Summary
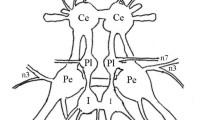
Intracellular recordings have been made from the somata of two metathoracic flight motoneurons, one innervating an elevator muscle of the hindwing, the tergosternal muscle 113 and the other a depressor, the first basalar muscle 127. The locust,Ghortoicetes terminifera was mounted ventral side uppermost with the thorax restrained and opened for access to the thoracic ganglia. Patterns of electrical activity recorded from the thoracic muscles were similar to those shown by a locust during flight when tethered in a more normal posture. In flight the left and right 113 motoneurons each receive a single impulse together at every stroke of the wing, with the 127 muscles active in approximate antiphase. A spike in a 113 motoneuron causes a delayed wave of excitation simultaneously upon itself and its contralateral partner (Fig. 2). The epsp's which form these waves summate and may cause a spike which follows the original one with a delay equal to the wingbeat period. The delayed excitation of the contralateral motoneuron is of larger amplitude than the ipsilateral one so that spikes in either motoneuron must activate separate but symmetrical pathways. A single spike may cause multiple waves in either motoneuron, each separated by intervals equal to the wingbeat period (Fig. 3). In the pathway must be neurons capable of reverberation.
A spike in a 113 motoneuron causes a delayed excitation of the ipsilateral 127 motoneuron so that its membrane potential is lowered antiphasically to that of 113 (Fig. 17). A spike in a 127 motoneuron has no effect on the 113 motoneurons. In flight these pathways causing delayed excitation may co-ordinate the motoneurons.
The left and right 113 motoneurons receive common synaptic inputs from at least two sources (Fig. 8). These occur as bursts of epsp's at intervals approximately equal to or multiples of the wingbeat period and in the absence of flight. Epsp's of sufficient amplitude cause a spike in the motoneuron which is in the correct phase in the flight pattern relative to any other active motoneurons (Fig. 9). During sustained flight epsp's contribute to the wave of depolarization that the motoneuron undergoes at each wingbeat (Fig. 11). In the absence of the epsp's the motoneuron does not oscillate on its own. At the end of flight bursts of epsp's may continue at the flight frequency long after all activity in the muscles has ceased.
Similar content being viewed by others
References
Bentley, D. R.: Intracellular activity in cricket neurons during the generation of behaviour patterns. J. Insect Physiol.15, 677–699 (1969).
Burrows, M.: The morphology of an elevator and a depressor motoneuron of the hindwing of a locust. J. comp. Physiol.83, 165–178 (1973).
Burrows, M., Hoyle, G.: Neural mechanisms underlying behavior of the locustSchistocerca gregaria. III. Topography of limb motoneurons in the metathoracic ganglion. J. Neurobiol. (in press) (1973).
Hoyle, G., Burrows, M.: Intracellular studies on identified neurons of insects. Fed. Proc.29, 589 (Abstract), (1970).
Hoyle, G., Burrows, M.: Neural mechanisms underlying behavior in the locustSchistocerca gregaria. I. Physiology of identified neurons in the methathoracic ganglion. J. Neurobiol. (in press) (1973).
Iwasaki, S., Wilson, D. M.: Neuropile potentials following wing sensory nerve stimulation in locusts. Comp. Biochem. Physiol.17, 693–695 (1966).
Kendig, J. J.: Motor neurone coupling in locust flight. J. exp. Biol.48, 389–404 (1968).
Mulloney, B., Selverston, A. I.: Antidromic action potentials fail to demonstrate known interactions between cells. Science177, 69–72 (1972).
Page, C. H.: Unit responses in the metathoracic ganglion of the flying locust. Comp. Biochem. Physiol.37, 565–571 (1970).
Pitman, R. M., Tweedle, C. D., Cohen, M. J.: Branching of central neurons: intracellular cobalt injection for light and electron microscopy. Science176, 412–414 (1972).
Pond, C. M.: Neuromuscular events and wing movements at the start of flight ofPeriplaneta americana andSchistocerca gregaria. J. comp. Physiol.78, 192–209 (1972).
Snodgrass, R. E.: The thoracic mechanism of a grasshopper, and its antecedents. Smithson. misc. Colln.82 (2), 1–111 (1929).
Usherwood, P. N. R., Grundfest, H.: Peripheral inhibition in skeletal muscle of insects. J. Neurophysiol.28, 497–518 (1965).
Waldron, I.: Mechanisms for the production of the motor output pattern in flying locusts. J. exp. Biol.47, 201–212 (1967).
Waldron, I., Wilson, D. M.: Latency relationships between loosely co-ordinated locust flight motor neurons. J. exp. Zool.170, 293–300 (1969).
Wilson, D. M.: The central nervous control of flight in a locust. J. exp. Biol.38, 471–490 (1961).
Wilson, D. M.: Central nervous mechanisms for the generation of rhythmic behaviour in Arthropods. Symp. Soc. exp. Biol.20, 199–228 (1966).
Wilson, D. M.: The nervous control of insect flight and related behavior. Adv. Insect Physiol.5, 289–338 (1968).
Wilson, D. M.: Neural operations in Arthropod ganglia. In: The Neurosciences. Second study program (F. O. Schmitt, ed.), p. 397–409. New York: Rockefeiler University Press 1970.
Wilson, D. M., Gettrup, E.: A stretch reflex controlling wingbeat frequency in grasshoppers. J. exp. Biol.40, 171–185 (1963).
Wilson, D. M., Waldron, I.: Models for the generation of the motor output pattern in flying locusts. Proc. I.E.E.E.56, 1058–1064 (1968).
Wilson, D. M., Weis-Fogh, T.: Patterned activity of co-ordinated motor units studied in flying locusts. J. exp. Biol.89, 643–667 (1962).
Author information
Authors and Affiliations
Additional information
Beit Memorial Research Fellow.
Rights and permissions
About this article
Cite this article
Burrows, M. The role of delayed excitation in the co-ordination of some metathoracic flight motoneurons of a locust. J. Comp. Physiol. 83, 135–164 (1973). https://doi.org/10.1007/BF00696892
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00696892