Summary
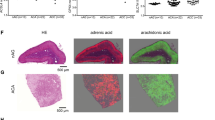
Mitotane is an important adrenalytic drug for the treatment of adrenal cancer whose use is limited by toxicity. Reports from another laboratory indicated that a methylated homolog of Mitotane (Mitometh) tested in guinea pigs possessed comparable adrenalytic activity but was less toxic than Mitotane. This observation prompted us to undertake a comparative study of these two drugs on the basis that Mitometh may be a superior agent for the treatment of adrenal cancer. Preliminary studies in guinea pigs failed to show a significant adrenalytic effect for either Mitotane or Mitometh. Thus, we extended the study to 13 mongrel dogs weighing 12–15 kg that were treated daily with Mitometh or Mitotane (50–100 mg/kg) for 6 or 12 days. Cortisol decreased to undetectable levels and adrenocorticotropic hormone (ACTH) rose to 10 times the baseline levels within 72 h in Mitotane-treated animals. Despite the achievement of similar drug levels, Mitometh treatment in dogs failed to suppress cortisol or increase ACTH. To determine whether these differences were due to differences in bioavailability, we measured the relative concentration of Mitotane and Mitometh in homogenates of adrenal cortex obtained from Mitotane- and Mitometh-treated dogs. The adrenal concentration of Mitometh determined in Mitometh-treated dogs was 5 times higher than the concentration of Mitotane measured in Mitotane-treated animals. Whereas the adrenal glands of Mitotane-treated dogs showed hemorrhage and necrosis, the Mitometh-treated animals showed no adrenal damage. Despite the lack of adrenalytic activity, Mitometh maintained its toxicity as demonstrated by microscopic evidence of hepatic necrosis and an increase in hepatic enzymes. The adrenalytic effects of both agents was also studied in vitro using a human functioning adrenal cortical carcinoma cell line. NCI-H295. Whereas Mitotane strongly suppressed cell growth, Mitometh had a weaker effect. We conclude that Mitometh is not likely to be effective in the therapy of adrenal cancer. Moreover, the results of this study are supportive of the view that metabolic transformation of Mitotane is in some way linked to its adrenalytic action.
Similar content being viewed by others
References
Bleiberg MJ, Larson PS (1957) Studies on derivatives of 2,2bis-(p-chlorophenyl)-1,1-dichloroethane (DDD, TDE) with special reference to their effects on the adrenal cortex. J Pharmacol Exp Ther 121: 421–431
Cobey FA, Taliaferro I, Haag HB (1956) Effect of DDD and some of its derivatives on plasma 17-OH corticosteroids in the dog. Science 123: 140–141
Cobey FA, Taliaferro I, Haag HB (1958) Further observations on effect on plasma 17-OH corticosteroids in the dog of derivatives of 2,2-bis-(p-chlorophenyl)-1,1-dichloroethane (DDD, TDE). Proc Soc Exp Biol Med 97: 491–494
Cohen EN, Trudell JR, Edmunds HN, Watson E (1975) Urinary metabolites of halothane in man. Anesthesiology 43: 392–401
Copeland MF, Cranmer MF (1974) Effects ofo,p′-DDT on the adrenal gland and hepatic microsomal enzyme system in the beagle dog. Toxicol Appl Pharmacol 27: 1–10
Cueto C, Brown JHV, Richardson AP (1958) Biological studies of an adrenocorticolytic agent and the isolation of the active components. Endocrinology 62: 334–339
Fang VS (1979) Cytotoxic activity of 1-(o-chlorophenyl)-1-(p-chlorophenyl)-2,2-dichloroethane (Mitotane) and its analogs on feminizing adrenal neoplastic cells in culture. Cancer Res 39: 139–145
Finnegan JK, Henningar GR, Smith RB, Larson PS, Haag HB (1955) Acute and chronic toxicity studies on 2,2-bis-(p-ethylphenyl)-1,1-dichloroethane (Perthane). Arch Int Pharmacodyn Ther 103: 404–418
