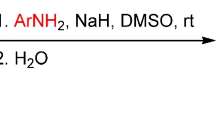
Conclusions
The reaction of 3-nitro-4,5-dihydro-6H-1,2-oxazine N-oxide with acetylenes in methanol is accompanied by cleavage of the nitro group and leads to the formation of theα-methoxy-3-substituted 4,5-dihydro-6H-1,2-oxazine derivatives.
Similar content being viewed by others
Literature cited
V. A. Tartakovskii, O. A. Luk'yanov, and S. S. Novikov, Izv. Akad. Nauk SSSR, Ser. Khim., 2246 (1966).
V. A. Tartakovskii, O. A. Luk'yanov, and S. S. Novikov, Dokl. Akad. Nauk SSSR,178, 123 (1968).
I. E. Chlenov, I. L. Sokolova, S. S. Novikov, and V. A. Tartakovskii, Izv. Akad. Nauk SSSR, Ser. Khim., 473 (1973).
P. S. Lauterbur, J. Chem. Phys.,26, 217 (1957).
G. Lery and G. Nelson, J. Am. Chem. Soc.,94, 4897 (1972).
K. S. Dhami and J. Stothers, Can. J. Chem.,43, 479 (1965).
P. Demarco, D. Doddrell, and E. Wenkevt, Chem. Commun., 1418 (1969).
D. Marr and J. Stothers, Can. J. Chem.,43, 596 (1965).
Determination of Organic Structures by Physical Methods, Vol. 2, Academic Press, New York (1962), p. 490.
H. Spisecke and W. Schneider, J. Chem. Phys.,35, 722 (1961).
A. Jones, M. Winkley, and G. Grant, Tetrahedron Lett., 5197 (1969).
D. Dorman and J. Roberts, J. Am. Chem. Soc.,92, 1355 (1970).
I. E. Chlenov, V. I. Khudak, L. N. Kolymagina, N. S. Morozova, and V. A. Tartakovskii, Izv. Akad. Nauk SSSR, Ser. Khim., 1867 (1970).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 2, pp. 382–386, February, 1974.
Rights and permissions
About this article
Cite this article
Chlenov, I.E., Sokolova, I.L., Khasapov, B.N. et al. Reaction of 3-nitro-4,5-dihydro-6H-1,2-oxazine N-oxide with acetylenes. Russ Chem Bull 23, 347–351 (1974). https://doi.org/10.1007/BF00924685
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00924685