Abstract
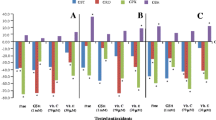
Four chromium salts with different oxidation states were tested for their influence in causing chromosome aberrations and sister-chromatid exchange in Chinese hamster ovary cellsin vitro. Cell cultures were treated with CrO3, K2Cr2O7, CrCl2 and Cr(NO3)3.9H2O at concentrations of 10−7, 10−6, 10−5 and 10−4 M for the aberration assay, and 10−8, 10−7, 10−6 and 10−5 M for the sister-chromatid exchange assay. It was noticed that Cr (VI) compounds-CrO3 and K2Cr2O7-considerably enhanced the frequencies of aberrations and sister-chromatid exchanges compared to the control cultures. CrCl2 and Cr(NO3)3.9H2O−Cr (II) and Cr (III) respectively-caused a slight increase in sister-chromatid exchange rates, but the frequencies of aberrations were almost unchanged compared to the controls. These investigations indicate a definite link between the metals and changes produced in the mammalian chromosomes, reaffirming the evidence of carcinogenic potential of Cr (VI) observed by other investigators.
Similar content being viewed by others
Abbreviations
- BrdU:
-
5-bromo-2′-deoxyuridine
- CHO:
-
Chinese hamster ovary
- SCE:
-
sister-chromatid exchange
References
Andersen O (1983) Effects of coal combustion products and metal compounds on sister-chromatid exchange (SCE) in a macrophage-like cell line. Environ. Health Prospect. 47: 239–253.
Costa M, Kraker AJ and Patierno SR (1984) Toxicity and carcinogenicity of essential and non-essential metals. Prog. Clin. Biochem. 1: 1–49.
De Flora S, Bagansco M, Serra D and Zanacchi P (1990) Genotoxicity of chromium compounds. A review. Mutation Res. 238: 99–172.
De Flora S and Wetterhahn KE (1989) Mechanisms of chromium metabolism and genotoxicity. Life Chem Rep. 7: 169–244.
Gainaldi G, Colella CM, Piras A and Mariani T (1982) Thioguanine resistance, ouabain resistance and sister-chromatid exchanges in V79/AP4 Chinese hamster cells treated with potassium dichromate. Chem-Biol. Interact. 42: 45–51.
Goto K, Maeda S, Kano Y and Sugiyama T (1978) Factors involved in differential Giemsa-staining of sister chromatids. Chromosoma 66: 351–359.
Huper WC and Payne WW (1962) Experimental studies in metal carcinogenesis. Chromium, nickel, iron, arsenic. Arch. Environ. Hlth. 5: 445–462.
Jennette KW (1979) Chromate metabolism in liver microsomes. Biol. Trace Elem. Rev. 1: 55–62.
Kawanishi S, Inoue S and Sano S (1986) Mechanism of DNA cleavage induced by sodium chromate (VI) in the presence of hydrogen peroxide. J. Biol. Chem. 261: 5952–5958.
Kochhar TS (1982) Effect of polycyclic hydrocarbons on the induction of chromosomal aberrations in absence of an exogenous metabolic activation system in cultured hamster cells. Experientia 38: 845–846.
Kochhar TS (1985) Inducibility of chromosome aberrations by steroid hormones in cultured Chinese hamster ovary cells. Toxicol. Lett. 29: 201–206.
Miller EC and Miller JA (1979) Overview on the relevance of naturally occurring carcinogens, promoters, and modulators of carcinogenesis in human cancer. In: EC Miller et al. (eds.) Naturally Occurring Carcinogens, Mutagens and Modulators of Carcinogenesis. (pp. 1–17), Japan Scientific Society Press, Baltimore.
Newton MF and Lilly LJ (1986) Tissue-specific clastogenic effects of chromium and selenium saltsin vitro. Mutation Res. 169: 61–69.
Nishioka H (1975) Mutagenic activities of metal compounds in bacteria. Mutation Res. 31: 185–189.
Paschin YV, Kozacyhenko VI and Sal'nikova LE (1983) Differential mutagenic response at HGPRT locus in V79 and CHO cells after treatment with chromate. Mutation Res. 122: 361–365.
Perry P and Wolff S (1974) New Giemsa method for differential staining of sister chromatids. Nature 251: 156–158.
Rodney PF, Robert JJ, Lay PA, Dixon NE, Raker RSU and Bonin AM (1989) Chromium (V)-induced cleavage of DNA: Are chromium (V) complexes the active carcinogens in chromium (VI)-induced cancer? Chem. Res. Toxicol. 2: 227–229.
Sen P, Conway K and Costa M (1987) Comparison of the localization chromosome damage induced by calcium chromate and nickel compounds. Cancer Res. 47: 2142–2147.
Shi X and Dalal NS (1988) The mechanism of chromate reduction by glutathione: ESR evidence for the glutathionyl radical and isolabel Cr (V) intermediate. Biochem. Biophys. Res. Commun. 156: 137–142.
Shi X and Dalal NS (1989) Chromium (V) and hydroxyl radical formation during glutathione reductase-catalyzed reduction of chromium (VI). Biochem. Biophys. Res. Commun. 163: 627–634.
Sirover MA and Loeb LA (1976) Infidelity of DNA synthesisin vitro: screening for potential metal mutagens or carcinogens. Science 194: 1434–1436.
Steel RGD and Torrie JH (1980) Principles and Procedures of Statistics. McGraw-Hill, Inc. New York, NY.
Sugiyama M, Wang Xin-Wei and Costa M (1986a) Comparison of DNA lesions and cytotoxicity induced by calcium chromate in human, mouse and hamster cell lines. Cancer Res. 46: 4547–4551.
Sugiyama M, Patierno SR, Cantoni O and Costa M (1986b) Characterization of DNA lesions induced by CaCrO4 in synchronous and asynchronous cultured mammalian cells. Molecular Pharm 29: 606–613.
Sunderman FW Jr (1977) Metal carcinogenesis. In: Goyer RA and Mehlman MA (eds.) Toxicology of Trace Elements. (pp. 257–295) John Wiley, New York.
Sunderman FW Jr (1984) Recent progress in nickel carcinogenesis. In: Merian E, Frei RW, Hardi W, and Schlatter C (eds.) Carcinogenic and Mutagenic Metal Compounds. (pp. 325–342) Gordon and Breach, London.
Tsuda H and Kato K (1977) Chromosomal aberrations and morphological transformation in hamster embryonic cells treated with potassium dichromatein vitro. Mutation Res. 46: 87–94.
Umeda M and Nishimura M (1979) Inducibility of chromosomal aberrations by metal compounds in cultured mammalian cells. Mutation Res. 67: 221–229.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kochhar, T.S., Howard, W.R. Effect of different oxidation states of chromium in causing chromosome alterations in cultured CHO cells. Cytotechnology 11, 183–188 (1993). https://doi.org/10.1007/BF00749868
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00749868