Abstract
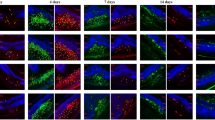
• Background: Although the choroidal neovascularization (CNV) is a common pathologic feature of a number of different eye diseases, its pathological mechanisms have not been fully elucidated. We investigated the expression of vascular endothelial growth factor (VEGF) in CNV using an experimental primate model. • Method: CNV was induced by intense laser photocoagulation in four monkey eyes. Single eyes were enucleated at 1, 3, 7 or 14 days after photocoagulation and examined immunohistochemically for VEGF, macrophage antigen, von Willebrand factor and glial fibrillary acidic protein (GFAP). Expression of VEGF mRNA was examined byin situ hybridization. • Results: One day after photocoagulation, the normal structure of the outer portion of the retina and the inner portion of the choroid was destroyed. Three days after photocoagulation, choroidal vascular endothelial cells migrated into the subretinal space through the defect in Bruch's membrane. Increased expression of VEGF was detected in the accumulating macrophages, migrating retinal pigment epithelial (RPE) cells and Muller cells. Maximal expression of VEGF was observed between 3 and 7 days after wounding, and many newly formed vessels extended into the subretinal space 7–14 days after photocoagulation. • Conclusion: VEGF derived from RPE cells, macrophages and Müller cells may play a role in the formation of CNV.
Similar content being viewed by others
References
Adams AP, Shima DT, Yeo K-T, Yeo T-K, Brown LF, Berse B, D'Amore PA, Folkman J (1993) Synthesis and secretion of vascular permeability factor/vascular endothelial growth factor by human retinal pigment epithelial cells. Biochem Biophys Res Commun 193:631–638
Aiello LP, Ferrara N, King GL (1994) Hypoxic regulation of vascular endothelial growth factor: characterization in retinal microvascular pericytes and pigment epithelial cells (abstract). Invest Ophthalmol Vis Sci 5:1868
Amin R, Puklin JE, Frank RN (1994) Growth factor localization in choroidal neovascular membranes of age-related macular degenerations. Invest Ophthalmol Vis Sci 35:3178–3188
Archer DB, Gardiner TA (1981) Electron microscopic features of experimental choroidal neovascularization. Am J Ophthalmol 91:433–457
Bignami A, Dahl D (1979) The radial glia of Müller in the rat retina and their response to injury. An immunofluorescence study with antibodies to the glial fibrillary acidic (GFA) protein. Exp Eye Res 28:63–69
Brown LF, Yeo K-T, Berse B, Yeo T-K, Senger DR, Dvorak HF, Van De Water L (1992) Expression of vascular permeability factor (vascular endothelial growth factor) by epidermal keratinocytes during wound healing. J Exp Med 176:1375–1379
Clauss M, Gerlach M, Gerlach H, Brett J, Wang F, Familletti PC, Pan YCE, Olander JV, Connolly DT, Stern D (1990) Vascular permeability factor: a tumor-derived polypeptide that induces endothelial cells and monocyte procoagulant activity, and promotes monocyte migration. J Exp Med 172:1535–1545
Ferrara N, Henzel WJ (1989) Pituitary follicular cells secrete a novel heparin-binding growth factor specific for vascular endothelial cells. Biochem Biophys Res Commun 161:851–858
Finkenzeller G, Marmé D, Weich HA, Hug H (1992) Platelet-derived growth factor-induced transcription of the vascular endothelial growth factor gene is mediated by protein kinase C1. Cancer Res 52:4821–4823
Gass JDM (1967) Pathogenesis of disciform detachment of the neuroepithelium. I. General concepts and classification. Am J Ophthalmol 573:1–13
Green WR, Wilson DJ (1986) Choroidal neovascularization. Ophthalmology 93:1169–1176
Guérin CJ, Anderson DH, Fisher SK (1990) Changes in intermediate filament immunolabeling occur in response to retinal detachment and reattachment in primates. Invest Ophthalmol Vis Sci 31:1474–1482
Hata Y, Nakagawa K, Ishibashi T, Inomata H, Ueno H, Sueishi K (1995) Hypoxia-induced expression of vascular endothelial growth factor by retinal glial cells promotes in vitro angiogenesis. Virchows Arch 426:479–486
Ishibashi T, Miller H, Orr G, Sorgente N, Ryan SJ (1987) Morphologic observations on experimental subretinal neovascularization in the monkey. Invest Ophthalmol Vis Sci 28:1116–1130
Ishibashi T, Inomata H, Sakamoto T, Ryan SJ (1995) Pericytes of newly formed vessels in experimental subretinal neovascularization. Arch Ophthalmol 113:227–231
Jakeman LB, Winer J, Bennett GL, Altar A, Ferrara N (1992) Binding sites for vascular endothelial growth factor are localized on endothelial cells in adult rat tissues. J Clin Invest 89:244–253
Jaye M, Howk R, Burgess W, Ricca GA, Chin I-M, Ravera MW, O'Brien SJ, Modi WS, Maciag T, Drohan WN (1986) Human endothelial cell growth factor: cloning, nucleotide sequence, and chromosome localization 233:541–545
Leung DW, Cachianes G, Kuang W-J, Goeddel DV, Ferrara N (1989) Vascular endothelial growth factor is a secreted angiogenic mitogen. Science 246:1306–1309
Malecaze F, Clamens S, Simorre-Pinatel V, Mathis A, Chollet P, Favard C, Bayard F, Plouet J (1994) Detection of vascular endothelial growth factor messenger RNA and vascular endothelial growth factor-like activity in proliferative diabetic retinopathy. Arch Ophthalmol 112:1476–1482