Gazdar AF, Oie HK, Shackleton CH (1990) Establishment and characterization of a human adrenocortical carcinoma cell line that expresses multiple pathways of steroid biosynthesis. Cancer Res 50: 5488–5496
Gold B, Brunk G (1984) A mechanistic study of the metabolism of 1,1 dichloro-2,2-bis (p-chlorophenyl) ethane (DDD) to 2,2-bis (p-chlorophenyl) acetic acid (DDA). Biochem Pharmacol 33: 979–982
Hart M, Reagan RL, Adamson RH (1973) The effect of isomers of DDD on the ACTH-induced steroid output, histology and ultrastructure of the dog adrenal cortex. Toxicol Appl Pharmacol 24: 101–113
Hunter WM, Greenwood FC (1987) Preparation of iodine-131 labeled human growth hormone of high specific activity. Nature 194: 495–496
Jensen BL, Caldwell MW, French LG, Briggs DG (1987) Toxicity, ultrastructural effects and metabolic studies with 1-(o-chlorophenyl)-1-(p-chlorophenyl)-2,2-dichloroethane (o,p′-DDD) and its methyl analog in the guinea pig and rat. Toxicol Appl Pharmacol 87: 1–9
Larson PS, Hennigan GR, Finnegan JK, Blackwell Smith R, Haag HB (1955) Observations on the relation of chemical structure to the production of adrenal cortical atrophy or hypertrophy in the dog by derivatives of DDD, TDE. J Pharmacol Exp Ther 115: 408–412
Martz F, Straw JA (1980) Metabolism and covalent binding of 1-(o-chlorophenyl)-1-(p-chlorophenyl)-2,2-dichloroethane (o,p′-DDD). Correlation between adrenocorticolytic activity and metabolic activation by adrenocortical mitochondria. Drug Metab Dispos 8: 127–130
Nelson AA, Woodard G (1949) Severe adrenal cortical atrophy (cytotoxic) and hepatic damage produced in dogs by feeding 2,2-bis (parachlorophenyl)-1,1-dichloroethane (DDD or TDE). Arch Pathol 48: 387–394
Nichols J (1961) Studies on an adrenal cortical inhibitor. In: Moon HD (ed) The adrenal cortex. Paul B. Hoeber, New York, pp 84–107
Nichols J, Prestley WF, Nichols F (1961) Effects ofm,p′-DDD in a case of adrenal cortical carcinoma (a preliminary report). Curr Ther Res 3: 266–271
Nichols J, Warren JC, Mantz FA (1962) ACTH-like excretion from carcinoma of the ovary. The clinical effects ofm,p′-DDD. JAMA 182: 713–718
Pohl LR, Krishna G (1978) Study of the mechanism of metabolic activation of chloramphenicol by rat liver microsomes. Biochem Pharmacol 27: 335–341
Reif D, Sinsheimer JE (1975a) Metabolism of 1-(o-chlorophenyl)-1-(p-chlorophenyl)-2,2 dichloroethane (o,p′-DDD) in rats. Drug Metab Dispos 3: 15–25
Reif VD, Sinsheimer JE (1975b) In vitro biotransformations ofo,p′-DDD andp,p′-DDD by bovine adrenal. J Agric Food Chem 23: 996–999
Reif VD, Sinsheimer JE, Ward JC, Schteingart DE (1974) Aromatic hydroxylation and alkyl oxidation in the metabolism of Mitotane (o,p′-DDD) in humans. J Pharm Sci 63: 1730–1736
Schteingart DE, Tsao HS, Taylor CI, McKenzie AK, Victoria RS, Therrien B (1980) Sustained remission of Cushing's disease with Mitotane and pituitary irradiation. Ann Intern Med 92: 613–619
Sinsheimer JE, Freeman CJ (1987) Mitotane [1-(o-chlorophenyl)-1-(p-chlorophenyl)-2,2-dichloroethane] metabolism in perfusion studies with dog adrenal glands. Drug Metab Dispos 15: 267–269
Skehan P, Storeng R, Scudiero D, Monks A, McMahon J, Vistica D, Warren JT, Bokesch H, Kenney S, Boyd MR J (1990) New colorimetric cytotoxicity assay for anticancer-drug screening. J Natl Cancer Inst 82: 1107–1112
Vague P, Oliver C, Jaquet P, Vague J (1971) Le dosage radioimmunologique de l'ACTH plasmatique: resultats chez le sujets normaux. Eur J Clin Biol Res 16: 485–493
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schteingart, D.E., Sinsheimer, J.E., Counsell, R.E. et al. Comparison of the adrenalytic activity of mitotane and a methylated homolog on normal adrenal cortex and adrenal cortical carcinoma. Cancer Chemother. Pharmacol. 31, 459–466 (1993). https://doi.org/10.1007/BF00685036
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00685036