct interaction between TonB and TonB-dependent receptor proteins in the outer membrane ofEscherichia coli. Gene 64:147–153
Hussein S, Hantke K, Braun V (1981) Citrate-dependent iron transport system inEscherichia coli K12. Eur J Biochem 117:431–437
Köster W, Braun V (1990) Iron(III) hydroxamate transport inEscherichia coli: substrate binding to the periplasmic FhuD protein. J Biol Chem 265:Dec. issue
Lugtenberg B, van Alphen L (1983) Molecular architecture and functioning of the outer membrane ofEscherichia coli and other Gram-negative bacteria. Biochim Biophys Acta 737:51–115
Mende J, Braun V (1990) Import-defective colicin B derivatives mutated in the TonB box. Mol Microbiol 4:1523–1533
Plastow GS, Holland IB (1979) Identification of anEscherichia coli inner membrane polypeptide specified by aλ-tonB transducing bacteriophage. Biochem Biophys Res Commun 90:1007–1014
Postle K, Good R (1983) DNA sequence of theEscherichia coli tonB gene. Proc Natl Acad Sci USA 80:5235–5239
Postle K, Skare JT (1988)Escherichia coli TonB protein is exported from the cytoplasm without proteolytic cleavage of its amino terminus. J Biol Chem 263:11000–11007
Pugsley AP, Zimmermann W, Wehrli W (1987) Highly efficient uptake of a rifamycin derivative via the FhuA/TonB-dependent uptake route inEscherichia coli. J Gen Microbiol 133:3505–3511
Roos U, Harkness RE, Braun V (1989) Assembly of colicin genes from a few DNA fragments. Nucleotide sequence of colicin D. Mol Microbiol 3:891–902
Schneider R, Hartmann A, Braun V (1981) Transport of the iron ionophore ferrichrome inEscherichia coli K12 andSalmonella typhimurium LT2. FEMS Microbiol Lett 11:115–119
Schöffler H, Braun V (1989) Transport across the outer membrane ofEscherichia coli via the FhuA receptor is regulated by the TonB protein of the cytoplasmic membrane. Mol Gen Genet 217:378–383
Sun TP, Webster RE (1987) Nucleotide sequence of a gene cluster involved in entry of E colicins and single-stranded DNA of infecting filamentous bacteriophages intoEscherichia coli. J Bacteriol 169:2667–2674
Tabor S, Richardson CC (1985) A bacteriophage T7 RNA polymerase/promoter system for controlled exclusive expression of specific genes. Proc Natl Acad Sci USA 82:1074–1078
Van hove B, Staudenmaier H, Braun V (1990) Novel two-component transmembrane transcription control: regulation of iron dicitrate transport ofEscherichia coli K12. J Bacteriol 172:Dec. issue
Watanabe NA, Nagasu T, Katsu K, Kitoh K (1987) E-0702, a new cephalosporin, is incorporated intoEscherichia coli cells via thetonB-dependent iron transport system. Antimicrob Agents Chemother 31:497–504
Zimmermann L, Hantke K, Braun V (1984) Exogenous induction of the iron dicitrate transport system ofEscherichia coli K12. J Bacteriol 159:271–277
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Braun, V., Günter, K. & Hantke, K. Transport of iron across the outer membrane. Biol Metals 4, 14–22 (1991). https://doi.org/10.1007/BF01135552
Issue Date:
DOI: https://doi.org/10.1007/BF01135552