Miller H, Miller B, Ryan SJ (1986) Newly-formed subretinal vessles: fine structure and fluorescein leakage. Invest Ophthalmol Vis Sci 27:204–213
Miller H, Miller B, Ryan SJ (1986) The role of retinal pigment epithelium in the involution of subretinal neovascularization. Invest Ophthalmol Vis Sci 27:1644–1652
Miller JW, Adamis AP, Shima DT, D'Amore PA, Moulton RS, O'Reilly MS, Folkman J, Dvorak HF, Brown LF, Berse B, Yeo T-K, Yeo K-T (1994) Vascular endothelial growth factor/vascular permeability factor is temporally and spatially correlated with ocular angiogenesis in a primate model. Am J Pathol 145:574–584
Murata T, Ishibashi T, Khalil A, Hata Y, Yoshikawa H, Inomata H (1995) Vascular endothelial growth factor plays a role in hyperpermeability of diabetic retinal vessels. Ophthalmic Res 27:48–52
Okada M, Matsumura M, Ogino N, Honda Y (1990) Müller cells in detached human retina express glial fibrillary acidic protein and vimentin. Graefes Arch Clin Exp Ophthalmol 228:467–474
Pertovaara L, Kaipainen A, Mustonen T, Orpana A, Ferrara N, Saksela O, Alitalo K (1994) Vascular endothelial growth factor is induced in response to transforming growth factor-β in fibroblastic and epithelial cells. J Biol Chem 269:6271–6274
Pierce EA, Avery RL, Foley ED, Aiello LP, Smith LEH (1995) Vascular endothelial growth factor/vascular permeability factor expression in a mouse model of retinal neovascularization. Proc Natl Acad Sci USA 92:905–909
Ryan SJ (1979) The development of an experimental model of subretinal neovascularization in disciform macular degeneration. Trans Am Ophthalmol Soc 77:707–745
Ryan SJ (1982) Subretinal neovascularization: natural history of an experimental model. Arch Ophthalmol 100:1804–1809
Sato Y, Rifkin DB (1989) Inhibition of endothelial cell movement by pericytes and smooth muscle cells: activation of a latent transforming growth factor-β1-like molecule by plasmin during co-culture. J Cell Biol 109:309–315
Schweigerer L, Malerstein B, Neufeld G, Gospodarowicz D (1987) Basic fibroblast growth factor is synthesized in cultured retinal pigment epithelial cells. Biochem Biophys Res Commun 143:934–940
Schweigerer L, Neufeld G, Friedman J, Abraham JA, Fiddes JC, Gospodarowicz D (1987) Capillary endothelial cells express basic fibroblast growth factor, a mitogen that promotes their own growth. Nature 325:257–259
Sehested M, Hou-Jensen K (1981) Factor VIII related antigen as an endothelial cell marker in benign and malignant diseases. Virchows Arch (Pathol Anat) 391:217–225
Shen H, Clauss M, Ryan J, Schmidt A-M, Tijburg P, Borden L, Connolly D, Stern D, Kao J (1993) Characterization of vascular permeability factor/vascular endothelial growth factor receptors on mononuclear phagocytes. Blood 81:2767–2773
Shweiki D, Itin A, Soffer D, Keshet E (1992) Vascular endothelial growth factor induced by hypoxia may mediate hypoxia-initiated angiogenesis. Nature 359:843–845
Simorre-Pinatel V, Guerrin M, Chollet P, Penary M, Clamens S, Malecaze F, Plouet J (1994) Vasculotropin-VEGF stimulates retinal capillary endothelial cells through an autocrine pathway. Invest Ophthalmol Vis Sci 35:3393–3400
Stavri GT, Zachary IC, Baskerville PA, Martin JF, Erusalimsky JD (1995) Basic fibroblast growth factor upregulates the expression of vascular endothelial growth factor in vascular smooth muscle cells: synergistic interaction with hypoxia. Circulation 92:11–14
Sunderkötter C, Steinbrink K, Goebeler M, Bhardwaj R, Sorg C (1994) Macrophages and angiogenesis. J Leukoc Biol 55:410–422
Tanihara H, Yoshida M, Matsumoto M, Yoshimura N (1993) Identification of transforming growth factor-β expressed in cultured human retinal pigment epithelial cells. Invest Ophthalmol Vis Sci 34:413–419
Tassignon M-J, Stempels N, Nguyen-Legros J, De Wilde F, Brihaye M (1991) The effect of wavelength on glial fibrillary acidic protein immunoreactivity in laser-induced lesions in rabbit retina. Graefe's Arch Clin Exp Ophthalmol 229:380–388
Teeters VW, Bird AC (1973) A clinical study of the vascularity of senile disciform macular degeneration. Am J Ophthalmol 75:53–65
Yoshimura N, Matsumoto M, Shimizu H, Mandai M, Hata Y, Ishibashi T (1995) Photocoagulated human retinal pigment epithelial cells produce an inhibitor of vascular endothelial cell proliferation. Invest Ophthalmol Vis Sci 36:1686–1691
Wallow IH (1984) Repair of the pigment epithelial barrier following photocoagulation. Arch Ophthalmol 102:126–135
Wells JA, Murthy R, Chibber R, Nunn A, Molinatti PA, Kohner EM, Gregor ZJ (1996) Levels of vascular endothelial growth factor are elevated in the vitreous of patients with subretinal neovascularization. Br J Ophthalmol 80:363–366
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ishibashi, T., Hata, Y., Yoshikawa, H. et al. Expression of vascular endothelial growth factor in experimental choroidal neovascularization. Graefe's Arch Clin Exp Ophthalmol 235, 159–167 (1997). https://doi.org/10.1007/BF00941723
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00941723