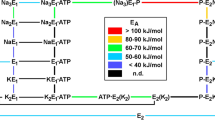
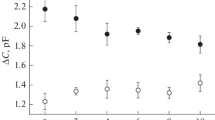
Abstract
A particulate (Na + K)-ATPase preparation from dog kidney bound [48V]-ortho-vanadate rapidly at 37°C through a divalent cation-dependent process. In the presence of 3 mM MgCl2 theK d was 96 nM; substituting MnCl2 decreased theK d to 12 nM but the maximal binding remained the same, 2.8 nmol per mg protein, consistent with 1 mol vanadate per functional enzyme complex. Adding KCl in the presence of MgCl2 increased binding, with aK 0.5 for KCl near 0.5 mM; the increased binding was associated with a drop inK d for vanadate to 11 nM but with no change in maximal binding. Adding NaCl in the presence of MgCl2 decreased binding markedly, with anI 50 for NaCl of 7 mM. However, in the presence of MnCl2 neither KCl nor NaCl affected vanadate binding appreciably. Both the nonhydrolyzable, β,γ-imido analog of ATP and nitrophenyl phosphate, a substrate for the K-phosphatase reaction that this enzyme also catalyzes, decreased vanadate binding at concentrations consistent with their acting at the low-affinity substrate site of the enzyme; the presence of KCl increased the concentration of each required to decrease vanadate binding. Oligomycin decreased vanadate binding in the presence of MgCl2, whereas dimethyl sulfoxide and ouabain increased it. With inside-out membrane vesicles from red blood cells vanadate inhibited both the K-phosphatase and (Na + K)-ATPase reactions; however, with the K-phosphatase reaction extravesicular K+ (corresponding to intracellular K+) both stimulated catalysis and augmented vanadate inhibition, whereas with the (Na + K)-ATPase reaction intravesicular K+ (corresponding to extracellular K+) both stimulated catalysis and augmented vanadate binding.
Similar content being viewed by others
References
Beauge', L. A. (1979). InNa, K-ATPase: Structure and Kinetics J. C. Skou and J. G. Norby, eds., Academic Press, London, pp. 373–387.
Beauge', L. A., and Glynn, I. M. (1978).Nature 272 551–552.
Beauge', L. A., and Glynn, I. M. (1979).Nature 280 510–512.
Beauge', L. A., Cavieres, J. J., Glynn, I. M., and Grantham, J. J. (1980).J. Physiol. 301 7–23.
Blostein, R., Pershadsingh, H. A., Drapeau, P., and Chu, L. (1979). InNa,K-ATPase: Structure and Kinetics J. C. Skou and J. G. Norby, eds., Academic Press, London, pp. 233–245.
Bond, G. H., and Hudgins, P. M. (1979).Biochemistry 18 325–331.
Cantley, L. C., Jr., Cantley, L. G., and Josephson, L. (1978).J. Biol. Chem. 253 7361–7368.
Fahn, S., Koval, G. J., and Albers, R. W. (1966).J. Biol. Chem. 241 1882–1889.
Glynn, I. M., and Karlish, S. J. D. (1975).Annu. Rev. Physiol. 37 13–55.
Grisham, C. M., and Mildvan, A. S. (1974).J. Biol. Chem. 249 3187–3197.
Karlish, S. J. D., Yates, D. W., and Glynn, I. M. (1978).Biochim. Biophys. Acta 525 252–264.
Jorgensen, P. L. (1974).Biochim. Biophys. Acta 356 26–52.
Liang, S. M., and Winter, C. G. (1976).Biochim. Biophys. Acta 452 552–565.
Mercer, R. W., and Dunham, P. B. (1981).J. Gen. Physiol., in press.
Post, R. L., Hegyvary, C., and Kume, S. (1972).J. Biol. Chem. 247 6530–6540.
Robinson, J. D. (1967).Biochemistry 6 3250–3258.
Robinson, J. D. (1969).Biochemistry 8 3348–3355.
Robinson, J. D. (1975a).Biochim. Biophys. Acta 384 250–264.
Robinson, J. D. (1975b).Biochim. Biophys. Acta 397 194–206.
Robinson, J. D. (1976a).Biochim. Biophys. Acta 429 1006–1019.
Robinson, J. D. (1976b).Biochim. Biophys. Acta 440 711–722.
Robinson, J. D. (1980a).Biochim. Biophys. Acta 598 543–553.
Robinson, J. D. (1980b).J. Bioenerg. Biomembr. 12 165–174.
Robinson, J. D. (1981).Biochim. Biophys. Acta 642 405–417.
Robinson, J. D., and Flashner, M. S. (1979).Biochim. Biophys. Acta 549 145–176.
Smith, R. L., Zinn, K., and Cantley, L. C. (1980).J. Biol. Chem. 255 9852–9859.
Steck, T. L. (1974).Methods Membr. Res. 2 245–281.
Swann, A. C., and Albers, R. W. (1978).Biochim. Biophys. Acta 523 215–227.
Tobin, T., Akera, T., Baskin, S. I., and Brody, T. M. (1973).Mol. Pharmacol. 9 336–349.
Yamaguchi, M., and Tonomura, Y. (1980a).J. Biochem. 88 1365–1375.
Yamaguchi, M., and Tonomura, Y. (1980b).J. Biochem. 88 1377–1385.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Robinson, J.D., Mercer, R.W. Vanadate binding to the (Na + K)-ATPase. J Bioenerg Biomembr 13, 205–218 (1981). https://doi.org/10.1007/BF00743200
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00743200